

Mithun Singh Rajput*, Rashmi Dahima
School of Pharmacy, Devi Ahilya Vishwavidyalaya, Takshashila Campus, Khandwa Road, Indore-452001, M.P., India
*Address for Correspondence
Dr. Mithun Singh Rajput,
Postdoctoral Fellow,
School of Pharmacy, Devi Ahilya Vishwavidyalaya, Takshashila Campus, Khandwa Road, Indore-452001, M.P., India.
Abstract
Background: α-Glucosidase is a membrane bound intestinal enzyme that transforms polysaccharides into simple absorbable monosaccharides and glucose within the gastrointestinal tract and thus absorbed glucose contributes to postprandial hyperglycemia. Therefore, inhibition of α-glucosidase is a prominent therapeutic strategy to control diabetes mellitus. Objective: In this study, an attempt has been made to investigate the ability of dimethyl cardamonin (DMC) to inhibit enzyme α-glucosidase as well as in combination with an antidiabetic drug, acarbose and to study inhibition kinetics of DMC. Material and Methods: The inhibitory effect of various dose of DMC (0.5–100.0μg/ml) on α-glucosidase was assessed using microtitre plate assay in vitro, so as to evaluate its antidiabetic potential. An enzyme kinetic analysis was performed and the type of inhibition was determined by Lineweaver-Burk plot obtained by plotting velocities of reaction and substrate concentrations reciprocally. Results: DMC exhibited satisfactory inhibitory activity against α-glucosidase with IC50 value 87.56μg/ml, which was much higher than acarbose (IC50 value of 262.39μg/ml). The Lineweaver–Burk plots for α-glucosidase inhibition by DMC indicated a noncompetitive enzyme inhibition. It was found that acarbose combined with DMC at all the three concentrations (¼IC50, ½IC50 and IC50) resulted in significant (p<0.05) inhibition compared with the individual compounds at the same concentration indicating synergistic inhibitory activity against α-glucosidase. Conclusion: The present study provides the first evidence that DMC could find application as α-glucosidase inhibitor. This study could facilitate effective utilization of DMC as an oral antidiabetic drug or functional food ingredient with a promising role in the formulation of medicines and nutrition supplements.
Keywords: α-glucosidase; chalcones; diabetes; dimethyl cardamonin
Introduction
Diabetes mellitus, a group of prolonged metabolic illness results from insulin resistance, inadequate insulin secretion, excessive or inappropriate glucagon secretion, or their combination, affects more than 400 million people and 1.5 million related deaths as of 2015 worldwide according to the International Diabetes Federation (Guariguata et al., 2014). Prolonged diabetes leads to severe ailments counting neuropathy, nephropathy, retinopathy and cardiovascular problems and enhances the threat of mortality (Asmat et al., 2016). Clinically existing oral antidiabetic medications employ their beneficial outcome by numerous mechanisms comprising promotion of insulin secretion and glucose absorption and metabolism. Drugs available to treat diabetes are insulin, dipeptidyl peptidase inhibitors, peroxisome proliferator-activated receptor agonists, sulfonylureas, α-glucosidase inhibitors and glucagon like peptide-1 analogs. However, therapeutic efficacies of these drugs are sometimes inadequate and reported to be associated with severe side effects e.g. weight gain, hypoglycemia, edema, lactic acidosis and gastrointestinal intolerance etc (Chaudhury et al., 2017). Hence, quest for novel safer antidiabetic medicines is a foremost stuff of research and drug discovery. An effective recommendation for the controlling of diabetes mellitus is the inhibition of enzyme, α-glucosidase (van de Laar, 2008).
α-Glucosidase is a membrane bound intestinal enzyme that converts polysaccharides into simple absorbable monosaccharides and glucose within the gastrointestinal tract and thus absorbed glucose contributes to postprandial hyperglycemia (Bell, 2004). Therefore, inhibition of α-glucosidase is a prominent therapeutic strategy to control postprandial hyperglycemia in type 2 diabetes. Acarbose, voglibose and miglitol are clinically available α-glucosidase inhibitors and given orally as monotherapy or in blend with other oral antidiabetic medications. This category of drugs imply ample side effects like meteorism, abdominal distention, diarrhea and other cardiac hazards (Fisman et al., 2008). Due to the limited effectiveness and accompanying undesirable side effects of available antidiabetic drugs, attention has been increased leading to the use of natural sources, especially medicinal plants for the alternative management of type 2 diabetes mellitus and some natural products like flavonoids are recognized to have well established α-glucosidase inhibitory property (Şohretoglu et al., 2017).
