

Prasanthi D.*, Deepthi P.
G. Pulla Reddy College of Pharmacy, Mehdipatnam, Hyderabad, Telangana-500028, India
*Corresponding author:
Dr. D. Prasanthi, Associate Professor,
Department of Pharmaceutics,
G. Pulla Reddy College of Pharmacy, Mehdipatnam, Hyderabad, Telangana-500028, India.
Abstract
Objective: The objective is to study the effect of different terpenes on permeation and optimize the better terpene for transdermal itraconazole hydrogel in onychomycosis treatment. Materials and methods: Itraconazole hydrogels were prepared by dispersion method using synthetic polymer (Acrypol 974 and Acrypol 980) as gelling agents. Terpenes nerolidol, limonene, geraniol, carvone at 2.5% and 5% were used. The prepared gels were evaluated for pH, drug content, spreadability, viscosity, in-vitro studies, ex-vivo studies, skin irritation studies and stability studies. Results: The solubility of itraconazole (0.04±0.21mg/ml) was found to be highest with nerolidol (5%) in combination with DMSO (3%) and propylene glycol (1%) in water (5.99±1.5mg/ml). All the formulations have shown good physicochemical properties. In in-vitro diffusion studies through dialysis membrane the trend of release was following the order nerolidol>carvone>geraniol>limonene. Ex-vivo permeation studies of F5 and F13 have shown flux of 28.68±2.3µg/cm2/hr and 31.41±1.67µg/cm2/hr respectively, permeability coefficient of 585cm/hr×10ˉ³ and 641cm/hr×10ˉ³respectively, enhancement ratio of 12.97 and 14.21 respectively, in comparison to pure drug flux of 2.21±0.2 µg/cm2/hr. Skin irritation studies have shown to be non-irritant. FTIR compatibility studies indicate drug and excipients were compatible. The optimized formulation F13 was stable for one month at room temperature. Conclusion: Based on these results, it can be concluded that small alcoholic, liquid terpene with high degree of unsaturation and high lipophilicity (Nerolidol) can provide good permeation of itraconazole.
Keywords: Itraconazole, Acrypol 974, Acrypol 980, Onychomycosis, Terpenes
Introduction
Transdermal drug delivery systems (TDDS) are dosage forms designed to deliver a therapeutically effective amount of drug across a patient’s skin (John, 2014). Transdermal drug delivery provides advantages over conventional drug delivery like avoidance of first pass metabolism, predictable and extended duration of activity, utility of short half-life drugs, improving physiological & pharmacological response, avoiding the fluctuation in drug levels, minimizing undesirable side effects, inter-and intra-patient variations (Preeti et al., 2012). TDDS provides a means to sustain drug release as well as reduce the intensity of action and thus reduce the side effects associated with its oral therapy (Kaur et al., 2015). Topical preparations are applied to the skin for local or systemic effects (Saroha et al., 2013). In some cases, the base may be used alone for its therapeutic properties, such as emollient, soothing or protective action (Upadhyay et al., 2014). However, the limited dermal and transdermal delivery of many small and large molecules is a significant challenge because of the unyielding barrier properties of the skin (Rai et al., 2016; Prateek et al., 2013).
Terpenes classes of chemical enhancers are used in transdermal formulations for facilitating penetration of drugs (Liu et al., 2011). Terpenes which are derived from plant essential oils are naturally occurring hydrocarbons based on combinations of the isoprene units (Fox et al., 2011). They were reported to have high percutaneous enhancement, less toxicity, abilities and low cutaneous irritancy at low concentrations (1-5%) (Dwibhashyam and Ratna, 2010). The effect of a specific terpene on skin depends upon its chemical structure and physicochemical properties, such as its lipophilicity, size and chiralty, boiling point and energy of vaporization and degree of unsaturation (Aqil et al., 2007). Terpenes can increase skin permeation by one or more of the mechanisms: interacting with stratum corneum lipids and/or keratin, and increasing the solubility of drug into stratum corneum lipids (Sapra et al., 2008).
Itraconazole is a potent triazole antifungal agent & highly hydrophobic drug which has very poor water solubility, and used in the treatment of onychomycosis. The most common symptom of onychomycosis is the nail becoming thickened and discoloured: white, black, yellow or green (Sawant and Mohite, 2015). As the infection progresses the nail can become brittle, with pieces breaking off or coming away from the toe or finger completely. If left untreated, the skin can become inflamed and painful underneath and around the nail (Jaiswal et al., 2015). There may also be white or yellow patches on the nailbed or scaly skin next to the nail, and a foul smell. There is usually no pain or other bodily symptoms, unless the disease is severe (Neeraj and Goindi, 2014). Research articles on itraconazole topical films (Mohamed et al., 2013), nanoemulsion (Sampathi et al., 2015), gellified emulsion (Deveda et al., 2010) and liposomes (Sampathi et al., 2014) in transdermal drug delivery are available, but terpenes as chemical penetration enhancers haven’t been worked.
So our objective of present work is to develop topical gel (with terpenes) of the very poorly soluble drug itraconazole which is useful in the treatment of Onychomycosis. Gel formulations were prepared using Acrypols as gelling agent. Itraconazole showed maximum solubility in dimethyl sulfoxide (DMSO), so it was used as chemical enhancer along with different terpenes. Formulations were evaluated for physico-chemical properties and ex-vivo permeation studies. The gel formulations provide delivery of an effective concentration of a triazole compound such as itraconazole to the dermis.
