

Karina Milagros R. Cui-Lim1,2,3, Christina A. Binag3
1College of Science, University of Eastern Philippines
2Research Office, University of Eastern Philippines
3 Research Center for Natural Sciences, University of Santo Tomas, Manila 1008, Philippines
Address for Corresponding Author
Dr. Karina Milagros R. Cui-Lim
College of Science, University of Eastern Philippines
Catarman Northern Samar, 6400, Philippines
Abstract
Objective: Nanowires of polythiophene (PTp) were electrochemically synthesized using the pores of the prepared Anodic Alumina Oxide (AAO) templates. The anodization experiments were performed using Al foil to create alumina pores having pore diameters of 150-250 nm. Materials and methods: The electrochemical polymerization synthesis of PTp nanowires was carried out using a potentiostatic condition in a three-electrode system. The best electropolymerization conditions were 0.1 M thiophene (Tp) monomer, 0.1 M tetrabutylammonium perchlorate (TBAClO4) dopant, in 10.00 mL acetonitrile, at a temperature of 0 + 1 0C, without stirring the polymerization solution using 1.6 V at 2 mA/cm2 current density. Four-point probe conductivity tests were done to determine the electrical properties of the PTp nanowires formed. PTp nanowires revealed a higher conductivity of 11.30 S/cm (0.50% RSD, n=3) compared to the bulk polymerized Al foil (1.45 S/cm). Results: The Scanning Electron image of PTp nanowires showed uniform cylindrical nodules with diameter of ~150 nm. In vitro testing unveiled a novel application of the fabricated PTp nanowires for its antimicrobial activity (100% reduction of colony growth) against S. aureus and E. coli. The antimicrobial property of the nanowire was further confirmed using V. fischeri (bioluminescent bacteria). Conclusion: In this study, the facile AAO template synthesis method utilized for PTp nanowires growth gave higher conductivity that could find wider environmental and medical applications.
Keywords: nanowires, polythiophene, template synthesis, anodic alumina oxide, SEM, antimicrobial property
Introduction
Conducting polymers are emerging as promising resources for the synthesis of nanostructured materials (Joo et. al., 2004). They are particularly appealing because they exhibit electrical, electronic, magnetic, and optical properties similar to metals or semiconductors while retaining their flexibility, ease of processing and modifiable electrical conductivity (Maynor, 2001). Polythiophene is very stable in air and water however its conductivity is lower compared to other known polymers such as polyaniline (PAn), polypyrrole (PPy) and polyacetylene (PAc). These properties of PTp are utilized in the development of nanowires (Park et. al., 2010).
The scientific and technological aspects of nanostructured materials are currently attracting considerable attention. Several methods have been proposed and applied to the synthesis of nanostructures such as molecular beam epitaxy, microlithography and template-assisted methods. A method which entails synthesizing the desired material within the pores of a membrane is called template synthesis. Template synthesis is an elegant chemical or electrochemical approach for the fabrication of nanostructures, particularly in view of its flexibility to produce nanostuctures with a different composition. The prepared wires, tubules and fibrils can be composed of CPs, semiconductors, carbon, metals and other materials (Kyotani, 1996).
Research has currently been focused on the fabrication, characteristics, and applications of nanoscale polymers. The AAO template method has been used for the fabrication of nanowires, as it has many advantages like having relatively high conductivity, flexible, low cost, and controlled chemistry (Yanchun et. al., 2005).
In this paper, we report the synthesis, characteristics and application of PTp nanowires using the prepared porous alumina template. The different electrochemical polymerization conditions were optimized in this study to identify the best parameters for the control of conductivity and morphology of PTp nanowires.
Materials and methods
Materials: All the reagents were prepared using analytical grade reagents and distilled water. The Tp monomer was purified using simple distillation. Electrochemical experiments were performed with a potentiostatic condition in a one-compartment cell at room temperature. A platinum and a silver-silver chloride served as the auxiliary and reference electrode, respectively. Anodic alumina membranes were employed as the template for nanowire growth.
