

Mital Bhadaniaa,*, Falguni Majmudarb, M. A. Karmurc
a,*Dept. of Product Management, McLeon Pharmaceuticals, Gandhinagar, India
bDept. of Pharmacology, Smt. NHL Municipal Medical College, Ahmedabad, India
cSaurastra Diabetes Centre, Rajkot, India
*Author for correspondence
Dr. Mital Bhadania
McLeon Pharmaceuticals
B08 Ariane Casa, Opp. IOC Petrol Pump
Adalaj-382421 Gandhinagar Gujarat, India
Abstract
Objective: This study was intended to improve diabetes care among patients with type 2 diabetes mellitus with premixed insulin analogue and human premixed insulin therapy. To evaluate safety and efficacy of premixed insulin add-on to metformin and sulphonylurea in routine clinical practice. Material and methods: This was a 26 week, multi-centre, parallel group, and open label study in type 2 diabetes mellitus patients. Patients were treated at various diabetes clinic and referral clinics in Gujarat India. The data included demographic characteristics, medical history, and the treatment regimens were collected during their routine clinical visit. Results: Out of 226 patients; 177 patients were completed the study, of which 103 (58.2%) were male and 74 (41.8%) were female. The mean±SD; A1C% was 10.04 ± 1.68; age was 60.61 ± 6.96 years; duration of diabetes mellitus was 5.12 ± 3.16 years at baseline. Combination of premixed insulin analogue was significantly achieved % reduction at target of <7% A1C as compared to human premixed insulin. Premixed insulin analogue significantly reduced A1C, FBG, PPBG as compared to the dual and triple combination of human premixed insulin. Premixed insulin analogue group showed very less hypoglycemic events during the study period compared to other insulin group. Conclusion: Premixed insulin analogue lowers A1C to a greater degree and minimize the risk of hypoglycemic events in T2DM patients compared to human insulin even though poor glycemic control. Patient and physician continuous education on drug therapy will leads to achieve adequate glycemic control.
Keywords: Diabetes mellitus, Premixed insulin analogue, Human premixed insulin, Glycosylated haemoglobin
Introduction
Quality of diabetes care in India is a challenge. Diabetes management is depending upon the expertise, awareness, attitude, and perceptions among health care professionals in diabetes (Joshi, 2015). Currently, India is a diabetic home of more than 69 million individuals and it is expected to be 123 million by the year 2040. In India, doctor population ratio is 1:1800, as the availability of specialists in the area, is mostly from the urban area and since about 70% initial diagnosis is being done by non-specialists, the key challenge of diabetes care (Bhalla et al., 2016).
Type 2 diabetes mellitus (T2DM) treatment includes oral anti-diabetic (OAD) drugs and insulin treatment with lifestyle adjustment. The overall goal is to achieve acceptable glycemic control and adequately reduce associated complications in long-term (Chawla et al., 2016). A number of studies, most prominently the UK prospective diabetes studies (UKPDS) suggest that proper glycemic control reduces the development and progression of T2DM vascular complications (Stratton et al., 2000; UKPDS, 1998). Due to the progressive deterioration in T2DM, the majority of subjects require multiple therapies to attain the recommended glycemic targets set by, for example, the American diabetes association (ADA). It is recommended that initiation of insulin therapy should begin when glycosylated haemoglobin (A1C) has deteriorated to > 7.0% after maximum attention to dietary control and oral antidiabetic therapy (American diabetes association, 2011).
Considering the fact, Gujarat is one of the richest and developed states of India. Many of Gujarati people have more oil and sugar content in diet and less physically active lifestyle. As a result, Gujarat is to the forefront of contributors of diabetic patients in India and high prevalence of obesity and hypertension (Pandya et al., 2012). Studies show that majority of patients are not achieving the recommended glycemic target despite a broad range of anti-diabetic therapies (Hoerger et al., 2007; Juming et al., 2012; Linong et al., 2012).
In T2DM, insulin is usually initiated in subjects who have failed to achieve the glycemic targets, % A1C should be <6.5 and ≤7.0 recommended by ADA and American college of Endocrinology (ACE) guideline respectively (American diabetes association, 2003; Field, 2003).
Human premixed insulin (HPI), also known as biphasic human insulin 30, contains a fixed mixture of 70% neutral protamine hagedorn (NPH) human insulin isophane suspension and 30% soluble regular human insulin. When injected 30 min before a meal, soluble component aim to lower post-prandial blood glucose (PPBG), while NPH provides basal insulin coverage (Shah et al., 2009).
Premixed insulin analogue (PIA) is a novel biphasic insulin analogue which targets PPBG, provides up to 24 hours of basal insulin coverage given within 15 minutes of a meal. PIA comprises 30% soluble insulin aspart and 70% protaminized insulin aspart. The soluble fraction is absorbed rapidly, and the protamine crystallised fraction has a delayed absorption, similar to that of human insulin NPH (Novomix, 2001; Rolla, 2002).
