

Ramesh Kumar Verma*, Neeraj Sharma
Department of Pharmaceutical Chemistry, Bhagwant University, Ajmer, Rajasthan, India
*Address for Corresponding Author
Ramesh Kumar Verma
Department of Pharmaceutical Chemistry, Bhagwant University, Ajmer, Rajasthan, India
Abstract
India is very well known for medicinal aromatic plants used for the treatment of various diseases which are day by day diminishing from the nature. Green plants represent a big source of bioactive compounds. Alpinia galanga (Linn.) of the Zingiberaceae family is one of those medicinally important plants. Many countries cultivate this plant, including Indonesia. This plant has numbers of benefits, ranging from being used as a food flavoring, which creates a distinctive aroma in cooking. Active compounds such as 1,8-cineol, α-fenchyl acetate, β-farnesene, β-bisabolene, α-bergamotene, β-pinene, and 1’-acetoxychavicol acetate from the Alpinia galanga plant. Among all the bioactive compounds, 1,8-cineol known as a marker compound in the Alpinia spp provides strong biological activity. This plant can also be used as a treatment for various diseases. The rhizome of the plant is used as a carminative, digestive tonic, anti-emetic, anti-fungal, anti-tumor, Anti-helmintic, anti-diuretic, anti-ulcerative, anti-dementia. The extract of rhizome shows anti-tubercular activity, hypothermia, bronchial catarrh, tonic, stomachic, and stimulant. It is also used as pungent, bitter, heating, stomachic, improve appetite, disease of heart, aphrodisiac tonic, expectorant, used in heal, ache, lumbago, rheumatic pains, chest pain, diabetes, burning of the liver, kidney disease, disinfectants. The rhizome is also used as an anti-microbial, anti-bacterial, anti-inflammatory, and flavoring agent. The seeds are used as cardiotonic, diuretic, hypotonic, gastric lesions, antiplatelet, anti-tumor, and anti-fungal. The tubers of this plant are used as carminative, irritant action, whooping cough in children, Bronchitis, anti-asthma, dyspepsia, fever, and diabetes mellitus.
Keywords: Rasna, Kulanjan, Sugandha Vacha, Greater galangal, Alpinia galanga
Introduction
Treatment with herbal plants is widely used in developing countries, especially in those countries whose communities have a poor economy (Ansari et al., 2020). Herbal medicines are either organic or natural. Pure herbal medicine is obtained from plant extracts that have medicinal benefits, without a mixture of artificial chemicals (synthetic) and without a mixture of animal components. There is currently a rapidly increasing demand for the use of medicines from herbal plants throughout the world (Solikhah et al., 2020).
Plant and plant products are being used as a source of medicine for a long Alpinia galangal wild, family Zingiberaceae is used in medication culinary, and cosmetics for centuries (Bensky et al., 1992; Mohd et al., 2003).
Alpinia galanga is also known as Greater galangal in English and Kulanjan in Hindi. Most the South Indian physicians of traditional Ayurveda and Siddha medicine system use Alpinia galanga to treat various kinds of diseases including diabetes mellitus (Basu and kirtikar, 2001).
The rhizomes are characterized externally by a dark reddish-brown color, and cuttings of the inner rhizome are characterized by the presence of a dark center surrounded by a wider and paler layer on the outer rim, that also darkens considerably when the rhizome is dried during processing. The rhizomes of galanga have a strong aromatic odor and a spicy or pungent taste (Farnsworth et al., 1992).
The seed of A. galanga is used in emaciation and cleaning of the mouth, it stimulates the digestive power and appetite. It is also used as a purgative. Usually, the rhizome is used as a spice and a source of essential oil. Young shoots and flowers are used as vegetables or as spices (Arambewela et al., 2006).
Galangal is widely used to treat breathing diseases, stomach diseases, diarrhea, and stomach cramps. Galangal can also function as an antimicrobial replacement for antibiotics (Mayachiew et al., 2010; Yang and Eilerman et al., 1999). Galangal is also effective for treating fever, abnormal menstruation, and increasing male fertility (Abubakar et al., 2018). Galangal rhizome began to be used in several formulations to prevent cancer and tumors and is also used for the treatment of other diseases such as rheumatism, inflammation, diabetes, and neurological disorders (Arambewela et al., 2006). Galangal is a mixture that has begun to be used by the community to overcome several chronic diseases (Srivastava and Shanker et al., 2012).
From the leaves, stems, rhizomes, and roots of Alpinia galanga, the presence of essential oil is reported. Those are mono and sesquiterpene as well as (E) - methyl cinnamate in nature. They are responsible for the characteristic odour as well as for the reported use in (folk) medicine and in food products of A. galangal (Jirovetz et al., 2003). Alpinia galanga contained flavonoids and volatile oils (Jain et al., 2012; Yu, 1981). The previous studies, the plant possessed many pharmacological activities, including antibacterial, antifungal, antiviral, Antiprotozoal, immunomodulatory, antioxidant effect, antidiabetic, antiplatelet, hypolipidemic, and many other pharmacological effects (De-Pooter et al., 1985; Kiuchi et al., 2002). This review is a combination of chemical constituents, and pharmacological and therapeutic effects of Alpinia galanga based on various current studies.
Taxonomy (Udjiana, 2008)
Kingdom : Plantae
Division : Magnoliophyta
Class : Liliopsida
Subclass : Zingiberidae
Order : Zingiberales
Family : Zingiberaceae
Subfamily : Alpinioideae
Tribe : Alpinieae
Genus : Alpinia
Species : Alpinia galanga
Botanical description
Alpinia galanga (Zingiberaceae) commonly known as Greater galangal; root-stock tuberous, slightly aromatic. Leaves oblong-lanceolate, acute, glabrous, green above, paler beneath, with slightly callus white margins, sheaths long, glabrous; ligule short and rounded. Flowers greenish-white, in dense flowered, 30 cm Panicles; bracts ovate-lanceolate. Calyx tubular, irregularly 3-toothed. Corolla lobes oblong, claw green, blade white, striated with red, rather more than 1 cm long, broadly elliptic, shortly 2-lobed at the apex, with a pair of subulate glands at the base of the apex, with a pair of subulate glands at the base of the claw. Fruit have size of a small cherry, orange red (Gupta, 2010).
 Figure. 1 (a) Whole plant (b) Alpinia galanga Rhizomes and roots of Alpinia galanga
Figure. 1 (a) Whole plant (b) Alpinia galanga Rhizomes and roots of Alpinia galanga
Geographical distribution
It is found in India, China, Indonesia, and Arabic gulf areas, Malaysia, Egypt, and Sri Lanka. It grows in open sunny places, forests, and brushwood. It is commonly cultivated in the mid and low-country in Sri Lanka. The plant is distributed in Himalayas and Southern region of the Western Ghats in India. It is often cultivated in Konkan and North Kanara (Shetty et al., 2015).
Traditional uses
Alpinia galanga is an important medicinal plant in different traditional systems of medicine to treat several diseases, including microbial infections, inflammations, rheumatic pains, chest pain, and dyspepsia, fever, burning of the liver, kidney disease, tumours, diabetes, and even HIV (Ramesh et al., 2011). The plant has an active role in the treatment of eczema, bronchitis, coryza, mobile, pityriasis Versicolor, otitis internal, gastritis, ulcers, and cholera. The seed is used for emaciation and to clean the mouth. It stimulates the digestive power and appetite and acts as a purgative. The rhizome is generally used as a spice. It is also a good source of essential oil. The flowers and young shoots are also used as a vegetable or as a spice (Arambewela et al., 2006).
Active compounds
Active compounds from the various parts A. galanga were widely studied by many researchers. Many active compounds were successfully isolated and identified by previous researchers. The major active compounds found in A. galanga are 1,8-cineol, α-fenchyl acetate, β-farnesene, β-bisabolene, α-bergamotene, β-pinene, and 1’-acetoxychavicol acetate. 1, 8-cineole is known as a marker compound for Alpinia spp and was reported as the most abundant compound in most of the studies on A. galanga (Abdullah et al., 2015).