Chalcone is simply one of the many types of flavonoid phytochemical group. Dimethyl cardamonin or 2’, 4’-dihydroxy-6’-methoxy-3’, 5’-dimethylchalcone (DMC) has been isolated from many plant species like C. operculatus, M. serrata, S. samarangense and S. campanulatum etc (Memon et al., 2014). DMC is known to possess hepatoprotective, cytoprotective, anti-inflammatory, antifungal, antibacterial, antituberculosis, antiviral, antispasmodic, antidiarrhoeal, antiapoptotic, antitumor, antimicrobial and antioxidant effect (Yu et al., 2015). DMC exerts antidiabetic and antihypergycemic effect by protecting the impaired insulin secretion induced by glucotoxicity in pancreatic β-Cells. DMC also promoted glucose uptake and imposed a paradoxical effect on adipocyte differentiation in 3T3-L1 cells (Huet al., 2014). However, studies on α-glucosidase inhibitory property of DMC are lacking. Therefore, in this study, we investigated the ability of DMC to inhibit enzyme α-glucosidase in vitro as well as in combination with acarbose, an antidiabetic drug.
Materials and methods
Drugs and solutions
DMC (purity ≥98%) was isolated in our laboratory from green leaves of Syzygium campanulatum Korth as per the method described by Memon et al. (2014). α-Glucosidase from Saccharomyces cervisiae (EC 3.2.1.20), p-nitrophenyl-a-D-glucopyranoside (pNPG) and acarbose were purchased from Sigma Aldrich (Sigma-Aldrich Chemicals Pvt. Ltd. Bengaluru, India). All other drugs were dissolved in distilled water. The doses for all freshly prepared drug solutions were expressed in terms of their free bases.
α-Glucosidase inhibition assay of DMC
α-Glucosidase inhibitory activity was determined using the method described by Rengasamy et al. (2013) with slight modifications using a 96-well microtiter plate. Briefly, yeast α-glucosidase (0.1 unit/ml) was dissolved in 0.1M potassium phosphate buffer (pH 6.8), this was used as the enzyme solution. The substrate, 0.375 mM of pNPG was prepared in the same buffer (pH 6.8). Various sample of DMC and acarbose (0.5–100.0 μg/ml) were prepared in dimethylsulfoxide (DMSO). Each sample (20 μl) and enzyme solution (20 μl) were mixed in the microtiter plate. The reaction was initiated by adding 40 μl substrate. The reaction mixture was incubated at 37 ˚C for 40 min. After incubation, 80 μl (0.2M) sodium carbonate in 0.1M potassium phosphate buffer (pH 6.8) was added to each well to quench the reaction. The amount of p-nitrophenol (pNP) released was quantified using an 96-well microplate reader at 405 nm. The control experiment contained the same reaction mixture, but the sample solution was replaced with the same volume of phosphate buffer. Acarbose dissolved in DMSO, was used as a positive control. The determinations were carried out in triplicate. The percentage inhibition (%) was calculated by using the following equation:
% Inhibition = [(Acontrol - Asample)/Acontrol] ×100
Where, Acontrol is the absorbance of the control and Asample is the absorbance of the sample. The IC50, which is the concentration of the sample required to inhibit the enzyme was determined for each sample.
Determination of the mechanism of α-glucosidase inhibition by DMC
An enzyme kinetic analysis was performed based on the α-glucosidase inhibition assay described above. The concentration of α-glucosidase was kept constant at 0.1 unit/ml and the pNPG concentrations varied from 0.16 to 2.65 mM in the absence and presence of DMC (12.5, 25.0 and 50.0 μg/ml). The type of inhibition was determined by Lineweaver-Burk plot obtained by plotting velocities of reaction (vertical axis) and substrate concentrations (horizontal axis) reciprocally (Gu et al., 2009).
α-Glucosidase inhibition of DMC in combinations with acarbose
On the basis of IC50 data, a series of three concentrations (¼IC50, ½IC50 and IC50) of DMC as well as its combined mixtures with acarbose were prepared to investigate the combined inhibitory effect of the samples with the control drug on α-glucosidase (Gao et al., 2013). Reaction was performed according to above α-glucosidase inhibition assay.