Materials and methods
Materials
Itraconazole was obtained as gift sample from Suven Life Sciences limited (Hyderabad, India). Acrypol 974 &Acrypol 980 were purchased from Corel Pharma Ltd. (Ahmedabad, India). Nerolidol, Geraniol, Limonene, Carvone were purchased from Alfa Aesar Ltd (USA). Dimethyl sulfoxide, triethanolamine & Propylene glycol were purchased from S.D. Fine Chemicals Ltd (India). All other chemicals used were of analytical grade.
Drug excipient compatibility studies
The spectrum analysis of itraconazole and polymer was studied by Fourier Transform Infra-Red (FTIR) Spectroscopy. FTIR spectra were recorded by preparing potassium bromide (KBr) disks using a Shimadzu Corporation (Kyoto, Japan) facility (model - 8400S). Potassium bromide (KBr) disks were prepared by mixing few mg of sample with potassium bromide by compacting in a hydrostatic press under vacuum at 6-8 tons pressure. The resultant disc was mounted in a suitable holder in IR spectrophotometer and the IR spectrum was recorded from 4000 cm-1 to 200 cm-1. The resultant spectrum was compared for any spectral changes. They were observed for the presence of characteristic peaks for the respective functional group in the compound. FTIR study of optimized formulation F13 was carried out to check compatibility of drug and excipients (Neeraj and Goindi, 2014).
Solubility studies
Solubility studies were done using orbital shaker bath. Saturated solutions of itraconazole were prepared by adding an excess amount of drug to 10 ml solvent (distilled water, propylene glycol, DMSO, propylene glycol+propanol+distilled water, propylene glycol+DMSO+distilled water, propylene glycol+DMSO+terpene+distilled water). Saturated solutions were kept in an orbital shaker for 24 hours at room temperature, later it was centrifuged at 3000 rpm for 15 min. Aliquots were filtered through Whatman No. 41 filter paper. The filtrates were diluted appropriately in distilled water and assayed spectrophotometrically at 264 nm (Kaur et al., 2014, Srinivas et al., 2014).
Preparation of gels
Itraconazole hydrogel was prepared using dispersion method (Ramchandani and Sangameswaran, 2013). Accurately weighed quantity of synthetic gums Acrypol 974, Acrypol 980 according to formulation table 1 was soaked in distilled water for 2-3 hrs, propylene glycol was added slowly with stirring. Weighed amount of itraconazole was dissolved in DMSO 3%. The drug solution was added slowly with stirring (400-600 rpm) to the above polymeric solution and triethanolamine was added until gel was formed. To this gel, terpenes were added according to table 1 and mixed properly to obtain a homogenous gel formulation.
Table 1. Formulation of itraconazole hydrogel using terpenes
|
Ingredients %(w/w) |
F1 |
F2 |
F3 |
F4 |
F5 |
F6 |
F7 |
F8 |
F9 |
F10 |
F11 |
F12 |
F13 |
F14 |
F15 |
F16 |
|
Itraconazole |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
|
Acrypol 974 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
- |
- |
- |
- |
- |
- |
- |
- |
|
Acrypol 980 |
- |
- |
- |
- |
- |
- |
- |
- |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
DMSO |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
|
Nerolidol |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
_ |
|
Limonene |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
|
Geraniol |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
|
Carvone |
_ |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
_ |
_ |
_ |
2.5 |
_ |
_ |
_ |
5 |
|
Propylene glycol |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
Triethanolamine |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
|
Distilled water |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
10g |
Physico-chemical evaluation of gels
The prepared gel formulations were evaluated for physico-chemical properties like homogeneity, spreadability, pH, drug content and viscosity.
Homogeneity: It was determined by visual inspection for the appearance and presence of any aggregates (Kumari et al., 2015).
Spreadability: The spreadability of the formulated gels was determined by measuring the spreading diameter of 1g of gel between 20x20 cm glass plates after 1 min. The mass of the upper plate was standardized at 10 g (Mahdi et al., 2018). The spreadability was calculated by using the formula:
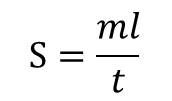
Where,
S = spreadability
m = weight tied to the upper glass slide
l = length of the glass slide
t = time taken in seconds
Determination of pH: The pH of itraconazole gel formulations was determined by using digital pH meter. One gram of gel was dispersed in 100 ml of distilled water and stored for two hours at constant temperature. The measurement of pH of each formulation was done in triplicate and average values were calculated (Sawant et al., 2015).
Drug content: Itraconazole content in gel was measured by dissolving 500 mg of gel (equivalent to 5mg of drug) in 10 ml solvent (pH 7.4PBS+0.5% SLS) by Sonication. The solution was passed through the Whatmann filter paper no.42 and filtered. Absorbance was measured after suitable dilution at 264 nm in UV/VIS spectrophotometer. The experiment was done in triplicate and average values were calculated (Sawant et al., 2015).
Determination of viscosity: Viscosity of prepared gels was determined by VISCOlab 3000 viscometer that contains a piston style electromagnetic sensor and integrated thermometer that provides continuous viscosity and temperature reading. The sample 1-2 ml was applied in the measurement chambers and the results were displayed on the screen of VISCOlab 3000. The determination of viscosity for each formulation was done in triplicate and average values were calculated (Sawant et al., 2015).
In-vitro diffusion studies
Diffusion studies were performed using Franz diffusion cell. The cell was locally fabricated and the volume of receptor compartment was 25 ml. The dialysis membrane used for diffusion studies was placed between donor and receptor compartment. Gel formulation (500 mg of gel) was uniformly applied on membrane and clamped together. The receptor compartment was filled with pH 7.4phosphate buffer saline & 0.5% SLS and maintained by continuous stirring with a magnetic bead. At predetermined time intervals, 1ml samples were withdrawn and replaced with an equal volume of buffer. The samples were analyzed after appropriate dilution using spectrophotometer.