Phytochemistry Chemical investigations of Alpinia galanga include galango flavonoid, 1’S-1’-acetoxychavicol acetate (ACE), phenylpropanoids and phydroxybenzaldehyde (1’S-1’- acetoxychavicol acetate and 1’S-1’-acetoxyeuginol acetate), acetoxycineoles (trans and cis)-2-and 3-acetoxy- 1, 1, 8-cineoles, 1’-acetoxychavicol acetate (galangal acetate), β-Sitosterol diglucoside (AG-7) and β-sitsteryl Arabinoside (AG-8), hydroxy-1,8-cineole glucopyranosides, (1R, 2R, 4S)-and (1S, 2S, 4R)-trans-2-hydroxy-1,8-cineole β-D-glucopyranoside, and (1R, 3S, 4S)-trans-3-hydroxy-1, 8- cineole β-D-glucopyranoside (Abdullah, et al., 2015; Morikawa et al., 2005; Misawa et al., 2008).
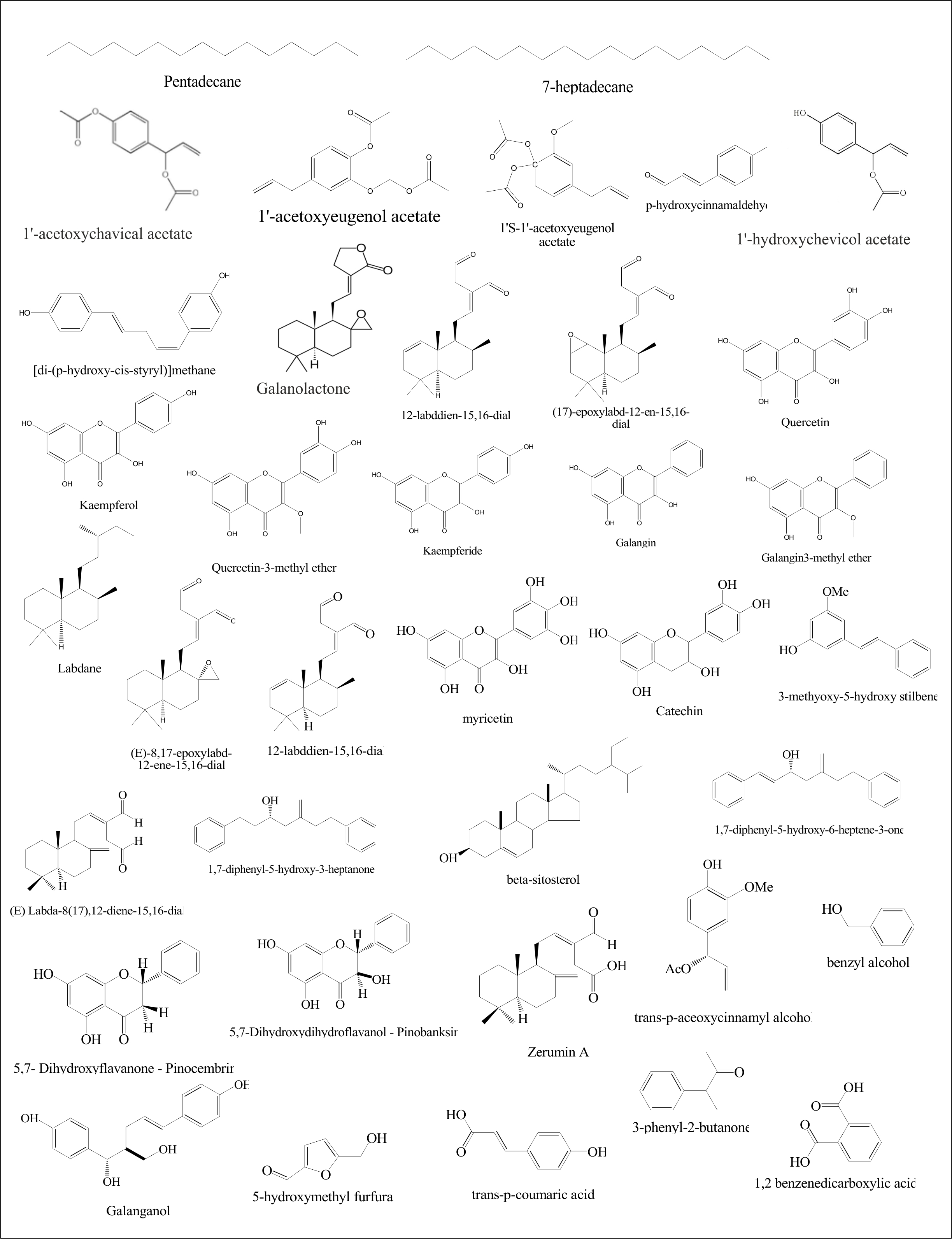 Figure 2. Chemical structures of phytoconstituents of Alpinia galanga
Figure 2. Chemical structures of phytoconstituents of Alpinia galanga
Pharmacological activities
Anti-allergic Activity
The Antiallergic activity of 80% aqueous acetone extract of the rhizomes of Alpinia galanga which was found to inhibit release of β -hexosaminidase, as a marker of antigen-IgE-mediated degranulation in RBL-2H3 cells (Matsuda et al., 2003). Compounds 1 'S-1'-acetoxychavicol acetate and 1 'S-1'-acetoxyeugenol acetate isolated from Alpinia galanga rhizome, were also examined for their antiallergy activities in RBL-2H3 cells (Yoshikawa et al., 2004).
Anti-cancer Activity
The DL-1'-Acetoxychavicol acetate (I) and DL-1'-acetoxyeugenol acetate (II) isolated from Alpinia galanga evaluated anti-tumor activity against Sarcoma 180 ascites in mice (Itokawa et al., 1987) and also evaluated the anti-tumor activity of diterpene compounds I (15α- and 15β-isomer) and II, isolated from the seeds of Alpinia galanga (Itokawa and Morita, 1988). The two new skeletal diterpenes, named galanal A (I), B (II), and two new labdane-type diterpenes, named galanolactone (III), (E)-8β(17),12-labddiene-15,16-dial, isolated from Alpinia galanga act as cytotoxic activity (Morita and Itokawa, 1988). The Ethyl trans-cinnamate (III) and Et 4-methoxy-trans-cinnamate (IV) from galanga root oil, which was found to exhibit significant activity in the mouse liver and intestines (Zheng et al., 1993). The ethanolic extract by brine shrimp lethality bioassay of Alpinia galanga (Khattak et al., 2005), [MCF7 (breast adenocarcinoma) and LS174T (colon adenocarcinoma)] cell lines of the methanol extracts, water extracts and volatile oils of the fresh rhizomes of Alpinia galanga (Zaeoung et al., 2005). The mechanism of cell death of human leukemic HL-60 and U937 cells induced by 4’-hydroxycinnamaldehyde (4’-HCA) isolated from Alpinia galanga. 4’-HCA was found to be cytotoxic to both cell lines in a dose-dependent manner (p<0.05) as demonstrated by MTT assay (Banjerdpongchai et al., 2011).
Anti-diabetic Activity
The methanolic extract of Alpinia galanga rhizome shows the hypoglycemic activity in rabbits. Which significantly lowered the blood glucose (Akhtar et al., 2002). The ethanolic extract of rhizome of Alpinia galanga (EEAG) in normoglycaemic and hyperglycemic rats. Single-dose of EEAG (50, 100, and 200 mg/kg) was administered orally in normoglycaemic, glucose (1.5 g/kg) fed hyperglycemic and alloxan-induced diabetic rats (n=5). Single-dose administration of EEAG (200 mg/kg) induced a significant (P<0.05) decrease in blood glucose level in glucose-fed hyperglycemic rats at ½ h compared to control glucose-fed rats and in alloxan-induced diabetic rats at 6h after EEAG treatment (Chudiwal et al., 2008), and of phenolic and methanolic also act as Antidiabetic and anti-inflammatory activities of extract of rhizome of Alpinia galanga (Jaju et al., 2009).