Statistical analysis
Results were expressed as mean ± S.E.M. The data were analyzed by one way analysis of variance (ANOVA) tracked by Tukey's multiple comparison tests or Welch’s corrected t test. Regression analysis for calculating IC50 values was done using GraphPad Prism software (version 5.03). Probability values less than 0.05 were considered statistically significant in all the cases.
Results
α-Glucosidase inhibition assay of DMC
The inhibitory effect of DMC on α-glucosidase was assessed so as to evaluate its antidiabetic potential. Lower IC50 values show higher inhibition. DMC exhibited satisfactory inhibitory activity against α-glucosidase with IC50 value 87.56 μg/ml, that was much higher activity than acarbose (IC50 value of 262.39 μg/ml) [F(5, 5) = 9.794, p < 0.0001, R2 = 0.9922] (Table 1).
Table 1. α-Glucosidase inhibitory activity of DMC
|
Test sample |
IC50 value (μg/ml) |
R2 value |
|
DMC |
87.56 ± 1.93 |
0.9651 |
|
Acarbose |
262.39 ± 6.04* |
0.9824 |
Results are expressed as Mean ± S.E.M. (n = 3). Data was analyzed by one way repeat measure ANOVA followed by Welch’s corrected t test. Significance: *p< 0.0001 when compared with DMC group, DMC: dimethyl cardamonin. Lower IC50 values show higher inhibition.
Mechanism of α-glucosidase inhibition by DMC
To conclude the mechanism of inhibition, three different concentrations of DMC were used (12.5, 25.0 and 50.0 μg/ml) as inhibitors in kinetic experiment to reveal the kind of inhibition. Potential interfering by DMC was studied at five various pNPG concentrations i.e. 0.16-2.65 mM. To obtain velocities of reactions, the absorbance was first plotted against time and the velocities were subsequently plotted against the reciprocal of substrate concentration to construct Lineweaver-Burk plots. The Lineweaver–Burk plots for α-glucosidase inhibition by DMC generated straight lines, which intersected at the same point on X-axis in the second quadrant, indicating noncompetitive inhibition (Figure 1).
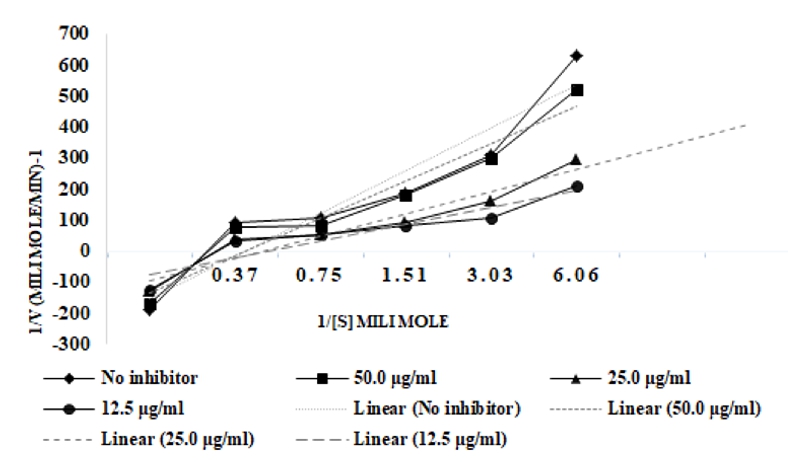
Figure 1. Lineweaver-Burk plot of DMC against α-glucosidase at different concentrations of pNPG
Synergistic activity of DMC with acarbose
Acarbose is a competitive α-glucosidase inhibitor (Ag, 1994), thus it was of interest to establish whether DMC, as noncompetitive inhibitor, might interact synergistically with acarbose in inhibiting α-glucosidase. The experiment was performed at three different concentrations at ¼IC50, ½IC50 and IC50. It was found that acarbose combined with DMC at all the three concentrations (¼IC50, ½IC50 and IC50) resulted in significant (p< 0.05) inhibition compared with the individual compounds at the same concentration indicating synergistic inhibitory activity against α-glucosidase [F(2, 18) = 55.83, p< 0.0001 (the column factor) and F(2, 18) = 54.20, p< 0.0001 (the row factor)] (Figure 2).