Insulin is recommended as an add-on to OAD drug preferably metformin, according to the current guidelines of the ADA and European association for the study of diabetes (Nathan et al., 2006). The study design was to evaluate and raise awareness among physician and patients about insulin therapy.
Research design and methodology
The aim of this observational study was to evaluate the safety and effectiveness of human premixed insulin and premixed insulin analogue on T2DM patients from Gujarat under normal clinical practice. The study design was a 26 week, multicentre, parallel group, and open label. The study protocol was approved by the Medilink ethics committee protocol number A-11-11 and was in accordance with the Declaration of Helsinki and ICH Good Clinical Practice (Helsinki, 2008, ICH, 1996). The study site selection was done depend on the investigator qualification and facility require for diabetes practice. The study investigators were educated on appropriate use of human premixed insulin and premixed insulin analogue therapy to improve glycemic control by personal visit of co-investigator. The study site staffs were also educated on ICH Good Clinical Practice for proper site management as per protocol. Written informed consent was obtained from all patients. Data were collected at baseline (visit 1), approximately at 13th week (visit 2) and at approximately 26th week (visit 3). The frequency and timing of visits were based on the accepted standard of care for T2DM management (Shah et al., 2009).
Study population
The study was conducted at 7 sites in Gujarat, India. All the sites were screened and subjected to treatment as per protocol. All patients were recruited by their physicians during their routine clinical practice. This study design was included subjects, who did not achieve adequate glycemic control (desire A1C level), treatment with metformin and combination with other hypoglycemic agents, was therefore necessary.
Patients were selected and screened based on the inclusion criteria: All the type 2 diabetes patients, >18 years of age, men or women were eligible for the study. A1C > 9.0 % at time of diagnosis. A1C > 7.0 % even after 6 months of therapy with other anti-diabetic agents (OADs and/or insulin). Exclusion criteria: type 1 diabetes, hypersensitivity to premixed insulin or to any of the excipient (Valensi, 2009; Sharma et al., 2012; Venkataraman et al., 2009).
Procedure
Prior to any study related activity, the investigator gave the oral information to subject or parents/the subject’s legally acceptable representative, as applicable. The subjects were educated in subjects’ mother language on general diabetes education, diet, foot care, exercise and self monitoring blood glucose procedure. The subjects were also informed of study related risks and benefits and they could withdraw from the study at any time. The responsibility for seeking informed consent (IC) remained with the investigator. The written IC was signed and personally dated by the investigator. A voluntary signed and dated informed consent form (ICF) was obtained at visit-1 prior to any study related activities. The study variable includes demographic variables, e.g. age and diabetes duration. The primary clinical measurements were glycosylated haemoglobin (A1C), fasting blood glucose (FBG) and post-prandial blood glucose (PPBG). Study includes the measurement of adverse events or serious adverse events as safety variables. It includes the number of minor and major hypoglycemic episodes; tested at baseline, 13th week and 26th week visit. Other safety variables include lipid profile were measured at baseline and 26th week.
Statistical analysis
Continuous variables were summarized with descriptive statistics expressed as mean ± standard deviation (SD). The data were analyzed using ANOVA followed by Tukey’s multiple comparison tests and paired t-test by Graph pad prism 5.01 software. p<0.05 were considered as statistically significant. Categorical data were summarized with the number (N) and percentage (%) of subjects in each category. The continuous endpoints were defined as change from baseline to week 26.
Results
Results shows (table 1) 226 patients at baseline, 24 (10.61%) were on metformin alone and 53(23.45%) patients were on oral dual combination of metformin plus sulphonylurea, 62 (27.43%) patients on oral dual combination with insulin, while 87 (38.49%) patients were on oral triple combination with insulin. Table shows the summary of subject study completion status full analysis set where 41 (18.1%) patients were dropped out due to loss of follow up and 8 (3.5%) patients due to discontinued drug therapy during the study period. No major adverse drug reaction was found in any of study group. The demographic data for these patients are presented in table 1. The mean patient age was 60.61 ± 6.96 years and mean age of male and female were 60.07% and 61.37%, respectively. The mean T2DM duration was 5.12 ± 3.16 years, among that male and female diabetes duration, were 4.96 ± 3.21 and 5.36 ± 3.11 years respectively.
Glycemic control
The observation shows (table 1) shows the baseline mean A1C was 10.04 ± 1.68%. Figure 1 represents the summary of A1C% from baseline to week 26th effective analysis set at A1C% level 7 and 6.5% achievement. All group was significantly reduced A1C % (p<0.001) at 26th week visit compared to baseline visit. % reduction of A1C% in group Metformin, Met+SU, Met+HPI, Met+SU+HPI, Met+PIA, and Met+SU+PIA were 10.07 ± 6.8%, 13.54 ± 9.55%, 30.22 ± 5.9%, 36.37 ± 6.19%, 36.30 ± 7.9% and 40.19 ± 7.7% respectively. Combination of metformin and premixed insulin analogue group (p<0.05) and add on to sulphonylureas (p<0.001) were significantly increased % reduction of A1C as compared to the combination of metformin and human premixed insulin (Figure 2).