Anti-inflammatory activity
The total alcoholic extract (TAE) and total aqueous extracts (TAQ) of Alpinia galanga rhizomes evaluated in acute (carrageenan-induced paw edema; M1) and sub-acute (cotton-pellet-induced granuloma; M2) rat models (Satish et al., 2003; Nagashekhar et al., 2006). Antinociceptive activity were found the ethanolic (95%) extract of rhizome of Alpinia galanga (EEAG). EEAG was examined for analgesic activity using hot plate, formalin-induced paw licking, and acetic acid-induced writhing method (doses 200 mg/kg and 400 mg/kg administered orally) Shivgunde et al., 2008). Yu also reported p-coumaryl diacetate (CDA) was found to have an anti-inflammatory activity which was isolated from Alpinia galanga (Yu et al., 2009). The Anti-arthritic activity of petroleum ether, chloroform, alcoholic extracts of the Alpinia galanga rhizomes in the presence of chemically active compounds by standard methods and evaluated for their antiarthritic activity by using Complete Freund’s Adjuvant (CFA) induced rat model. Application of all the three extracts exhibited statistically significant edema inhibition when compared with the arthritic control group (Chandur et al., 2010). The anti-psoriasis activity of ethanolic extract of Alpinia galanga by using a HaCaT keratinocyte cell line as an in-vitro model (Chanachai et al., 2011). Petroleum ether, Chloroform, Methanolic, and Aqueous methanolic (1:1) extracts of Alpinia galanga in carrageenan-induced paw edema in Wistar rats and compared to a positive control drug-using Ibuprofen. These extracts were given orally at a concentration of 500 mg/kg between 1 hour before carrageenan injection. Methanolic extract of Alpinia galanga showed maximum inhibition of 79.51 % on carrageenan-induced rat paw edema (Unnisa and Thahera, 2011). The ethanolic extract of A. galanga rhizome by scientifically validated anti-inflammatory screening technique on rats by carrageenan-induced pleurisy rats. the ethanolic extract had significant activity in rats in all the tested groups A. galanga 100, 200, and 400 mg with P < 0.005 compared to that of control (Subash et al., 2016).
Antimicrobial activity
The antifungal activities of two new skeletal diterpenes, named galanal A (I) B (II), and two new labdane-type diterpenes, named galanolactone (III), (E)-8β(17),12-labddiene-15,16-dial, isolated from Alpinia galanga (Morita and Itokawa, 1988). The antibacterial activity against different multi-resistant Gram-positive and Gram-negative bacteria of ether and ethyl acetate extracts of Alpinia galanga. Both extracts of Alpinia galanga had significant effects on Staphylococcus aureus and Klebsiella pneumoniae (Elsamma et al., 1996). The anti-fungal activity of essential oils, of Alpinia galanga against five dermatophytes (Trichophyton mentagrophytes, T. rubrum, Microsporum canis, Microsporum nanum and Epidermophyton floccosum), three filamentous fungi (Aspergillus niger, Aspergillus fumigatus and Mucor sp.) and five strains of yeast (Saccharomyces cerevisiae, Cryptococcus neoformans, Candida albicans, Candida tropicalis and Torulopsis glabrata). The anti-fungal property evaluation was carried out by broth microdilution and disc gel diffusion methods (Ibrahim Jantan et al., 2003). Endophytic actinomycetes activity of roots of Alpinia galanga against phytopathogenic fungi (Colletotrichum musae and Fusarium oxysporum), and tested against Candida albicans. The strain identified as Streptomyces aureofaciens CMUAc130 was the most effective in antifungal activity among those investigated (Taechowisan and Lumyong, 2003).
The antifungal activities against Trichophyton longifusum [Keratinomyces longifusus] (65% and 60%, respectively) from the ethanolic extract of Alpinia galanga. This extract was found quite inert in antibacterial bioassay involving Escherichia coli, Bacillus subtilis, Shigella flexneri, Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella typhi (Khattak et al., 2005). The anti-microbial action of ethanolic extract of Alpinia galanga (galangal) on Staphylococcus aureus 209P and Escherichia coli NIHJ JC-2 by using an agar disc diffusion assay. The galangal extract had the strongest inhibitory effect against Staphylococcus aureus (Oonmetta-aree, 2006). The anti-amoebic activity of chloroform, methanol, and water extracts from Alpinia galanga. The extracts were incubated with 2x10(5) E. histolytica trophozoites/ml of the medium at 37ºC under anaerobic conditions for 24 h. The cultures were examined with an inverted microscope and scored (1-4) according to the appearance and numbers of the trophozoites. The IC (50) of a standard drug, metronidazole, was 1.1 µg/ml (Sawangjaroen et al., 2006).
The Anti-bacterial effect of essential oil of Alpinia galanga was obtained by hydro-distillation and two different solvent extractions (petroleum ether and ethanol) against Escherichia coli, Staphylococcus aureus, Bacillus cereus and Listeria monocytogenes which were tested by a disc diffusion assay (Krittika et al., 2007). The antimicrobial activity of galangal (Alpinia galanga) extract using disc diffusion and agar dilution methods against Staphylococcus aureus. The minimum inhibitory concentration (MIC) value of galangal extracts were found to be 13.97 and 0.78 mg/ml and the minimum biocidal concentration (MBC) value was 2.34 mg/ml, respectively (Mayachiew and Devahastin, 2008). The antimicrobial activity of different solvent extracts of Alpinia galanga against gram-positive and gram-negative bacteria and some fungal strains was studied (Rao et al., 2008). The anti-fungal activity of crude ethanolic extract of Alpinia galanga rhizomes which was tested against selected zoonotic dermatophytes (Microsporum canis, Microsporum gypseum, and Trichophyton mentagrophyte) and the yeast-like Candida albicans. A broth dilution method was employed to determine the inhibitory effect of the extract and compared it to those of ketoconazole and griseofulvin (Trakranrungsie et al., 2008).
The crude acetone extract of the rhizomes of Alpinia galanga exhibited anti-plasmid activity against Salmonella typhi, Escherichia coli and vancomycin-resistant Enterococcus faecalis with an efficiency of 92%, 82% and 8% at 400 micro g/ml SIC respectively (Latha et al., 2009). The anti-bacterial activity of essential oil of Alpinia galanga against Escherichia coli, Staphylococcus aureus, Bacillus cereus, and Listeria monocytogenes which were tested by a disk diffusion assay (Natta et al., 2009). Anti-microbial activity of various extracts of Alpinia galanga which were screened against the common foodborne bacteria such as Escherichia coli, Salmonella enteriditis, Clostridium perfringens, Staphylococcus aureus, Campylobacter jejuni, Bacillus cereus and fungi such as Saccharomyces cerevisiae, Hansenula anomala, Mucor mucedo, Candida albicans using disc diffusion method (Sunilson et al., 2009). The anti-fungal activity of methanol extract of flowers of Alpinia galanga against Micrococcus luteus and Aspergillus niger which showed the largest zone of inhibition. The antimicrobial activity was also screened by using the disc diffusion method (Wong et al., 2009). The antifungal activity of the leaf extracts of Alpinia galanga, which was evaluated on the plant pathogenic fungus; C. gloeosporioides isolated from mango. Different antifungal assays were employed, i.e. Agar-Disc Dilution assay as the primary screening assay, followed by determination of Minimum Inhibition Concentration (MIC), and the rate of sporulation assay. Methanol crude extract reduced the radial growth of C. gloeosporioides by 66.39%, followed by chloroform crude extract 63.26%, and 61.56% for acetone crude extracts (Johnny et al., 2010). The anti-bacterial activity of methanol, acetone, and Di-ethyl ether extract of Alpinia galanga against pathogens viz. Bacillus subtilis MTCC 2391, Enterobacter aerogene, Enterobacter cloacae, Enterococcus faecalis, Escherichia coli MTCC 1563, Klebsiella pneumoniae, Pseudomonas aeruginosa MTCC 6642, Salmonella typhimurium, Staphylococcus aureus and Streptococcus epidermis using agar well diffusion method (Rao et al., 2010).