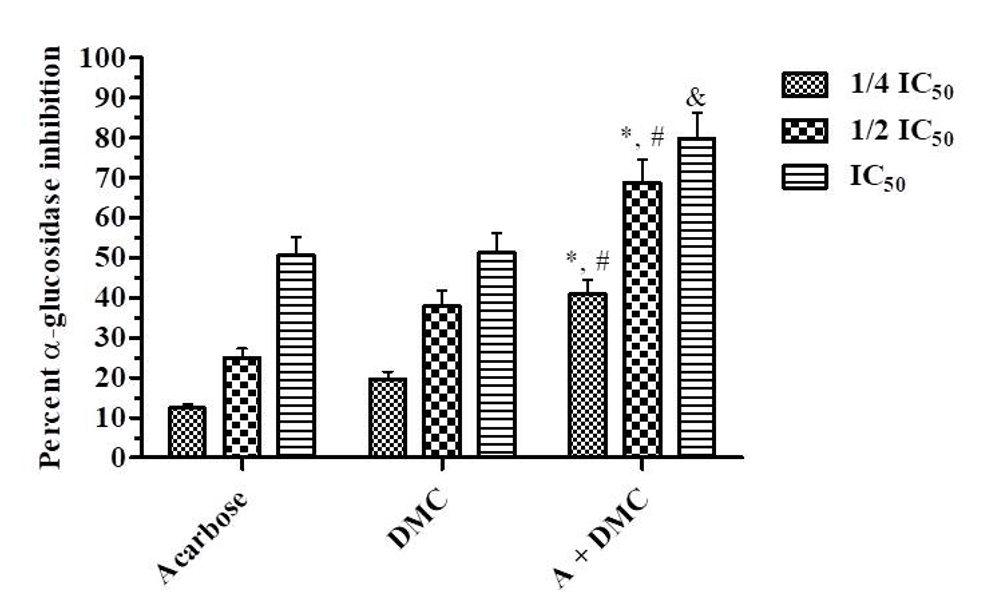
Figure 2. Percentage inhibition of α-glucosidase by acarbose, DMC and combined acarbose with DMC at different concentration on the basis of IC50. Results are expressed as Mean ± S.E.M. (n = 3). Data was analyzed by one way repeat measure ANOVA followed by Tukey’s multiple comparison test. Significance: *p< 0.001 when compared with acarbose group, #p< 0.01 when compared with DMC group, &p< 0.05 when compared with both acarbose and DMC group. A: acarbose; DMC: dimethyl cardamonin.
Discussion
Recently the pressure to develop new drugs for type 2 diabetes has been stimulated by the worldwide increase in the incidence of this disease (Nathan, 2007). α-Glucosidase is the key catalyzing enzyme involved in the process of carbohydrate digestion and glucose release. Inhibition of α-glucosidase is one very effective way of delaying glucose absorption and lowering the postprandial blood glucose level, which can potentially suppress the progression of diabetes mellitus (Thao et al., 2018).
In this study, we investigated the effects of DMC on α-glucosidase and the present study provides the first evidence that DMC could find application as α-glucosidase inhibitor. DMC exhibited satisfactory inhibitory activity against α-glucosidase with IC50 value 87.56 μg/ml, which was much higher than acarbose (IC50 value of 262.39 μg/ml). The Lineweaver–Burk plots for α-glucosidase inhibition by DMC indicated a noncompetitive enzyme inhibition. It was found that acarbose combined with DMC at all the three concentrations (¼IC50, ½IC50 and IC50) resulted in significant inhibition compared with the individual compounds at the same concentration indicating synergistic inhibitory activity against α-glucosidase. These finding suggest that the combination of acarbose with DMC having different inhibitory mechanisms could inhibit α-glucosidase activity more effectively at low doses compared with the single compound, resulting in a reduction of postprandial blood glucose in type-2 diabetes and avoiding adverse effects due to acarbose.
The antidiabetic effect of DMC has been reported in many studies. DMC is known to protect the impaired insulin secretion induced by glucotoxicity in pancreatic β-Cells. DMC also promoted glucose uptake and imposed a paradoxical effect on adipocyte differentiation in 3T3-L1 cells (Hu et al., 2014). Our results therefore provide a novel mechanism for the hyperglycemic effect of DMC. However, additional dose-response studies, in animals are needed to compare dosing efficacy.
Conclusion
The results presented in this study showed that DMC exhibited a noncompetitive inhibitory effect against α-glucosidase and may be considered as an alternative natural antidiabetic agent to control postprandial blood glucose levels. In addition, DMC displayed synergistic inhibition of α-glucosidase in combination with acarbose, suggesting its clinical use to reduce the dose and adverse effects related to acarbose. This study could facilitate effective utilization of DMC as an oral antidiabetic drug or functional food ingredient with a promising role in the formulation of medicines and nutrition supplements.