Release rate was calculated by plotting the amount of drug permeated/cm2 versus square root time. The slope is release rate (µg/cm²/hr½) (Rajitha et al., 2014).
Ex-vivo permeation studies
The experimental protocol was approved by the institutional animal ethical committee (IAEC) in G. Pulla Reddy College of Pharmacy with Registration number 320/CPCSEA. Male wistar rats (1.5-2 kg) were used for permeation study. The animal was sacrificed by cervical dislocation and hair was removed from abdomen using an animal hair clipper. Abdominal skin section was excised and observed for existence of cuts and wounds. The fat adhering on dermis was removed using scalpel and finally it was washed under tap water. The skin was stored at −20°C and used within a week (Rajitha et al., 2014).
The thawed rat skin was mounted onto diffusion cell such that the dermis side was in constant contact with receptor solution. 500 mg of gel was applied to the stratum corneum facing the donor compartment and the hydrodynamics in the receptor compartment were maintained by stirring on magnetic stirrer at 600 rpm. 1ml sample was withdrawn at predetermined time intervals for 12 hours and drug content was analyzed by UV-VIS double beam spectrophotometer at 264 nm. Ex-vivo permeation rate studies such as % drug release, cumulative amount permeated in 24 hrs (Q24), steady state transdermal flux (SSTF), permeability coefficient, lag time and enhancement ratio for percutaneous absorption of itraconazole were calculated.
Steady state flux (Jss): The steady state flux is obtained by plotting the cumulative amount of drug permeated in micrograms per square centimeter versus time in hours and the slope is the flux.
Lag time is X intercept of this graph.
Permeability coefficient (Kp) was calculated with the following equation:
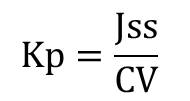
Where,cv is the total donor concentration of the formulation.
Enhancement ratio (ER) is used to evaluate the effect of permeation enhancer on diffusion and permeation of selected drug molecules and is calculated by
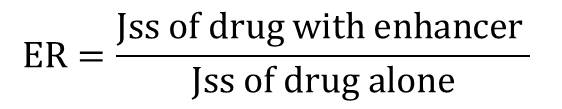
Where, Jss - Steady state flux
Model dependent kinetics
Various models were tested for explaining the kinetics of drug release. To analyze the mechanism of the drug release rate and kinetics of the dosage form, the obtained data was fitted into zero order, first order, Higuchi, and Korsemeyer-peppas release model (Mahdi et al., 2018).
Skin-irritation studies
The animals study was approved by the IAEC. The institutional ethical approval number is 320/CPCSCEA. A primary skin irritation test was performed since skin is the vital organ through which the drug is transported. The test was carried out on three healthy rabbits weighing between 1.5-2 kg. The test was conducted on an unbraided skin of rabbits. Before placing the formulations, the unbraided skin was cleaned with rectified spirit. The placebo gel was kept on the right dorsal surface of first rabbit, whereas formulation F5 was placed on the left dorsal surface of the rabbit. Formulation F13 was placed on the left dorsal surface of second rabbit, whereas the third rabbit was kept as control. The rabbits were under observation for 24hrs and the application sites were graded according to a visual scoring scale (Prasanthi and Lakshmi, 2013).
Stability studies
Stability studies were carried out by keeping optimized formulations in glass containers with polypropylene closure for one month at room temperature. Known amount of gel was taken out at different time intervals like 0, 1st, 2nd, 4th week and was analyzed for appearance, pH, drug content and viscosity (Rajitha et al., 2014).
Statistical analysis
All the evaluation parameters were performed with three samples and data was analysed statistically using Graph Pad Prism 5 software.
Results and discussion
Drug excipient compatibility studies
Fourier transformer infrared spectrophotometer (FTIR) studies were done to verify if there was any interaction between the pure drug and polymers employed. The FTIR graphs of pure drug and optimized formulation F13 are given in figure 1 and figure 2. The peaks 1698, 1229, 2970 & 3464 cm-1 identified in the pure drug indicating C=O stretching, C-N stretching, C-H stretching, N-H stretching were relatively same when compared with FTIR spectrum of optimized formulation F13 given in figure 2. Indicating compatibility of polymers with pure drug.