Table 1. Treatment administered. SU, Sulphonylureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue
|
Anti-diabetic Medication |
Baseline |
26 Week |
Male |
Female |
|
|
N (226) |
% |
N (177) |
N (103) |
N (74) |
|
|
Biguanide (Metformin) |
24 |
10.61 |
23 |
16 |
7 |
|
Dual Combination of Metformin |
53 |
23.45 |
- |
- |
- |
|
Metfromin+Sulphonylurea (Glimepiride, Glibenclamide, Gliclazide, Glipizide) |
53 |
23.45 |
30 |
18 |
12 |
|
Oral Dual Combination with Insulin |
62 |
27.43 |
- |
- |
- |
|
Metformin + HPI 30/70 |
31 |
13.71 |
24 |
14 |
10 |
|
Metformin + PIA 30/70 |
31 |
13.71 |
31 |
18 |
13 |
|
Oral Triple Combination with Insulin |
87 |
38.49 |
- |
- |
- |
|
Metformin + SU+ HPI 30/70 |
50 |
22.12 |
35 |
16 |
19 |
|
Metformin + SU + PIA 30/70 |
37 |
16.37 |
34 |
21 |
13 |
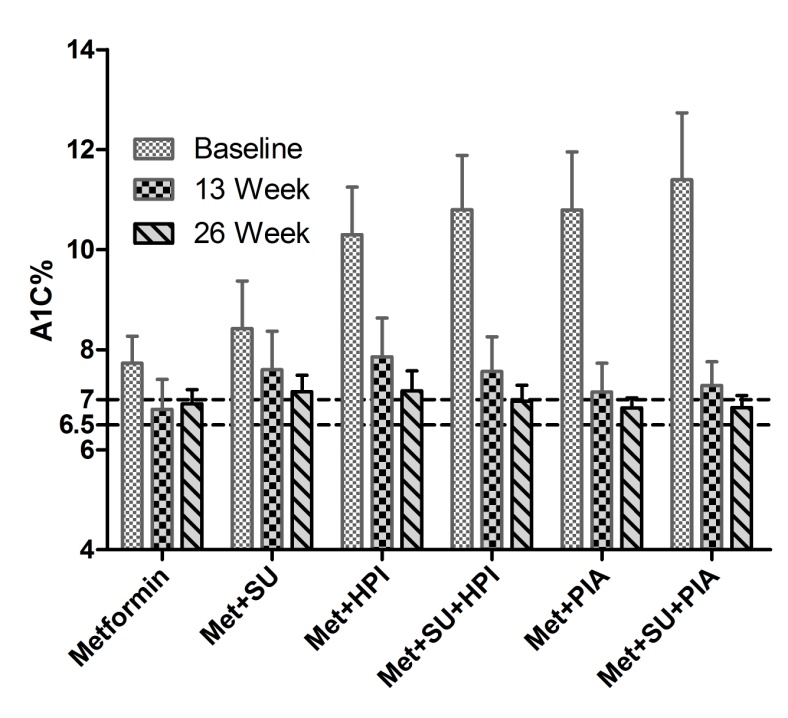 Figure 1. Effect of group combination with metformin on A1C% from baseline to week 26 effective analysis set. Values are Mean ± SD. A1C, glycosylated haemoglobin; Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 1. Effect of group combination with metformin on A1C% from baseline to week 26 effective analysis set. Values are Mean ± SD. A1C, glycosylated haemoglobin; Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 2. Effect of group combination with metformin on % reduction of A1C from baseline to week 26 effective analysis set. Values are Mean ± SD. One way ANOVA followed by Tukey’s multiple comparison tests. ***indicates p<0.001 as compared to Met+SU group, +++ indicates p<0.001 as compared to Met+HPI group, +indicates p<0.05 as compared to Met+HPI group. A1C, glycosylated haemoglobin; Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 3 represents % A1C % achievement at 6.5 and 7.0 based on ADA and ACE guideline. Metformin combination with premixed insulin analogue was significantly achieved % reduction at 7% A1C compared to human premixed insulin and metformin group, while add on to sulphonylurea achieved 6.5 % A1C.