Antimicrobial activity of hexane, ethanolic and aqueous extracts from galangal (Alpinia galanga) which had strong antimicrobial activity against Staphylococcus aureus and/or Listeria monocytogenes (Weerakkody et al., 2010). The antimicrobial activity of acetone, ethanolic and aqueous extract A. galanga exhibits anti-Mycobacterium tuberculosis activity with multiple modes of action. Since the activity of the extracts was observed under reducing oxygen concentrations, it may be effective in treating the dormant and non-replicating bacteria of latent TB (Gupta et al., 2014).
The ethanolic extract prevents the infectious diseases Vibrio harveyi and white spot syndrome virus in Pacific white shrimp (Litopenaeus vannamei). A commercial diet mixed with galangal ethanol extract was fed to shrimp for 1 or 2 months. The number of V. harveyi in the hemolymph of the galangal diet group was significantly lower than that in the control diet group (P < 0.05), indicating the higher clearance ability of the galangal diet group (Chaweepack et al., 2015). The Minimum Inhibitory Concentration (MIC) of essential oils and crude extracts was evaluated by broth dilution method against foodborne bacteria Bacillus subtilis, Escherichia coli, Staphylococcus aureus, Salmonella Typhimurium, and Vibrio cholera. MIC of crude extract and essential oils of galangal and ginger against all tested microorganisms were relatively high (Hamad et al., 2016). The methanolic extract of Alpinia galanga exhibited antibacterial activity against all the three tested gram-positive bacterial strains. The minimum inhibitory concentration of A. galangal against S. aureus, S. epidermidis, and L. monocytogenes was less than 1mg/ml. Based on the result obtained, A. galangal has higher total phenolic content (122 ±2.6 mg GE/g), followed by total flavonoid content (110 ± 4.4 mg QE/g). However, the DPPH content in this plant extract was the least with a value of 20 ± 1.0 mg TE/g (Muniandy et al., 2019).
Antioxidant activity
1'-Acetoxychavicol acetate (ACA) is a unique phenylpropanoid compound in Alpinia galanga that has been evaluated as an effective aroma, pungent and anti-oxidative component. In addition, the anti-oxidative activity of linoleic acid was examined. Although ACA indicated the highest activity, related compounds also showed significant anti-oxidative activity (Kubota et al., 2001). The antioxidant activity of ethanolic extract of Galangal (Alpinia galanga). Antioxidant activity of extract at neutral pH was higher than at acidic pH ranges (Juntachote and Berghofer, 2005). Antioxidant activity of the methanol, aqueous extracts, and volatile oil of the fresh rhizomes of Alpinia galanga, which was assessed for free radical scavenging activity against 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical (Zaeoung et al., 2005). Antioxidant activity of rhizomes of Alpinia galanga, measured by DPPH and β-carotene-linoleic acid method after extraction with two different solvents- methanol and dichloromethane (Vankar et al., 2006).
The antioxidant activity of the essential oils of Alpinia galanga was determined by using two complementary methods: 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay and 2,2’-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) free radical decolorization assay. The results obtained indicated that the essential oil of Alpinia galanga possessed stronger antioxidant activity with the IC50 values of 550 and 3721 μg/ml, respectively (Chowwanapoonpohn and Tachakittirungrod, 2007). Antioxidant and tyrosinase inhibition properties of leaves and rhizomes of Alpinia galanga by using total phenolic content (TPC) and Ascorbic acid equivalent antioxidant capacity (AEAC) methods. The most outstanding was the FIC value of Alpinia galanga leaves which was more than 20 times higher than that of rhizomes. It displayed the strongest tyrosinase inhibition activity (Chan et al., 2008). The antioxidant activity of galangal (Alpinia galanga) extract, which was evaluated by the β-carotene bleaching method, was 70.3%, respectively (Mayachiew and Devahastin, 2008).
The naturally occurring phenolic compounds, such as chavicol analogs, have been shown to have potent antioxidant activity in the rhizomes of Alpinia galanga (Jung et al., 2009). The p-coumaryl diacetate (CDA) isolated from Alpinia galanga was found to have antioxidant activity (Yu et al., 2009). The antioxidant activity of methanol extract of Alpinia galanga leaves was evaluated for total phenolic content. The AOA was investigated using 1,1-diphenyl-2-picrylhydrazyl (DPPH), reducing power (RP), ferrous ion chelating as well as β -carotene bleaching assays. It was found that Alpinia galanga leaves and flowers showed the highest chelating and β-carotene bleaching abilities (Wong et al., 2009). Antioxidant Activity of ethanolic extract of Alpinia galanga which showed the potent scavenging activity by DPPH method with the IC50 value of 69.5±1.375 μg/ml, by lipid peroxidation method with the IC 50 value of 77±1.876 μg/ml, hydrogen peroxide radical scavenging activity with the IC 50 value 55±1.59 μg/ml, ABTS radical scavenging method with the IC 50 value 0.086±1.10 μg/ml (Srividya et al., 2010).
The methanolic extracts of galangal samples irradiated showed the highest antioxidant activity and phenolic compounds compared with those from non-irradiated samples (Araby et al., 2013), it was measured by 1, 1-diphenyl-2-picrylhydrazyl (DPPH) scavenging potential and Ferric reducing antioxidant power (FRAP) potential (Avasthi et al., 2015). The aqueous extract was filtered and used for DPPH, FRAP, TPC, and MDA tests to determine its antioxidant value (Hasan et al., 2020).
Anthelmintic Activity
The anthelmintic activity on alcoholic extracts of rhizomes of Alpinia galanga showed good in-vitro anthelmintic activity against human Ascaris lumbricoides (Raj,1975). The ethanolic extracts from the leaves of Eupatorium triplinerve and the rhizome of Alpinia galanga were compared for their anthelmintic activities, based on traditional claims (Subhash et al., 2012).
Anti-viral Activity
The anti-HIV agents and inhibitors for nuclear export of HIV-derived proteins such as Rev essential to HIV replication contain 1'-acetoxychavicol acetate (I). Thus, 1'-acetoxychavicol acetate, isolated from a Methanolic extract of Alpinia galanga fruits, inhibited the replication of HIV-1 NL43 in MT-4 cells without affecting the viability of MT-4 cells (Murakami and Tamura, 2005). The antiviral activity of methanol and aqueous extracts of Alpinia galanga, inhibited proteases from human immunodeficiency virus type 1 (HIV-1), hepatitis C virus (HCV), and human cytomegalovirus (HCMV) and it was found that methanol extract inhibits the enzymes more effectively than the aqueous extract (Sookkongwaree et al., 2006).
Anti-HIV activity of 1'S-1'-acetoxychavicol acetate isolated from Alpinia galanga rhizomes extract by blocking Reverse Transport (Ye and Li, 2006). Rev-export inhibitor (ACA) from the medicinal plant Alpinia galanga clarified the formation of the quinone methide intermediate ii to be essential for exerting the inhibitory activity (Murkami et al., 2010).
Hepatoprotective Activity
The hepatoprotective effect of the crude extract of Alpinia galanga at 200 and 400 mg kg-1 against paracetamol-induced hepatotoxicity in rats. The findings from the study showed that the crude extract of Alpinia galanga has protective effects against paracetamol-induced hepatotoxicity (Hemabarathy et al., 2009).