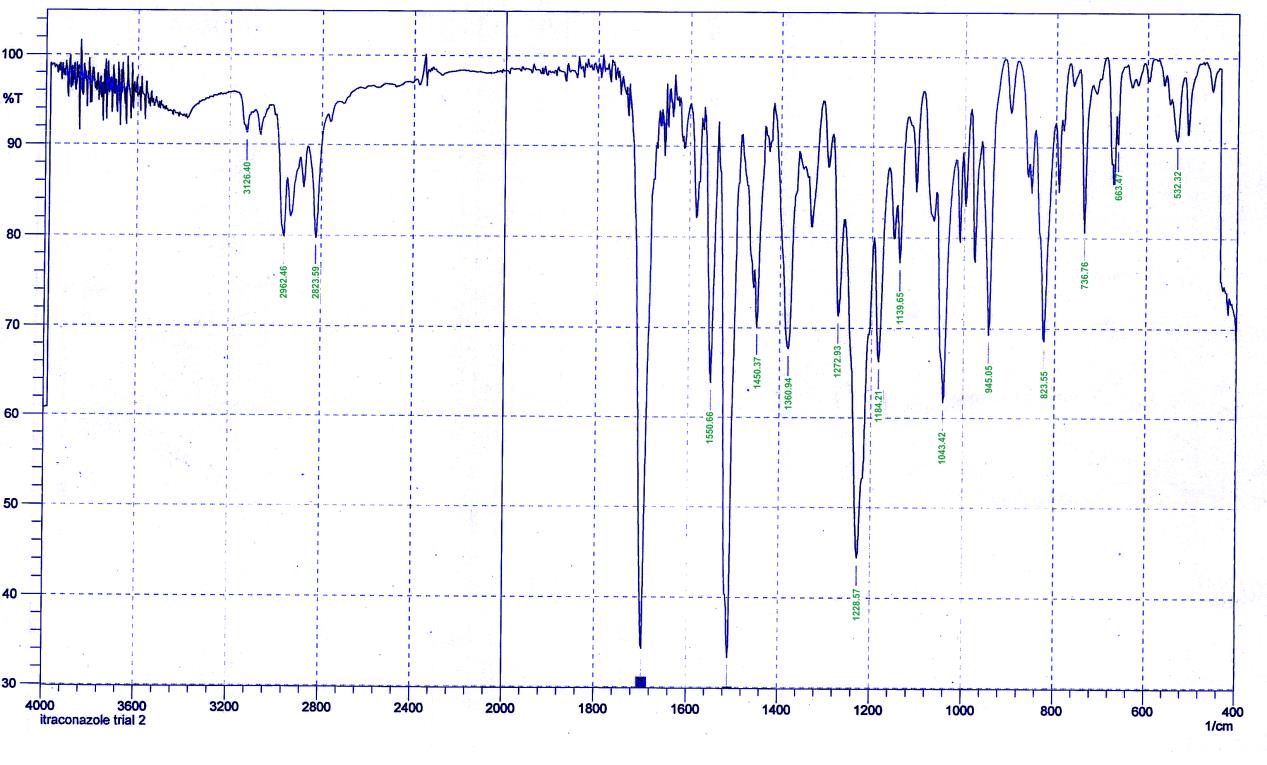 Figure 1.FTIR spectra of pure drug
Figure 1.FTIR spectra of pure drug
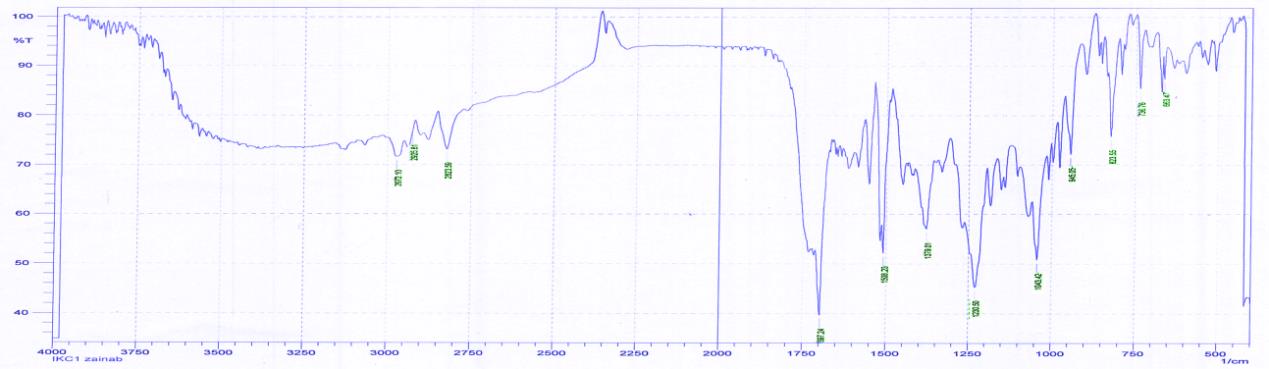
Figure 2. FTIR spectra of F13
Solubility studies
Solubility studies for pure itraconazole were carried in distilled water. The solvent taken for the solubility studies and results obtained are shown in table 2. Solubility of itraconazole is maximum in di-methyl sulfoxide (DMSO) 14.51±0.35mg/ml. From literature review, DMSO concentration upto 5% is non-irritant to skin (Prasanthi and Lakshmi, 2012a). Hence in the gel formulations DMSO 3% was added. With terpenes maximum solubility was observed with nerolidol (5%) in combination with DMSO (3%) and Propylene glycol (1%).
Table 2. Solubility studies for itraconazole
|
Medium (solvent) |
Solubility (mg/ml) |
|
Distilled water |
0.04±0.21 |
|
Propylene glycol |
0.24±0.25 |
|
DMSO |
14.51±0.35 |
|
Propylene glycol (1%) + DMSO (3%) +Distilled water |
2.78±0.42 |
|
Propylene glycol (1%) + Propanol (3%) +Distilled water |
0.09±1.52 |
|
Propylene glycol (1%) + DMSO (3%) +Nerolidol (5%) + Distilled water |
5.99±1.5 |
|
Propylene glycol (1%) + DMSO (3%) +Limonene (5%) + Distilled water |
1.23±1.51 |
|
Propylene glycol (1%) + DMSO (3%) +Geraniol (5%) + Distilled water |
0.71±2.35 |
|
Propylene glycol (1%) + DMSO (3%) +Carvone (5%) + Distilled water |
4.38±2.45 |
Physico-chemical evaluation of gels
Formulated itraconazole hydrogels were evaluated and results are tabulated in table 3. All the formulations were found to have good homogeneity without lumps, spreadability values varies from 10.4±0.23 to12.7±0.06 g.cm/s indicating that the gels are easily spreadable by small amount of shear. pH in the range of 5.61±0.07 to 6.87±0.06 indicating suitability for skin application, drug content in the range of 96.44±0.66% to 99.73±0.71% which shows effective loading and uniform distribution of drug in the formulation, viscosity in the range of 18100 to 19700cps.