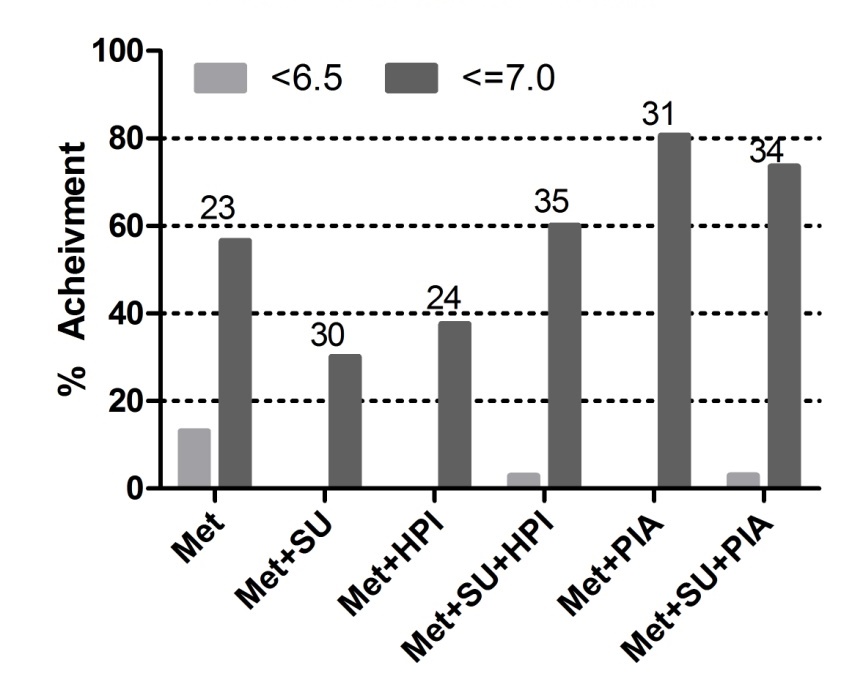 Figure 3. Effect of group on % achievement 6.5 and 7.0 based on ADA and ACE guideline on % A1C. Above bar graph number indicating number of patients in particular group. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 3. Effect of group on % achievement 6.5 and 7.0 based on ADA and ACE guideline on % A1C. Above bar graph number indicating number of patients in particular group. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
All the study groups were significantly reduced FBG (p<0.001) at 26 week visit compared to baseline. % reduction of FBG of study group Metformin, Met+SU, Met+HPI, Met+SU+HPI, Met+PIA, and Met+SU+PIA were 16.30 ± 14.0, 22.18 ± 16.43, 45.72 ± 12.96, 48.89 ± 16.25, 48.09 ± 16.58 and 54.62 ± 11.79 respectively. Insulin combination groups were significantly (p<0.001) reduced FBG level as compared to the metformin and sulphonylurea combination (Figure 4 and 5).
The observation data in figure 6 and 7 shows the effect of group combination with metformin on PPBG from baseline to week 26. Drug combinations were significantly (p<0.001) reduced PPBG on 26 week as compared to baseline. % reduction of PPBG of study group Met, Met+SU, Met+HPI, Met+SU+HPI, Met+PIA, and Met+SU+PIA were 9.4 ± 7.8, 24.79 ± 12.8, 42.11 ± 12.51, 51.32 ± 12.33, 47.95 ± 14.0 and 53.48 ± 8.18 respectively. The triple combination of metformin, sulphonylureas, and premixed insulin analogue group was significantly (p<0.01) increase % reduction of PPBG as compared to the combination of metformin and human premixed insulin.
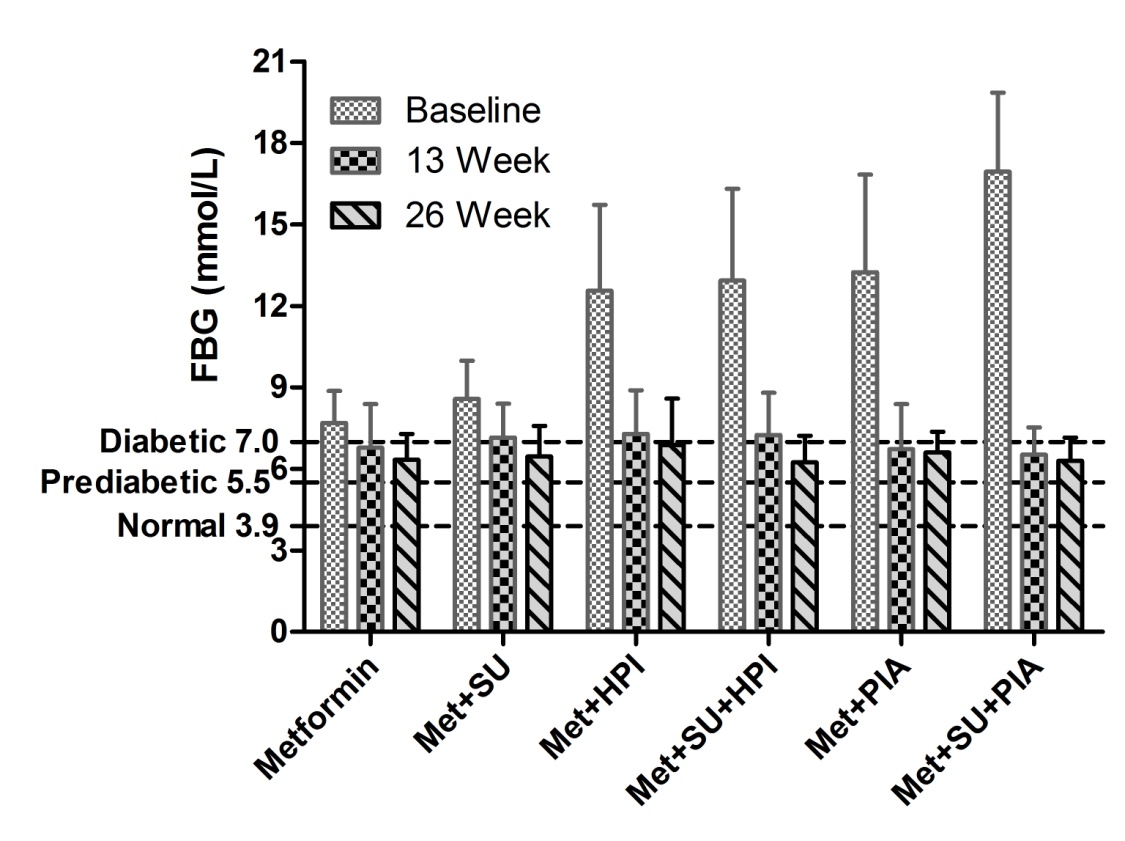
Figure 4. Effect of group combination with metformin on fasting blood glucose (FBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 5. Effect of group combination with metformin on % reduction of fasting blood glucose (FBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. One way ANOVA followed by Tukey’s multiple comparison tests. *** p<0.001 as compared to metformin and sulphonylurea combination group. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