Analgesic Activity
Analgesic activity of ethanolic extract of Alpinia galanga percentage inhibition rate of Aspirin (100mg/kg) was 82.15% compared to Alpinia galanga (100mg/kg) 19.63%, (200mg/kg) 33.02% and (400mg/kg) 57.13% by acetic acid-induced abdominal constrictions antinociceptive mice model. Alpinia galanga 400mg/kg (71.70%) had comparable percentage inhibition of nociception to standard group indomethacin (88.71%) in the formalin-induced nociceptive mice model. Among 20 compounds screened for pharmacokinetic and drug-like features, Galanal B had the binding free energy -56.664 when compared to control compound 2AZ5-56.000 (Subash et al., 2018). Ethanolic extract of Alpinia galanga rhizomes was given orally to experimental animals. EEAGR was evaluated for central analgesic activity by using the tail-flick method and peripheral analgesic activity by using the acetic acid-induced writhing test using aspirin (300 mg/kg b.w and 100 mg/kg b.w orally) as the standard drug respectively (Dasari et al., 2018).
Miscellaneous Activity
The Gastric antisecretory, antiulcer and cytoprotective properties of ethanolic extract of Alpinia galanga Wild. in rats. Which showed significantly reduced gastric secretion and marked cytoprotective activity; it was suggested that these properties may be responsible for the antiulcer activity of Alpinia galanga (Al-Yahya et al., 1990). The gastroprotective activity and the effects of 1'S-1'-acetoxychavicol acetate and related phenylpropanoids isolated from the rhizomes of Alpinia galanga on ethanol-induced gastric lesions in rats (Matsuda et al., 2003). Aphrodisiac activity of an isolated compound named (3)-shogaol from the extract of Alpinia galanga in guinea pigs (Jean and Cariel, 2002). The treatment on cytological and biochemical changes induced by cyclophosphamide in mice by the effect of Alpinia galanga from the ethanolic extract. The rhizomes of Alpinia galanga were also used to treat dyspepsia, gastralgia, seasickness, abdominal colic, and digestive and tonic (Qureshi et al., 1994). The insecticidal activity of 1'-acetoxychavicol acetate from the rhizome of Alpinia galanga had a molecular formula of C13H14O4 (Dadang and Ohsawa, 1998). The platelet activity from Alpinia galanga and their inhibitory effects on platelet-activating factor (PAF) binding to rabbit platelets, using 3H-PAF as a ligand (Jantan et al., 2005).
The anti-giardial activity of chloroform, methanol, and water extracts of the Alpinia galanga plant. The plant extracts and a standard drug, metronidazole, were incubated with 2x10(5) trophozoites of Giardia intestinalis per milliliter of growth medium in 96-well tissue culture plates under anaerobic conditions for 24 h. The cultures were examined with an inverted microscope and the minimum inhibitory concentration and the IC50 value for each extract were determined. The chloroform extracts from Alpinia galanga as "active", i.e. with an IC50 of <100 µg/ml (Sawangjaroen et al., 2005).
The effects of Alpinia galanga extract on metabolism and gene expression involved in the interleukin-1β (IL-1β) response of human chondrocyte and synovial fibroblast. Alpinia galanga extract was also found to inhibit IL-1β enhanced matrix breakdown of the cartilage explants in a dose-dependent manner (Pothacharoen et al., 2006). Antileishmanial activity of the (hexane, chloroform, and ethyl acetate) extracts and isolated constituents of rhizome of Alpinia galanga. Twelve compounds namely, methyleugenol (1), p-coumaryl diacetate (2), 1'-acetoxychavicol acetate (3), 1'-acetoxyeugenol acetate (4), trans-p-acetoxycinnamyl alc. (5), trans-3,4-dimethoxycinnamyl alc. (6), p-hydroxybenzaldehyde (7), p-hydroxycinnamaldehyde (8), trans-p-coumaryl alc. (9), galangin (10), trans-p-coumaric acid (11), and galanganol B (12) were isolated from these extracts. These compounds 2, 3, 4, and 5 were found most active in-vitro against promastigotes of L. donovani with IC50 values of 39.3, 32.9, 18.9, and 79.9 µM respectively (Kaur et al., 2010). The Neuroprotective Effect of Alpinia galanga (L.) fractions on Aβ(25–35) induced amnesia in mice. The increased habituation memory and decreased escape latency in behavioral parameters are indicative of the cognitive enhancement after treatment with Alpinia galanga fractions. Increment in Na+/K+-ATPase and antioxidant activity depicts brain membrane integrity improvement and free radical scavenging property. AChE level was decreased to improve cognition by enhancing cholinergic transmission (Singha et al., 2011). Two compounds, 1,7-bis(4-hydroxyphenyl)-1,4,6-heptatrien-3-one (BHPHTO) and bisdemethoxycurcumin (BDMC) from the rhizome of Alpinia galanga both the compounds on the human melanoma A2058 and showed that significantly inhibited the proliferation of melanoma cells in the cell viability assay. This research was also taken on the tests of B16-F10 cell line and showed minor inhibitory consequences of cellular tyrosinase activities and melanin contents (Lo et al., 2013).
Conclusion
From the various scientific research based on Alpinia galanga, the plant has a huge biological potential. Alpinia galanga is a common herbal plant, is widely used as a treatment for various diseases, and has a diverse pharmacological spectrum. Several chemicals present in the plant show wide pharmacological and medicinal properties. More research and evaluation needs to be done to isolate and identify different chemicals present in the plant which will be used for innumerable application for human welfare in the near future.
Conflict of interest: None
References
Abdullah F, Subramanian P, Ibrahim H, Malek SNA, Lee GS, Hong SL. 2015. Chemical Composition, Antifeedant, Repellent, and Toxicity Activities of the Rhizomes of Galangal, Alpinia galanga Against Asian Subterranean Termites, Coptotermes gestroi and Coptotermes curvignathus (Isoptera: Rhinotermitidae). Journal of Insect Science 15:1-7.
Abubakar IB, Malami I, Yahaya Y, Sule SM. 2018. A Review on The Ethnomedicinal Uses, Phytochemistry and Pharmacology of Alpinia officinarum Hance. Journal of Ethnopharmacology, 224:45-62.
Afzal U, Thahera Parveen D. 2011. Anti-inflammatory and acute toxicity studies of the extracts from the rhizomes of Alpinia galanga Willd, Pelagia Research Library Der Pharmacia Sinica, 2(2):361-367.
Akhtar MS, Khan MA, Malik MT. 2002. Hypoglycaemic activity of Alpinia galanga rhizome and its extracts in rabbits, Fitoterapia, 73(7/8):623-628.
Al-Yahya MA, Rafatullah S, Mossa JS, Ageel AM, Al-Said MS, Tariq M. 1990. Gastric antisecretory, antiulcer and cytoprotective properties of ethanolic extract of Alpinia galanga Willd. in rats, Phytotherapy Research, 4(3):112-114.
Ansori ANM, Fadholly A, Hayaza S, Joko R, Susilo K, Inayatillah B, Winarni D, Husen SA. 2020. A Review on Medicinal Properties Of Mangosteen (Garcinia Mangostana L.). Research Journal Pharmacy And Technology, 13(2): 974-82.
Arambewela L, Wijesinghe A. 2006. Sri Lankan Medicinal Plant Monographs and Analysis - Alpinia galanga.
Arambewela L, Wijesinghe A. 2006. Sri Lankan Medicinal Plant Monographs and Analysis - Alpinia galanga.
Arambewela LS, Arawwawala M, Owen NL, Jarvis B. 2007. Volatile oil of Alpinia galanga Willd. of Sri Lanka. Journal of Essential Oil Research, 19(5):455-6.