Table 3. Physico-chemical evaluation of itraconazolehydrogels
|
Formulation code |
Homogeneity |
Spreadability (g.cm/sec) |
pH |
Drug content (%) |
Viscosity (Cps) |
|
F1 |
+++ |
10.7±0.87 |
6.87±0.06 |
99.23±0.71 |
18300±30 |
|
F2 |
+++ |
11.1±0.04 |
6.77±0.12 |
96.44±0.66 |
18500±60 |
|
F3 |
+++ |
10.9±1.12 |
6.75±0.08 |
97.81±1.33 |
19500±80 |
|
F4 |
+++ |
10.4±0.23 |
6.71±0.14 |
96.55±1.87 |
19500±90 |
|
F5 |
+++ |
12.4±0.13 |
6.25 ±0.21 |
99.73±0.71 |
19500±50 |
|
F6 |
+++ |
12.7±0.06 |
5.81 ±0.22 |
97.25±0.89 |
19100±30 |
|
F7 |
+++ |
12.3±0.02 |
6.17 ±0.26 |
98.75±0.58 |
19100±30 |
|
F8 |
+++ |
12.8±0.53 |
5.61 ±0.07 |
97.25±0.89 |
19600±80 |
|
F9 |
+++ |
10.4±0.23 |
6.71±0.14 |
99.73±0.71 |
19500±70 |
|
F10 |
+++ |
11.1±0.39 |
6.47 ±1.11 |
98.11 ±0.52 |
19500±70 |
|
F11 |
+++ |
12.7±0.06 |
5.91 ±0.09 |
98.16 ±0.71 |
19700±60 |
|
F12 |
+++ |
12.1±0.15 |
6.25 ±0.21 |
99.73±0.71 |
19500±90 |
|
F13 |
+++ |
12.4±0.04 |
6.25 ±0.21 |
97.25±0.89 |
18300±80 |
|
F14 |
+++ |
12.3±0.02 |
5.81 ±0.22 |
98.75±0.58 |
18100±40 |
|
F15 |
+++ |
10.4±0.23 |
5.61 ±0.07 |
97.25±0.89 |
19500±90 |
|
F16 |
+++ |
12.7±0.06 |
6.25 ±0.21 |
96.55±1.87 |
19100±30 |
Note: All the values are expressed as mean± SD, n=3 ; homogeneity- (+-presence of lumps, ++- good, +++- excellent, clear & homogenous)
In-vitro diffusion studies: The gel formulations containing terpenes in 2% and 5% with DMSO 3% were found be physico-chemically compatible with gel properties. These formulations were further evaluated for in-vitro diffusion studies through dialysis membrane. The results are tabulated in table 4 & table 5. From the results it was observed gels with 5% terpenes in Acrypol 974 1% diffused more percentage of drug in 8hrs when compared to 2.5%. Similar pattern of release was also observed with Acrypol 980 1%. The release of drug showed a linear relationship with concentration of terpene. Significant change was observed with increase in concentration of terpene. From the in-vitro diffusion studies, formulation F5, F8, F13, F16 (containing nerolidol and carvone) have shown more than 90% drug release for 8 hrs. This was dependent on the nature of polymer used, as well as type of permeation enhancers used and their concentrations. The trend of release was following the order nerolidol>carvone>geraniol>limonene. This could be due to increase in lipophilicity of terpenes (nerolidol (5.36±0.38), carvone (2.23±0.25), geraniol (3.18±0.30) and limonene (4.58±0.23) log P values). It is observed limonene and geraniol with high log P when compared to carvone is not enhancing the release, as terpenes act as permeation enhancers and show effect on the surface of lipophilic skin by interacting with skin or by rupturing skin integrity which does not show on dialysis membrane (Prasanthi and Lakshmi, 2012b). It was found that the formulations containing nerolidol have more permeation activities. These optimized formulations (F5, F8, F13, F16) were further studied for ex-vivo permeation studies.