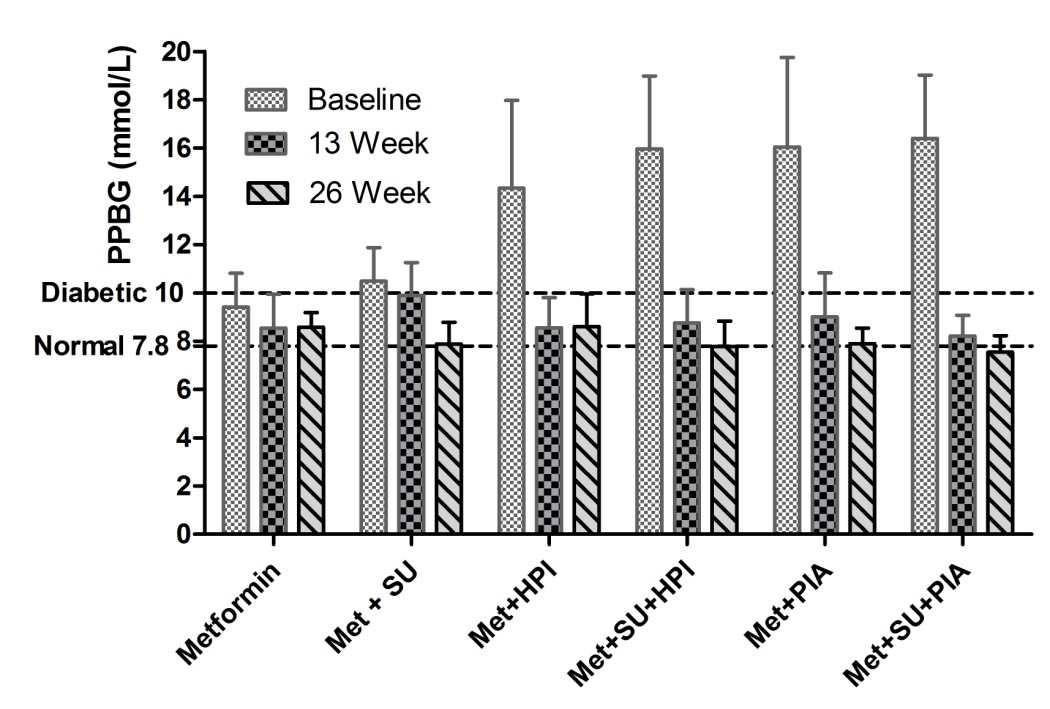 Figure 6. Effect of group combination with metformin on postprandial blood glucose (PPBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 6. Effect of group combination with metformin on postprandial blood glucose (PPBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
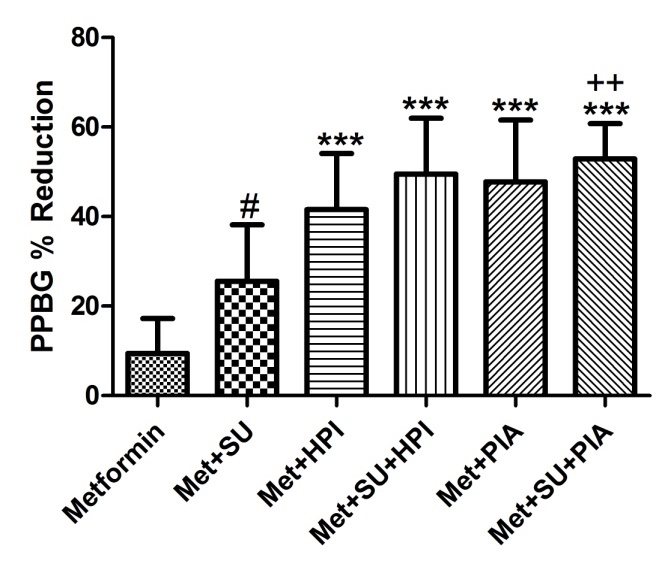 Figure 7. Effect of group combination with metformin on % reduction of postprandial blood glucose (PPBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. One way ANOVA followed by Tukey’s multiple comparison tests. # p<0.001 as compared to metformin, *** p<0.001 as compared to met+SU, ++p<0.01 as compared to metfromin and human premixed insulin combination. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 7. Effect of group combination with metformin on % reduction of postprandial blood glucose (PPBG) from baseline to week 26 effective analysis set. Values are Mean ± SD. One way ANOVA followed by Tukey’s multiple comparison tests. # p<0.001 as compared to metformin, *** p<0.001 as compared to met+SU, ++p<0.01 as compared to metfromin and human premixed insulin combination. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Hypoglycemia
Hypoglycemia occurred in 8.47% of total patients according to the questionnaire in case record form, although there was not observed major hypoglycemic events during the study period at any of study group. Around 20% of patients showed minor hypoglycemic event by human premixed insulin group. Combination of metformin, sulphonylurea, and premixed insulin analogue did not show any hypoglycemic event among study subjects (Figure 8).