Arambewela LS, Wijesinghe A. 2006. Sri Lankan Medicinal Plant Monograph and Analysis: Alpinia galanga (10th ed.). Industrial Technology Institute and National Science Foundation: Colombo.
Avasthi Sharma A, Jain S, Bhatnagar M, Ghosal S. 2015. Invitro antibacterial, antifungal, antioxidant and antihemolytic activities of Alpinia galanga, International journal of phytomedicine, 7:78-89.
Banjerdpongchai R, Punyati P, Nakrob A, Pompimon W, Kongtawelert P. 2011. 4’-Hydroxycinnamaldehyde from Alpinia galanga (Linn.) Induces Human Leukemic Cell Apoptosis via Mitochondrial and Endoplasmic Reticulum Stress Pathways, Asian Pacific Journal of Cancer Prevention, 12:593-598.
Basri AM, Taha H, Ahmad N. 2017. A review on the pharmacological activities and phytochemicals of Alpinia officinarum (Galangal) extracts derived from bioassay-guided fractionation and isolation. Pharmacognosy Reviews, 11(21):43-56.
Basu kirtikar. 2001. Indian Medicinal Plant, Second Edition, Volume 10, pp. 3378.
Bendjeddou D, Lalaoui K, Satta D. 2003. The immunostimulating activity of the hot water-soluble polysaccharide extracts of Anacydus pyrethrum, Alpinia galanga and Citrullus colocynthis. Journal of Ethnopharmacology, 88(2-3):155-160.
Bensky and Gamble 1992. Chines Materia Medica, Eastland Press, Scattle, WA, pp. 307.
Chan EWC, Lim YY, Wong LF, Lianto FS, Wong SK, Lim KK, Joe CE, Lim TY. 2008. Antioxidant and tyrosinase inhibition properties of leaves and rhizomes of ginger species, Food Chemistry, 109:477–483.
Chanachai S, Visa T, Tewin T. 2011. Effects of Thai Medicinal Herb Extracts with Anti-Psoriatic Activity on the Expression on NF-κB Signaling Biomarkers in HaCaT Keratinocytes, Molecules, 16:3908-3932.
Chandur U, Shashidhar S, Chandrasekar SB, Narasima Rao M. 2010. Phytochemical evaluation and screening of Anti-arthritic activity of Alpinia galanga (Linn.), International Journal of Pharmaceutical Sciences, 2(2):593-597.
Chaweepack T, Khomvilai C, Chaweepack S, Kamei K. 2015. Effect of Galangal (Alpinia galanga Linn.) Extract on the Growth Rate and Resistance to Vibrio harveyi and White Spot Diseases in Pacific White Shrimp (Litopenaeus vannamei), Journal of Agricultural Science, 7:9.
Chudiwal AK, Jain DP, Jain KS, Singhai AK, Somani RS. 2008. Antidiabetic potential of rhizomes of Alpinia galanga in alloxan- induced diabetic rats, Indian Journal of Pharmacology, 40(2):171.
Dadang Riyanto S, Ohsawa K. 1998. Lethal and antifeedant substance from the rhizome of Alpinia galanga Sw. (Zingiberaceae), Journal of Pesticide Science, 23(3):304-307.
Dasari S, Mohd R, Abdul. 2018. Evaluation of analgesic activity of ethanolic extract of Alpinia galanga rhizomes in experimental animal models, Indian Journal of Pharmacy and Pharmacology, 5(4):164-169.
De-Pooter HL, Omar MN, Coolsaet BA, Schamp NM. 1985. The essential oil of greater galanga (Alpinia galanga) from Malaysia. Phytochemistry, 24(1):93-6.
Elsamma T, Shanmugam J, Rafi MM. 1996. Antibacterial activity of plants belonging to Zingiberaceae family, Biomedicine, 16(2/3):15-20.
Eman A, Hoda GM, Ali, Abdeldaiem MH. 2013. Evaluation of Irradiated Galangal Rhizomes (Alpinia galanga) as natural food preservatives (Antioxidant and Antimicrobial) Journal of Applied Sciences Research, 9(1):743-757.
Farnsworth NR, Bunyapraphatsara N. 1992.Thai medicinal plants recommended for primary healthcare system, Medicinal Plants Information Center, Faculty of Pharmacy, Mahidol University, 402.
Gupta P, Bhatter P, D'souza D, Tolani M, Daswani P, Tetali P, Birdi T. 2014. Evaluating the Anti-Mycobacterium tuberculosis activity of Alpinia galanga (L.) Willd. Axenically under reducing oxygen conditions and in intracellular assays, BMC Complementary and Alternative Medicine, 14(84):1-8.
Gupta RK. 2010. Medicinal and Aromatic Plants. 1st edition CBS Publisher and Distributors Pvt. Ltd. New Delhi. pp. 468-9.
Hamad A, Alifah A, Permadi A, Hartanti D. 2016. Chemical constituents and antibacterial activities of crude extract and essential oils of Alpinia galanga and Zingiber officinale International Food Research Journal, 23(2):837-841.
Hemabarathy B, Siti B, Budin Feizal V. 2009. Paracetamol hepatotoxicity in rats treated with crude extract of Alpinia galanga, Journal of Biological Sciences, 9(1):57-62.
Indrayan A, Agrawal P, Rathi AK, Shatru A. 2009. Nutritive Value of Some Indigenous Plant Rhizomes Resembling Ginger. Natural product radiance, 8(5):507-13.
Itokawa H, Morita H, Sumitomo T, Totsuka N, Takeya K. 1987. Anti-tumour principles from Alpinia galanga, Planta Medica, 53(1):32-3.
Itokawa H, Morita H. 1988. Isolation of antitumor diterpenes from Alpinia, Japan, Kokai Tokkyo Koho, pp. 5.
Jaju S, Indurwade N, Sakarkar D, Fuloria N, Ali M. 2009. Isolation of galangogalloside from rhizomes of Alpinia galanga, International Journal of Green Pharmacy, 3(2):144-147.
Jantan I, Mohd Y, Chen MS. 2003. Antifungal activity of the essential oils of nine Zingiberaceae species, Pharmaceutical Biology, 41(5):392-397.
Jantan I, Rafi IAA, Jalil J. 2005. Platelet-activating factor (PAF) receptor-binding antagonist activity of Malaysian medicinal plants, International journal of phytotherapy and phytopharmacology, 12(1-2):88-92.
Jean D, Cariel L. 2002. Use of one or more shogaol(s) as an aphrodisiac, PCT International applications, pp. 12.
Jirovetz L, Buchbauer G, Shafi MP, Leela NK. 2003. Analysis of the essential oils of the leaves, stems, rhizomes, and roots of the medicinal plant Alpinia galanga from southern India. Acta Pharmaceutica, 53(2):73-82.
Johnny L, Kalsom UY, Rosimah N. 2010. The effect of herbal plant extracts on the growth and sporulation of Colletotrichum gloeosporioides, Journal of Applied Biosciences, 34:2218-2224.
Juntachote T, Berghofer E. 2005. Anti-oxidative properties and stability of ethanolic extracts of Holy basil and Galangal, Food Chemistry, 92(2):193-202.
Kaur A, Singh R, Dey CS, Sharma SS, Bhutani KK, Singh IP. 2010. Antileishmanial phenylpropanoids from Alpinia galanga (Linn.) Willd, Indian Journal of Experimental Biology, 48(3):314-317.
Khattak S, Shah SR, Waqar Ahmad HU, Ahmad M. 2005. Biological effects of indigenous medicinal plants Curcuma longa and Alpinia galanga, Fitoterapia, 76(2):254-257.
Kiuchi F, Keniji M, Itano Y, Ito M, Honda G, Qui TK. 2002. Screening of Natural Medicines Used in Vietnam for Trypanocidal Activity. Nature Medicine, 56(2):64-8.