Table 4. in-vitro diffusion studies of itraconazole hydrogels F1 to F8
|
Time (hr) |
F1 (%) |
F2 (%) |
F3 (%) |
F4 (%) |
F5 (%) |
F6 (%) |
F7 (%) |
F8 (%) |
|
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
0.5 |
7.5±0.7 |
7.18±0.9 |
7.18±0.2 |
10.31±0.3 |
10.61 ± 0.8 |
6.56±0.2 |
6.56±0.2 |
4.37±0.3 |
|
1 |
8.46±0.3 |
13.77±0.7 |
13.77±0.3 |
16.91±0.3 |
19.41±0.4 |
10.65±0.5 |
7.83±0.3 |
15.95±0.2 |
|
2 |
11.90±0.3 |
17.86±0.7 |
17.86±0.6 |
24.75±0.3 |
28.51±0.5 |
16.66±0.3 |
16.28±0.5 |
25.68±0.24 |
|
3 |
35.36±0.6 |
18.82±0.6 |
18.82±0.1 |
35.41±0.3 |
41.05±0.3 |
29.41±0.6 |
21.62±0.4 |
31.66±0.15 |
|
4 |
51.39±0.4 |
21.01±0.5 |
27.26±0.3 |
51.39±0.5 |
61.41±0.5 |
39.49±0.5 |
32.89±0.5 |
39.18±0.15 |
|
5 |
57.08±0.5 |
21.64±0.2 |
39.17±0.4 |
57.08±0.4 |
70.55±0.3 |
49.53±0.9 |
46.69±0.8 |
52.65±0.26 |
|
6 |
61.16±0.8 |
23.21±0.7 |
44.53±0.3 |
61.16±0.4 |
85.90±0.2 |
60.19±0.5 |
51.74±0.9 |
60.52±0.23 |
|
7 |
63.99±0.6 |
35.71±0.9 |
50.80±0.4 |
63.99±0.4 |
86.59±0.5 |
70.55±0.8 |
58.33±0.7 |
70.55±.04 |
|
8 |
76.81±0.3 |
62.95±0.6 |
59.89±0.2 |
68.38±0.3 |
90.65±0.2 |
78.03±0.5 |
72.73±0.5 |
82.15±0.6 |
|
Release rate(µg/cm2/hr1/2 ) |
984.2 |
1121.4 |
840.1 |
980.2 |
721.1 |
941.2 |
845.3 |
825.2 |
Note: All the values are expressed as mean± SD, n=3
Table 5. In-vitro diffusion studies of itraconazole hydrogels F9 to F16
|
Time (hr) |
F9 (%) |
F10 (%) |
F11 (%) |
F12 (%) |
F13 (%) |
F14 (%) |
F15 (%) |
F16 (%) |
|
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
0.5 |
5.31±0.3 |
3.75±0.7 |
4.37±.2 |
8.75±0.5 |
7.81±0.6 |
4.68±0.1 |
5.93±1.9 |
7.81±0.7 |
|
1 |
9.08±0.3 |
5.95±0.3 |
7.83±.3 |
16.91±.0.3 |
16.28±0.8 |
10.64±0.9 |
8.77±1.5 |
14.09±1.3 |
|
2 |
11.59±0.9 |
8.46±0.9 |
14.40±.5 |
23.19±0.8 |
27.87±0.2 |
19.73±1.3 |
14.09±4.7 |
23.18±0.6 |
|
3 |
22.54±0.7 |
15.34±3.5 |
19.74±.6 |
31.65±0.7 |
41.36±0.3 |
26.95±0.5 |
23.49±3.6 |
39.46±2.2 |
|
4 |
31.65±0.2 |
25.68±2.5 |
26.64±.5 |
49.81±0.5 |
57.97±3.2 |
31.67±0.4 |
39.46±2.7 |
55.15±2.8 |
|
5 |
41.37±0.5 |
36.35±.6 |
33.23±0.8 |
54.57±0.6 |
66.79±3.9 |
45.43±0.1 |
48.90±5.5 |
63.03±2.7 |
|
6 |
55.16±0.1 |
47.64±.2 |
47.94±0.5 |
57.71±0.6 |
82.14±3.4 |
61.43±0.2 |
58.32±2.6 |
79.01±2.5 |
|
7 |
81.15±1.2 |
69.25±0.2 |
54.87±0.3 |
76.79±0.3 |
95.01±0.3 |
72.74±0.6 |
70.85±1.9 |
92.81±1.3 |
|
8 |
86.26±0.5 |
79.2±0.5 |
61.46±0.7 |
80.93±0.8 |
99.44±0.1 |
84.04±0.5 |
80.28±3.9 |
94.43±3.4 |
|
Release rate(µg/cm2/hr1/2 ) |
994.2 |
771.2 |
721.4 |
1022.4 |
1161.1 |
1121.4 |
1021.2 |
1051.3 |
Note: All the values are expressed as mean± SD, n=3
Ex-vivo permeation studies
These studies were performed for optimized formulations using rat abdominal skin to know the amount of drug that has actually diffused across the tissue over the period of time, thus permeation parameters and kinetics of drug transport can be assessed. Ex-vivo permeation studies through rat abdominal skin (figure 3) indicate the enhancement of itraconazole permeation when compared with pure drug (8.2±1.8μg/cm2/hr), this may be due to the poor water solubility of itraconazole in water. From literature it is known terpenes can increase skin permeation by one or more of the mechanisms: interacting with stratum corneum lipids and/or keratin, and increasing the solubility of drug into stratum corneum lipids (Prasanthi and Lakshmi, 2012b). F13 has shown highest permeated amount and more than 90% of itraconazole was permeated from the transdermal gel formulations, this could be due to the improved itraconazole solubility by nerolidol and its effect on the lipid layer of stratum corneum.