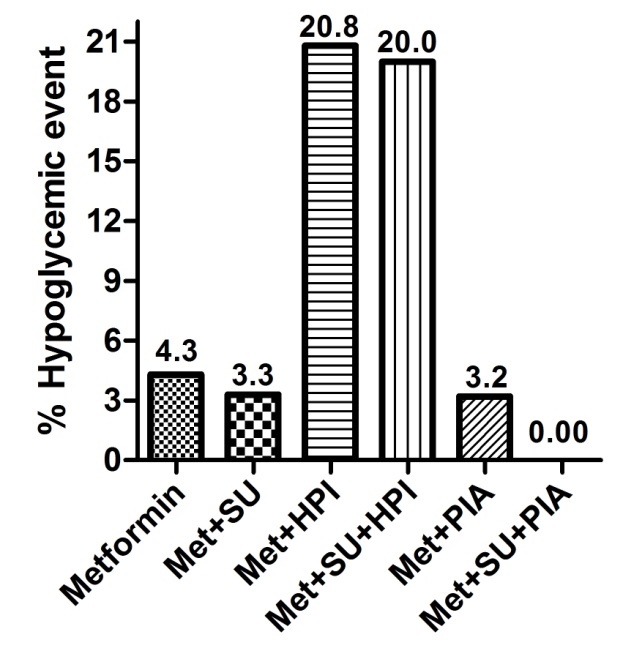 Figure 8. Summary of percentage of hypoglycemic events occurs during treatment period. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Figure 8. Summary of percentage of hypoglycemic events occurs during treatment period. Met, Metfromin; SU, Sulphonulureas; HPI, Human premixed insulin; PIA, Premixed insulin analogue.
Other safety variable
As other safety variable parameter considered total cholesterol (TC), HDL-cholesterol, LDL-cholesterol and triglyceride (TG) from baseline to week 26. Study result did not show any significant change during the study (Figure 9).
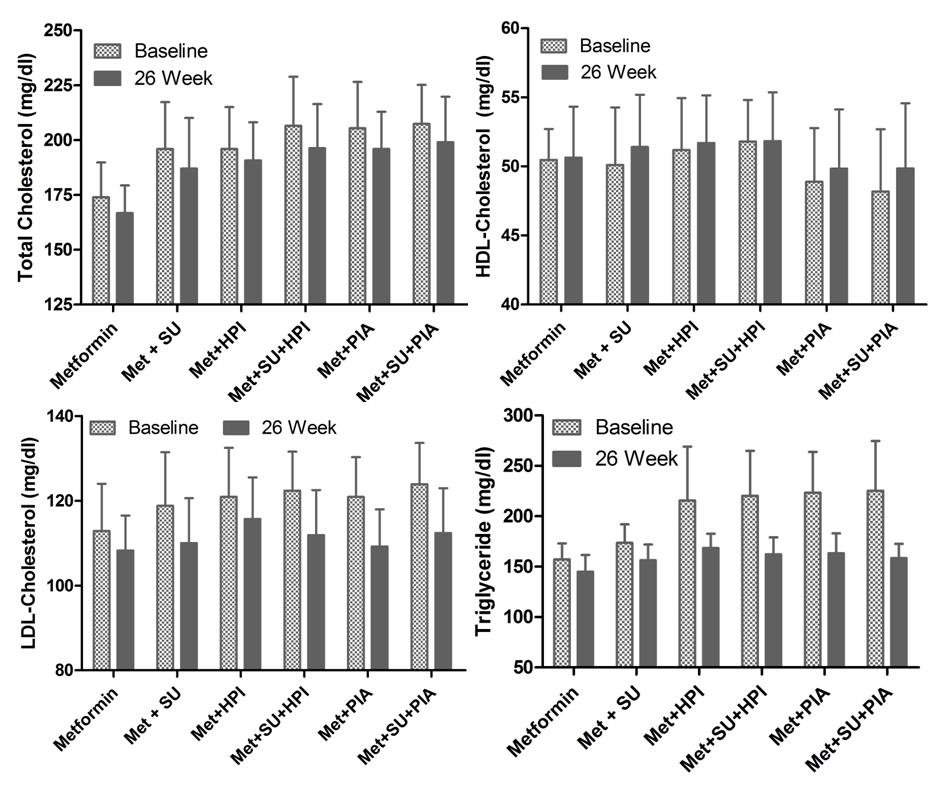 Figure 9. Effect of group combination with metformin on total cholesterol (TC), HDL-Cholesterol, LDL-Cholesterol and Triglyceride (TG) from baseline to week 26 safety analysis set. Values are Mean ± SD followed by paired t tests.