Kubota K, Ueda Y, Yasuda M, Masuda A. 2001. Occurrence and antioxidative activity of 1'-acetoxychavicol acetate and its related compounds in the rhizomes of Alpinia galanga during cooking, Food flavors and chemistry, Proceedings of the 10th International Flavor Conference, Paros, Greece, pp. 601-607.
Latha C, Shriram VD, Jahagirdar SS, Dhakephalkar PK, Rojatkar SR. 2009. Antiplasmid activity of 1'-acetoxychavicol acetate from Alpinia galanga against multi-drug resistant bacteria, Journal of Ethnopharmacology, 123(3):522-525.
Lo C-Y, Liu P-L, Lin L-C, Chen Y-T, Hseu Y-C, Wen Z-H, Wang H-M. 2013. Antimelanoma and Antityrosinase from Alpinia galangal Constituents, Hindawi Publishing Corporation, The Scientific World Journal, Volume 2013, Article ID 186505, 5 pages.
Matsuda H, Morikawa T, Managi H, Yoshikawa M. 2003. “Antiallergic Principles from Alpinia galanga and Structural Requirements of Phenylpropanoids for Inhibition of Degranulation and Release of TNF- Alpha And IL-4 In RBL-2H3 Cells”, Bioorganic & Medicinal Chemistry Letters, 13(19):3197-3202.
Matsuda H, Pongpiriyadacha Y, Morikawa T, Ochi M, Yoshikawa M. 2003. Gastroprotective effects of phenylpropanoids from the rhizomes of Alpinia galanga in rats: structural requirements and mode of action, European Journal of Pharmacology, 471(1):59-67.
Mayachiew P, Devahastin S, Mackey BM, Niranjan K. 2010. Effects of Drying Methods and Conditions on Antimicrobial Activity of Edible Chitosan Films Enriched with galangal extract. Food Research International, 43(1):125-32.
Mayachiew P, Devahastin S. 2008. Antimicrobial and antioxidant activities of Indian gooseberry and galangal extracts, Food Science and Technology, 41(7):1153-1159.
Mayachiew P, Devahastin S. 2008. Antimicrobial and antioxidant activities of Indian gooseberry and galangal extracts, Food Science and Technology, 41(7):1153-1159.
Min HJ, Nam JW, Yu ES, Hong JH, Seo EK, Hwang ES. 2009. Effect of naturally occurring hydroxychavicol acetate on the cytokine production in T helper cells. International immunopharmacology, 9(4):448-54.
Misawa T, Aoyama H, Furuyama T, Dodo K, Sagawa M, Miyachi H, Kizaki M, Hashimoto Y. 2008. Structural development of benzhydrol-type 1-acetoxy- chavicol acetate (ACA) analogs as human leukaemia cell growth inhibitors based on quantitative SAR (QSAR) analysis. Chemical and pharmaceutical bulletin, 56(10): pp.1490-1495.
Mohd MS, Chin CB, Chen LL, Sim NL. 2003. Antifungal Activity of The Essectial Oils of Nine Zingiberaceae Spices, Pharmaceutical Biology, 41(5), 302-307.
Morikawa T, Ando S, Matsuda H, Kataoka S, Muraoka O, Yoshikawa M. 2005. Inhibitors of NO production from the rhizomes of Alpinia galanga: structures of new 8-9’ linked neolignans and sesquineolignan. Chemical and pharmaceutical bulletin, 53(6): pp.625-630.
Morita H, Itokawa H. 1988. Cytotoxic and antifungal diterpenes from the seeds of Alpinia galanga, Planta Medica, 54(2):117-20.
Morita H, Itokawa H. 1988. Cytotoxic and antifungal diterpenes from the seeds of Alpinia galanga, Planta Medica, 54(2):117-20.
Mundugaru R, Sivanesan S, Udaykumar P, Prabhu SN, Ravishankar B. 2018. Neuroprotective functions of Alpinia galanga in forebrain ischemia-induced neuronal damage and oxidative insults in rat Hippocampus. Indian journal of pharmaceutical education & research, 52(4):S77-85.
Muniandy P, Murugan P, Jeng-Yeou CN, Singh D, Daruliza K. 2019. A study of antibacterial efficacy of Alpinia galangal extracts against Staphylococcus aureus, Staphylococcus epidermidis and Listeria monocytogenes, Journal of Pharmaceutical Sciences and Research, 11(8):3061-3066.
Murakami H, Tamura S. 2005. Anti-HIV agents and HIV-derived protein nuclear export inhibitors containing 1'-acetoxychavicol acetate, and foods containing the compound, Japan Kokai Tokkyo Koho, pp. 8.
Murakami H, Tamura S. 2005. Anti-HIV agents and HIV-derived protein nuclear export inhibitors containing 1'-acetoxychavicol acetate, and foods containing the compound, Japan Kokai Tokkyo Koho, pp. 8.
Nagashekhar M, Shivaprasad HN. 2006. Antiinflammatory and analgesic activity of the topical preparation of Alpinia galanga willd, Biomed, 1(1):63-68.
Natta L, Orapin K, Krittika N, Pantip B. 2009. Essential oil from five Zingiberaceae for anti food-borne bacteria, International Food Research Journal, 16(3):337-346.
Nik Hasan MK, Kamarazaman IS, Azman M, Abd Rashid L. 2020. Preparation of Alpinia galanga Water Extract with High Antioxidant Properties, Asian Journal of Pharmacognosy, Asian Journal of Pharmacognosy, 4(1):43-48.
Norajit K, Laohakunjit N, Kerdchoechuen O. 2007. Antibacterial Effect of Five Zingiberaceae Essential Oils, Molecules, 12:2047-2060.
Oonmetta-aree J, Suzuki T, Gasaluck P, Eumkeb G. 2006. Antimicrobial properties and action of galangal (Alpinia galanga Linn.) on Staphylococcus aureus, Food Science and Technology, 39(10):1214-1220.
Padma SV, Tiwari V, Singh WL, Ningombam S. 2006. Antioxidant Properties of Some Exclusive Species of Zingiberaceae Family of Manipur, Electronic Journal of Environmental Agriculture & Food Chemistry, 5(2):1318-1322.
Pal Jain A, Singh Pawara R, Lodhia S, Singhaia A. 2012. Immunomodulatory and antioxidant potential of Alpinia galanga Linn. rhizomes. Pharmacognosy Communications, 2(3):30-7.
Pothacharoen P, Choocheep K, Pitak T, Pompimon W, Premanode B, Hardingham TE, Kongtawelert P. 2006. Effect of Alpinia galanga extract on cartilage degradation and on gene expression in human chondrocyte and synovial fibroblast metabolism, Central European Journal of Biology, 1(3):430-450.
Qureshi S, Shah AH, Ahmed MM, Rafatullah S, Bibi F, Al-Bekairi AM. 1994. Effect of Alpinia galanga treatment on cytological and biochemical changes induced by cyclophosphamide in mice, International Journal of Pharmacognosy, 32(2):171-177.
Raj RK. 1975. Screening of indigenous plants for anthelmintic action against human Ascaris lumbricoides: Part-II, Indian journal of physiology and pharmacology, 19(1):6-12.
Rama Rao M, Bhaskar Reddy I, Raja Gopal SV, Bhaskar D, Ramana T. 2008. A comparative study of antimicrobial activity of Curcuma amada and Alpinia galanga of zingiberaceae family, Asian Journal of Chemistry, 20(7):5293-5300.
Ramesh KV, Garima S, Pradeep S, Jha KK, Khose RL. 2011. Alpinia galanga an important medicinal plant: a review, Der Pharmacia Sinica, 2(1):142-54.
Rao K, Bhuvaneswari N, Lakshmi M, Giri A. 2010. Antibacterial activity of Alpinia galanga (L) wild crude extracts, Applied Biochemistry and Biotechnology, 162(3):871-884.