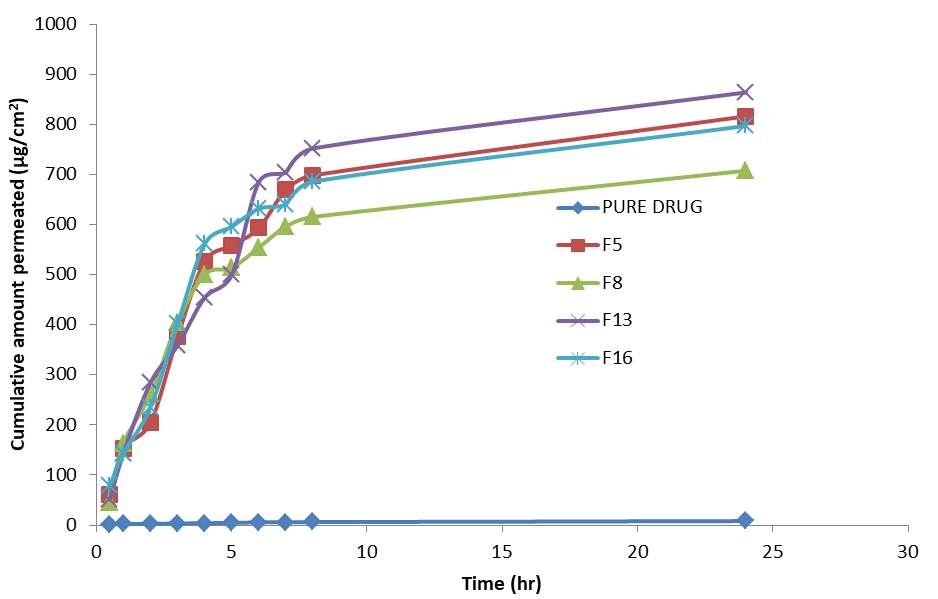 Figure 3. Ex-vivo permeation profiles for optimized itraconazole hydrogels
Figure 3. Ex-vivo permeation profiles for optimized itraconazole hydrogels
Permeability parameters of optimized gel formulations
The ex-vivo permeability parameters (flux, permeability coefficient, lag time and enhancement ratio) were calculated for all optimized formulation and the results were tabulated in table 6. It is observed from the permeability parameters that the nerolidol has more permeation enhancement than carvone. This could be due to increase in hydrophilicity of terpenes showed increase in flux, permeability coefficient and enhancement ratio. Log P values of optimized terpenes are nerolidol (5.36±0.38) and carvone (2.23±0.25), the permeated amount and flux also decreased showing a linear relationship between permeation and log P proving the influence of lipophilicity on permeation. It was found that, Nerolidol with highest lipophilicity showed maximum permeation (Prasanthi and Lakshmi, 2012b).
Table 6. Permeability parameters of optimized gel formulations
|
Formulation code |
Flux (µg/cm2/hr) |
Lag time (hr) |
Permeability coefficient (cm/hr) |
Enhancement ratio |
|
Pure drug |
2.21±0.2 |
10.5±1.2 |
442 |
1 |
|
F5 |
28.68±2.3 |
0.89±0.3 |
585.31±2.3 |
12.97 |
|
F8 |
22.97±0.5 |
1.09±1.5 |
468.78±0.5 |
10.39 |
|
F13 |
31.41±1.6 |
0.62±0.4 |
641.02±2.3 |
14.21 |
|
F16 |
26.72±1.2 |
0.71±0.8 |
545.31±1.2 |
12.09 |
Table 7. Stability study of F13 at room temperature for one month
|
Parameters |
Time in weeks for F 13 |
|||
|
O (Initial) |
1st week |
2nd week |
4thweek |
|
|
Appearance |
+++ |
+++ |
+++ |
+++ |
|
Drug content (%) |
97.25±0.89 |
97.18±0.14 |
97.08±0.23 |
97.08±0.22 |
|
Spreadability |
12.4±0.04 |
12.4±0.4 |
12.35±0.2 |
12.31±0.7 |
|
pH |
6.25±0.21 |
6.18±0.23 |
6.06±0.24 |
6.04±0.23 |
|
Viscosity |
18300±80 |
18300±80 |
18300±80 |
18300±80 |
Model dependent kinetics of optimized formulations: Model dependent kinetics was done for F13 optimized gel formulations in order to determine the release kinetics and release mechanism. It was found to follow zero order drug release kinetics and anamolous transport release mechanism.
Skin irritation studies
Skin irritation studies were conducted on depilated rabbit skin. Skin reaction at the site of application was assessed and scored according to Draize method. The formulations control, placebogel, F5 and F13 showed irritation potential of “0”, thus proving to be non-irritant. The “0” value in an irritancy test indicates that the applied formulations are generally non-irritant to the human skin. No obvious erythema and edema was observed after 24 hours of the application of the formulations.
Stability studies
The stability studies were conducted for one month at room temperature and the formulation F13 was found to be stable, without significant change in the appearance, drug content, viscosity and pH.
Conclusion
This study attempts to demonstrate the influence of various permeation enhancers at 2.5% and 5% concentration (nerolidol, limonene, geraniol, carvone) on the percutaneous permeation of itraconazole transdermal gel. Ex-vivo permeation studies revealed that the F5, F13 have shown better release of itraconazole in 8 hrs with flux of 28.68±2.3µg/cm²/hr and 31.41±1.67µg/cm2/hr respectively; permeability coefficient of 585 cm/hr×10ˉ³ and 641 cm/hr×10ˉ³ respectively and enhancement ratio of 12.97 and 14.21 respectively in comparison to pure drug flux of 2.21±0.2 µg/cm2/hr. Skin irritation studies proved that the formulations were non-irritant. The formulation F13 (containing nerolidol 5% &Acrypol 980 1%) which showed maximum permeability and enhancement was optimized. Formulation F13 was found to be stable for one month at room temperature. It can be concluded that terpenes with high lipophilicity are good penetration enhancers as linear relationship was observed with lipophilicity and permeation. Thus nerolidol (5%) as permeation enhancer can provide good permeation of itraconazole.
References
Aqil M, Ahad A, Sultana Y, Ali A. 2007. Status of terpenes as skin penetration enhancers. Drug Discovery Today, 12:1061-7.
Deveda P, Jain A, Vyas N, Khambete H, Jain S. 2010. Gellified emulsion for sustain delivery of itraconazole for topical fungal diseases. International Journal of Pharmacy and Pharmaceutical Sciences, 2(1):104-112.