Figure 9. Effect of group combination with metformin on total cholesterol (TC), HDL-Cholesterol, LDL-Cholesterol and Triglyceride (TG) from baseline to week 26 safety analysis set. Values are Mean ± SD followed by paired t tests.
Discussion
In India, diabetes mellitus is creating significant healthcare burden of both family and society because of increase in the level of morbidity and mortality. Nowadays, diabetes is being shown to be associated with a continuum of complications and to be occurring at a relatively younger age in the country (Kaveeshwar and Cornwall, 2014).
India is concerned; the aspects of current profile of diabetic patients and their management for diabetes care have been less focused in the research studies. In DiabCare Asia study, a multi-centre collaborative observational study in Asia shows 50% had poor control on A1C and 54% had late severe complications (Venkataraman et al., 2009).
As per DiabCare India 2011, T2DM begins at a young age in Indians, while patients have sub-optimal glycemic control. This indicates that there is a necessity for increasing awareness regarding advantages of good glycemic control and requirement of more structured intervention at an early stage of the disease. It would not be out of place to emphasize that the status of diabetes care in India needs to be further improved. To the best of knowledge, no such type of profiles has been reported from Gujarat. Nonetheless, the literature on the prevalence of diabetes is available from the south and north India. The main motivation for this analysis was to obtain the risk profile so as to prevent or decrease the burden of T2DM in Gujarat (Mohan et al., 2014; Patel et al., 2011).
In present observational study, total 226 T2DM patients were recruited and out of them, 177 patients had completed the study, 58.2% were male and 41.80% were female. The mean age was 60.61 ± 6.96 years and duration of diabetes mellitus was 5.12 ± 3.16 years.
A1C % level had reduced effectively from the baseline to week 26 by all treatment groups. Statistically significant (p<0.001) reduction in A1C % was seen. The highest percentage of reduction was achieved by premixed insulin analogue and its combination group. As we have discussed the guidelines of ADA and ACE, % A1C should be <6.5 and <7.0 respectively, which shows optimum glycemic target should be achieved.
Analysis of A1C % after 26 weeks of treatment showed that premixed insulin analogue was superior to human premixed insulin. The percentage of patients who were achieved the target <7.0 A1C % was 80.64% (premixed insulin analogue) and 37.5% (human premixed insulin), which indicates that optimum glycemic target was achieved by premixed insulin analogue group as compared to human premixed insulin group. Result represents that human premixed insulin and premixed insulin analogue were achieved the target of ≤6.5 A1C % when it was combined with sulphonylureas. Triple combination of human premixed insulin was given same result as compared to dual combination of premixed insulin analogue on % reduction of A1C. Moreover, dual combination of premixed insulin analogue was significantly reduced A1C % as compared to dual combination of human premixed insulin, indicating dual combination of premixed insulin analogue is good choice of diabetes management compared to dual and triple combination of human premixed insulin.
Insulin groups were significantly reduced fasting and postprandial blood glucose level at 26 week visit. Dual or triple combination of human premixed insulin and premixed insulin analogue group did not showed significant change in % FBG reduction, while triple combination of premixed insulin analogue was showed significant (p<0.01) % PPBG reduction as compared to dual combination of human premixed insulin.
Furthermore, around 20% of patients faced hypoglycemic events in the group of human premixed insulin. Only 3.2% hypoglycemic events were achieved by premixed insulin analogue group, while combination of premixed insulin analogue and sulphonylurea did not show any hypoglycemic events.
Conclusion
For initiating or intensifying insulin therapy in T2DM patients, the pharmacological property of premixed insulin analogue that makes it a possible choice for healthcare practitioners. Premixed insulin analogue lowers A1C to a greater degree and minimize the risk of hypoglycemic events in T2DM patients compared to human insulin. Premixed insulin analogue stands for a simple and effective tool for the modern diabetes management.