Raviraja Shetty G, Monisha S. 2015. Pharmacology of an Endangered Medicinal Plant Alpinia galanga – A Review, Research Journal of Pharmaceutical, Biological and Chemical Sciences. Research Journal of Pharmaceutical, Biological and Chemical Sciences, 6(1):499-511.
Satish R, Dhananjayan R. 2003. Evaluation of the anti-inflammatory potential of rhizome of Alpinia galanga Linn, Biomedicine, 23(1/2):91-96.
Sawangjaroen N, Phongpaichit S, Subhadhirasakul S, Visutthi M, Srisuwan N, Thammapalerd N. 2006. The anti-amoebic activity of some medicinal plants used by AIDS patients in southern Thailand, Parasitology research, 98(6):588-92.
Sawangjaroen N, Subhadhirasakul S, Phongpaichit S, Siripanth C, Jamjaroen K, Sawangjaroen K. 2005. The in vitro anti-giardial activity of extracts from plants that are used for self-medication by AIDS patients in southern Thailand, Parasitology research, 95(1):17-21.
Shivgunde PP, Shah P, Jain DP, Singhai AK, Somani RS. 2008. Antinociceptive activity of ethanolic extract of rhizomes of Alpinia galanga, Indian Journal Pharmacology, 40(2):172.
Singha Hanish JC, Alagarsamyb V, Prakash V. Diwana, S. Sathesh Kumar, Nishad JC, Narsimha Reddye Y. 2011. Neuroprotective effect of Alpinia galanga (L.) fractions on Aβ(25–35) induced amnesia in mice, Journal of Ethnopharmacology, 138:85-91.
Solikhah TI, Setiawan B, Ismukada DR. 2020. Antidiabetic Activity of Papaya Leaf Extract (Carica papaya L.) Isolated with Maceration Method in Alloxan-induces Diabetic Mice. Systematic Reviews in Pharmacy, 11(9): 774-778.
Sookkongwaree K, Geitmann M, Roengsumran S, Petsom A, Danielson UH. 2006. Inhibition of viral proteases by Zingiberaceae extracts and flavones isolated from Kaempferia parviflora, Die Pharmazie, 61(8):717-21.
Srivastava P, Shanker K. 2012. Pluchea lanceolata (rasayana): Chemical and biological potential of rasayana herb used in traditional system of medicine. Fitoterapia, 83(8):1371-85.
Srividya AR, Dhanabal SP, kumar S, kumar P. 2010. Antioxidant and Antidiabetic Activity of Alpinia galanga, International Journal of Pharmacognosy and Phytochemical Research, 3(1):6-12.
Subash KR, Bhanu Prakash G, Vijaya Chandra Reddy K, Manjunath K, Rao Umamaheswara K. 2016. Anti-inflammatory activity of ethanolic extract of Alpinia galanga in carrageenan-induced pleurisy rats, National Journal of Physiology, Pharmacy and Pharmacology, 6(5):468-470.
Subash KR, Britto Francis G, Kumar Katari S, Umamaheshwari A, Konda V, Reddy C, Bhanu PG. 2018. Analgesic activity of Alpinia galanga extract in mice models and TNF-alpha receptor computational docking analysis on its leads with pharmacokinetics prediction, International Journal of Basic & Clinical Pharmacology, 7(3):446-450.
Subash KR, Rao Jagan N, Varghese CB, Muthulakshmi Bhaarati G, Kumar SK. 2012. the anthelmintic activity of Eupatorium triplinerve and Alpinia galanga in Pheritima posthuman and ascardia galli, A Comparitive study, Journal of Clinical and Diagnostic Research, 6(6):947-950.
Subash KR, Rao NJ,Varghese C, Bhaarati GM, Kumar S. 2012. The anthelmintic activity of Eupatorium triplinerve and Alpinia galanga in Pheritima posthuman and ascardia galli, A Comparitive study, Journal of Clinical and Diagnostic Research, 6(6):947-950.
Sunilson JAJ, Suraj R, Rejitha G, Anandarajagopal K, Kumari AV, Promwichit P. 2009. In vitro antimicrobial evaluation of Zingiber officinale, Curcuma longa and Alpinia galanga extracts as natural food preservatives, American Journal of Food Technology, 4(5):192-200.
Tachakittirungrod S, Chowwanapoonpohn S. 2007. Comparison of Antioxidant and Antimicrobial Activities of Essential Oils from Hyptis suaveolens and Alpinia galanga Growing in Northern Thailand, Journal of Natural Science, 6(1):31-42.
Taechowisan T, Lumyong S. 2003. Activity of endophytic actinomycetes from root of zingiber officinale and Alpinia galanga against phytopathogenic fungi, Annals of Microbiology, 53(3):291-291.
Trakranrungsie N, Chatchawanchonteera A, Khunkitti W. 2008. Ethnoveterinary study for the anti-Dermatophytic activity of Piper betle, Alpinia galanga, and Allium ascalonicum extracts in vitro, Research in Veterinary Science, 84(1):80-84.
Udjiana S. 2008. Food preservation efforts using galangal extract. Journal of Separation Technology, 1(2).
Weerakkody Nimsha S, Caffin N, Turner Mark S, Dykes Gary A. 2010. In vitro antimicrobial activity of less-utilized spice and herb extracts against selected food-borne bacteria, Food Control, 21(10):1408-1414.
Wong LF, Lim YY, Omar M. 2009. Antioxidant and antimicrobial activities of some Alpinia species, Journal of Food Biochemistry, 33(6):835-851.
Wong LF, Lim YY, Omar M. 2009. Antioxidant and antimicrobial activities of some Alpinia species, Journal of Food Biochemistry, 33(6):835-851.
Yang X, Eilerman RG. 1999. Pungent Principal of Alpinia galanga (L.) Swartz and its Applications. Journal of Agricultural and Food Chemistry, 47(4):1657-62.
Ye Y, Li B. 2006. 1'S-1'-acetoxychavicol acetate isolated from Alpinia galanga inhibits human immunodeficiency virus type 1 replication by blocking Rev transport, Journal of General Virology, 87(7):2047-2053.
Yoshikawa M, Matsuda H, Morikawa T. 2004. Antiallergy agent obtained from Alpinia galanga and method for production thereof, Kokai Tokkyo Koho, pp. 9.
Yu ES, Min HJ, Lee K, Lee MS, Nam JW, Seo EK, Hong JH, Hwang ES. 2009. Anti-inflammatory activity of p-coumaryl alcohol-γ-O-methyl ether is mediated through modulation of interferon-γ production in Th cells, British Journal of Pharmacology, 156(7):1107-1114.
Yu ES, Min HJ, Lee K, Lee MS, Nam JW, Seo EK, Hong JH, Hwang ES. 2009. Anti-inflammatory activity of p-coumaryl alcohol-γ-O-methyl ether is mediated through modulation of interferon-γ production in Th cells, British Journal of Pharmacology, 156(7):1107-1114.
Yu JG. 1981. Identification of the chemical components of two Alpine species, Zhongyao tong bao Beijing, China, 13(6):34-6.
Zaeoung S, Plubrukarn A, Keawpradub N, Zaeoung S, Plubrukarn A, Keawpradub N. 2005. Cytotoxic and free radical scavenging activities of Zingiberaceous rhizomes, Journal of Science & Technology, 27(4):799-812.
Zaeoung S, Plubrukarn A, Keawpradub N. 2005. Cytotoxic and free radical scavenging activities of Zingiberaceous rhizomes, Journal of Science & Technology, 27(4):799-812.
Zheng GQ, Kenney PM, Lam Luke KT. 1993. Potential anticarcinogenic natural products isolated from lemongrass oil and galanga root oil, Journal of Agricultural and Food Chemistry, 41(2):153-6.