Dwibhashyam VS, Ratna VJ. 2010. Chemical penetration enhancers-an update. Indian Drugs, 47:5-18.
Fox LT, Gerber M, Plessis JD, Hamman JH. 2011. Transdermal drug delivery enhancement by compounds of natural origin. Molecules, 16:10507-40.
Jaiswal M, Kumar M, Pathak K. 2015. Zero order delivery of itraconazole via polymeric micelles incorporated in situ ocular gel for the management of fungal keratitis. Colloids and Surfaces B: Biointerfaces, 130(2):23–30.
John L. 2014. Review on transdermal drug delivery system. International Journal of Pharma Research and Health Sciences, 2(4):261-272.
Kaur D, Raina A, Singh N. 2014. Formulation and evaluation of Acrypol 940 based glibenclamide transdermal gel. International Journal of Pharmacy and Pharmaceutical Sciences, 6(8):434-440.
Kaur D, Rajinder. 2015. Novel approach: transdermal gel. International Journal of Pharma Research & Review, 4(10):41-50.
Kumari, Singh A, Saurabh SS, Rathore KS, Issara R. 2015. Formulation and evaluation of lycopene emulgel. Indo American Journal of Pharmaceutical Sciences, 2(6):1013-1027.
Liu CH, Chang FY, Hung DK. 2011. Terpene microemulsions for transdermal curcumin delivery: Effects of terpenes and co-surfactants. Colloids and Surfaces B: Biointerfaces, 8(2):63-70.
Mahdi AZ, Prasanthi D, Amoolya C, Zainab RH, Lakshmi PK. 2018. Effect of Transcutol and stearylamine on ibuprofen hydrophilic gel for transdermal delivery. International Journal of Drug Delivery Technology, 8(1):6-14.
Mohamed SM, Eid AMM, Elgadir MA, Mahdy AM. 2013. Preparation and release characteristics of itraconazole polymeric films for topical application. International Journal of Pharmacy and Pharmaceutical Sciences, 5(3):167-170.
Neeraj K, Goindi S. 2014. Statistically designed nonionic surfactant vesicles for dermal delivery of itraconazole: characterization and in-vivo evaluation using a standardized tinea pedis infection model. International Journal of Pharmaceutics, 472(1-2):224–240.
Prasanthi D, Lakshmi PK. 2012a. Effect of chemical enhancers in transdermal permeation of alfuzosin hydrochloride. International Scholarly Research Network ISRN Pharmaceutics, 1(1):1-8.
Prasanthi D, Lakshmi PK. 2012b. Terpenes: effect of lipophilicity in enhancing transdermal delivery of alfuzosin hydrochloride. Journal of Advanced Pharmaceutical Technology & Research, 3(4):216-223.
Prasanthi D, Lakshmi PK. 2013. Optimisation of transdermal gel formulations of tolterodine tartrate by experimental design. Turkish Journal of Pharmaceutical Sciences, 10(2):273-286.
Prateek C, Tomar RS, Ramchandani U, Manocha N, Agrawal H. 2013. Topical gel. International Journal of Pharmaceutical & Biological Archives, 4(4):606-613.
Preeti GK, Rohit RS, Chougule DD, Bhise SB. 2012. Formulation and evaluation of celecoxib gel. Journal of Drug Delivery & Therapeutics, 2(3):132-135.
Rai AK, Tiwari G, Tiwari R, Yadav P. 2016. Penetration enhancers: An approach to increase skin permeation and its effects with eutectic mixture. World Journal of Pharmacy and Pharmaceutical Sciences, 5(6):856-867.
Rajitha K, Lakshmi PK, Pranitha A, Prasanthi D. 2014. Transdermal permeation enhancement of ibuprofen and its solid dispersion. International Journal of Research in Ayurveda and Pharmacy, 5(4):508-514.
Ramchandani U, Sangameswaran B. 2013. Formulation and evaluation of topical gel of ketoprofen using different polymers. International Journal of Pharmaceutical and Biological Archives, 4(2):323-326.
Sampathi S, Ajimera T, Kuchana V. 2014. Preparation and evaluation of liposome entrapped hydrogel complex systems of itraconazole for enhanced transdermal permeation. Journal of Pharmaceutical and Scientific Innovation, 3(1):25-29.
Sampathi S, Mankala SK, Wankar J, Dodoala S. 2015. Nanoemulsion based hydrogels of itraconazole for transdermal drug delivery. Journal of Scientific and Industrial Research, 74(2):88-92.
Sapra B, Jain S, Tiwary AK. 2008. Percutaneous permeation enhancement by terpenes: Mechanistic view. AAPS Journal, 10:120-32.
Saroha K, Singh S, Aggarwal A, Nanda S. 2013. Transdermal gels-An alternative vehicle for drug delivery. International Journal of Pharmaceutical, Chemical and Biological Sciences, 3(3):495-503.
Sawant A, Mohite SK. 2015. Formulation and evaluation of itraconazole emulgel for topical drug delivery. Asian Journal of Pharmaceutical Science and Technology, 5(2):91-96.
Srinivas L, Hemalatha B, Kumar VST, Rao NM, Teja B. 2014. Studies on solubility and dissolution enhancement of itraconazole by complexation with Sulfo-Butyl7 Ether β Cyclodextrin. Asian Journal of Biomedical and Pharmaceutical Sciences, 4(38):6-16.
Upadhyay G, Verma S, Parvez N, Sharma PK. 2014. Recent trends in transdermal drug delivery system - A Review. Advances in Biological Research Imaging & Biointerphases, 8(1):1-8.