Conflicts of interest: None
Acknowledgements
The authors are thankful to patients involved and investigators Dr. Ramesh Goyal (MD Endo), Dr. Vinod Bhatt (MD), Dr. K P Patel (MD), Dr. Kaushal Jani (MD), Dr. Vidyut Shah (MD), Dr. Gajendra Balat (MD) and Dr. Paresh Patel (MD) for active participating in study.
References
American Diabetes Association. 2003. Standards of care for patients with diabetes mellitus. ADA Diabetes Care, 26(1):s33-50.
American Diabetes Association. 2011. Standards of medical care in diabetes 2011. ADA Diabetes Care, 34(1):s11-s61.
Bhalla S, Unnikrishnan R, Srivastava R, Tandon N, Mohan V, Prabhakaran D. 2016. Innovation in capacity building of primary-care physicians in diabetes management in India: a new slant in medical education. Lancet Diabetes Endocrinology, 4(3):200-2.
Chawla A, Chawala R, Jaggi S. 2016. Microvascular and macrovascular complications in diabetes mellitus: Distinct or continuum? Indian Journal of Endocrinology and Metabolism, 20 (4):546-51.
Field S. 2003. AACE diabetes guidelines. Endocrine Practice, 1:5-11.
Helsinki Declaration. 2008. Ethical principles for medical research involving human subjects. Last amended by the 59th WMA General Assembly, Seoul. World medical association.
Hoerger T, Segel J, Gregg E, Saaddine J. 2007. Is glycemic control improving in U.S. adults? Diabetes Care, 31(1):81-6.
ICH International conference on harmonisation. 1996. ICH harmonised tripartite guideline. Good Clinical Practice.
Joshi SR. 2015. Diabetes care in India. Ann Glob Health, 81 (6):830-8.
Juming LU, Linong JI, Xiaohui GUO. 2012. Glycemic control in patients with type 2 diabetes treated with oral antidiabetic drugs in urban area of china. Chinese Journal of Diabetes Mellitus, 4(7):402-6.
Kaveeshwar SA, Cornwall J. 2014. The current state of diabetes mellitus in India. Australas Medical Journal, 7(1):45-8.
Linong JI, Juming LU, Xiaohui GUO. 2003. Status of blood glucose control and treatment of type 2 diabetes in china. Chinese Journal of Diabetes Mellitus, 4(7):397-401.
Mohan V, Shah SN, Joshi SR, Seshiah V, Sahay BK, Banerjee S, Wangnoo SK, Kumar A, Kalra S, Unnikrishnan AG, Sharma SK, Rao PV, Akhtar S, Shetty RV, Das AK. 2014. Current status of management, control, complications and psychosocial aspects of patients with diabetes in India: Results from the DiabCare India 2011 study. Indian Journal of Endocrinology and Metabolism, 18(3):370-8.
Nathan DM, Buse JB, Davidson MB, heine RJ, Holman RR, Sherwin R, Zinman B. 2006. Management of Hyperglycaemia in Type 2 Diabetes: A consensus Algorithm for the Initiation and Adjustment of Therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care, 29:1963-72.
NovoMix® Mix 30/70. 2001. Prescriber’s Information. FDA Approved 11/01/2001.
Pandya H, Lakhani JD, Dadhania J, Trivedi A. 2012. The prevalence and pattern of dyslipidemia among Type 2 diabetic patients at rural based hospital in Gujarat, India. Indian Journal of Clinical Practice, 22 (12):36-44.
Patel M, Patel IM, Patel YM, Rathi SK. 2011. A hospital –based observational study of type 2 diabetic subjects from Gujarat, India. Journal of Health Population and Nutrition, 29(3):265-72.
Rolla A. 2002. Insulin analog mixes in the management of type 2 diabetes mellitus. Practical Diabetology, 21(4):36-43.
Shah S, Benroubi M, Borzi V, Gumprecht J, Kawamori R, Shaban J, Shestakova M, Wenying Y, Valensi P. 2009. Safety and effectiveness of biphasic insulin aspart 30/70 (Novomix 30) when switching from human premix insulin in patients with type 2 diabetes: subgroup analysis from the 6 month IMPROVE observational study. International Journal of Clinical Practice, 63(4):574-82.
Sharma SK, Seshiah V, Sahay BK, Das AK, Rao PV, Shah S, Akhtar S, Shetty R. 2012. Baseline characteristics of the IMPROVE control study population: A study to evaluate the effectiveness of a standardized healthcare professionals training program. Indian Journal of Endocrinology Metabology, 16(2):471–3.
Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, Hadden D, Turner RC, Holman RR. 2000. Association with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ Open, 321(7258):405-12.
UK prospective Diabetes Study (UKPDS) Group. 1998. Intensive blood glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet, 352(9131):837-53.
Valensi P. 2009. Biphasic insulin aspart 30/70 (BIAsp 30) in the treatment of type 1 and type 2 diabetes. Diabetes, Metabolic Syndrome Obesity, 2:61–71.
Venkataraman K, Kannan AT, Mohan V. 2009. Challenges in diabetes management with particular reference to India. International Journal of Diabetes in Developing Countries, 29(3):103-9.