Conflict of interest
We declare that we do not have any conflict of interest.
References
Ag H. 1994. Pharmacology of α-glucosidase inhibition. European Journal of Clinical Investigation 24(3): 3-10.
Asmat U, Abad K, Ismail K. 2016. Diabetes mellitus and oxidative stress – A concise review. Saudi Pharmaceutical Journal 24(5): 547‑553.
Bell DS. 2004. Type 2 diabetes mellitus: What is the optimal treatment regimen? American Journal of Medicine 8(116): Suppl:23S-29S.
Chaudhury A, Duvoor C, Reddy Dendi VS, Kraleti S, Chada A, Ravilla R, Marco A, Shekhawat NS, Montales MT, Kuriakose K, Sasapu A, Beebe A, Patil N, Musham CK, Lohani GP, Mirza W. 2017. Clinical review of antidiabetic drugs: Implications for type 2 diabetes mellitus management. Frontiers in Endocrinology 26(8): 6.
Fisman EZ, Michael M, Tenenbaum A. 2008. Non-insulin antidiabetic therapy in cardiac patients: Current problems and future prospects. In: Fisman EZ, Tenenbaum A (Eds), Cardiovascular Diabetology: Clinical, Metabolic and Inflammatory Facets Advances Cardiology, pp. 154-170, Basel, S. Karger AG Publishing.
Gao J, Xu P, Wang Y, Wang Y, Hochstetter D. 2013. Combined effects of green tea extracts, green tea polyphenols or epigallocatechin gallate with acarbose on inhibition against α-amylase and α-glucosidase in vitro. Molecules 18(9): 11614-11623.
Guariguata L, Whiting D R, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. 2014. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Research and Clinical Practice 103(2): 137–149.
Gu HJ, Lv JC, Yong KL, Chen X, Liu PP, Zhang XB. 2009. Antidiabetic effect of an active fraction extracted from dragon’s blood (Dracaena cochinchinensis). Journal of Enzyme Inhibition and Medicinal Chemistry 24(1): 136-139.
Hu YC, Hao DM, Zhou LX, Zhang Z, Huang N, Hoptroff M and Lu YH. 2014. 2′,4′-dihydroxy-6′-methoxy-3′,5′-dimethylchalcone protects the impaired insulin secretion induced by glucotoxicity in pancreatic β-Cells. Journal of Agricultural and Food Chemistry 62(7): 1602-1608.
Memon AH, Ismail Z, Aisha AF, Al-Suede FS, Hamil MS, Hashim S, Saeed MA, Laghari M, Abdul Majid AM. 2014. Isolation, characterization, crystal structure elucidation, and anticancer study of dimethyl cardamonin, isolated from Syzygium campanulatum Korth. Evidence-Based Complementary and Alternative Medicine 2014: 470179.
Nathan DM. 2007. Finding new treatment for diabetes--how many, how fast… how good? New England Journal of Medicine 356(5): 437-440.
Rengasamy KR, Aderogba MA, Amoo SO, Stirk WA, Van Staden J. (2013). Potential antiradical and alpha-glucosidase inhibitors from Ecklonia maxima (Osbeck) Papenfuss. Food Chemistry 141(2): 1412–1415.
Şohretoglu D, Sari S, Ozel A, Barut B. 2017. α-Glucosidase inhibitory effect of Potentilla astracanica and some isoflavones: Inhibition kinetics and mechanistic insights through in vitro and in silico studies. International Journal of Biological Macromolecoles 105(1): 1062-1070.
Thao NP, Binh PT, Luyen NT, Hung TM, Dang NH, Dat NT. 2018. α-Amylase and α-glucosidase inhibitory activities of chemical constituents from Wedelia chinensis (Osbeck.) Merr. leaves. Journal of Analytical Methods in Chemistry 2018: 2794904.
Van de Laar FA. 2008. Alpha-glucosidase inhibitors in the early treatment of type 2 diabetes. Vascular Health Risk Management 4(6): 1189–1195.
Yu WG, He H, Yao JY, Zhu YX, Lu YH. 2015. Dimethyl cardamonin exhibits anti-inflammatory effects via Interfering with the PI3K-PDK1-PKCα signaling pathway. Biomolecules and Therapeutics (Seoul) 23(6): 549-556.