Fabrication of AAO template: A 99.99 % 0.5 mm thick Al foil (Sigma) was degreased in acetone followed by ultrasonic cleaning (Kerry). Anodization was done in 0.3 M oxalic acid (Ajax) for 12 hours with the Al foil anode and C-rod cathode at constant current with vigorous stirring (Maynor, 2001). Different anodization conditions like varying the voltage (10 V, 5 V and 2 V) and temperature (0 0C, 15 0C, and 25 0C) were utilized to prepare the AAO template for growth of nanowires.
Electropolymerization of Tp Monomer: The PTp nanowires were synthesized through electrochemical polymerization using the prepared AAO template in various parameters. The electrolyte consisted of acetonitrile (Sigma) solvent, TBAClO4 dopant (Sigma), and Tp monomer (Sigma) at 1.6 V without stirring the solution. Different optimization conditions for the growth of PTp nanowires were tested, like polymerization time, type of dopants, type of solvents, current density, and temperature. The synthesis of PTp nanowires was carried out in a one-compartment cell with the anodized Al foil (anodic alumina oxide, AAO) as working electrode (WE), Pt wire as auxiliary electrode (AE), and Ag/AgCl reference electrode (RE). After the electrochemical polymerization, the coated-AAO was soaked with 0.4 M NaOH (Merck) for 60 minutes to dissolve the template.
Characterization of the PTp Nanowires: The morphologies of the porous alumina template and PTp nanowires grown on them were investigated with Scanning Electron Microscopy (SEM) (JEOL-JSM-5310) at the Physics Laboratory of De La Salle University, Manila. To determine the electrical properties of the nanowires, the conductivity was measured using the Four-point probe technique to eliminate or minimize contact resistance.
Application: Each of the pristine Al foil, bulk polymerized Al foil and PTp nanowires inoculated with the test microorganism was placed on each tube with saline solution. The test tubes were swirled for 15 minutes to disperse the cells of the microorganisms. One mL aliquot of the prepared saline solution where the pristine Al foil and PTp nanowires were inoculated with microorganism was placed on each Petri dish. These plates where then poured with the media. The medium was allowed to solidify and the plates were incubated for 24 hours. The number of colonies of the viable cells that grew on the surface of the medium in the plates labeled as pristine Al foil and PTp nanowires were counted and compared.
Results and discussion
Anodization of Al foil to produce an oxide film is carried out using electrochemical process in a 2- electrode set-up under galvanostatic condition. Porous alumina oxide grows in acid electrolytes like oxalic acid in which oxide can be deposited in the Al foil but also dissolves using template dissolving solvent.
Different anodization conditions like temperature and applied voltage could be used in order to produce a well-ordered and well-aligned array of nanopores. Figure 1 shows the SEM micrographs of the pristine Al foil and AAO template. The pristine Al foil showed the smooth and uniform image (Figure 2(a)), while the prepared AAO template showed the uniform formation of pores on a rough alumina oxide coating (Figure 2(b)). This anodization process used the best conditions of 0.3 M oxalic acid, 0 0C, at 10 V. Nanopores of diameters ~ 150 nm were observed and results showed that excessive pretreatments were not necessary to obtain well-ordered nanopores and only simple pretreatments like degreasing and oxide removing were enough.
 Figure 1. SEM micrograph images taken at 3500x magnification (a) pristine Al foil and (b) AAO template
Figure 1. SEM micrograph images taken at 3500x magnification (a) pristine Al foil and (b) AAO template
The described template procedure enables the fabrication of PTp nanowires with the attractive possibility to vary the diameter and appearance of nanowires by different optimization conditions. The PTp nanowires were synthesized using electrochemical polymerization on AAO template in a three-electrode system under potentiostatic condition using the optimized parameters presented on Table 1. In PTp nanowires growth, the potential is better controlled to prevent the PTp from being overoxidized thus, becoming more conductive and in this study the potential was kept constant at 1.6 V (Popat et. al., 2005). The polymerization of Tp monomer to produce PTp nanowires in the presence of a suitable dopant and solvent led to the formation of PTp radical cations that can couple with other radical cations and ultimately build up a conjugated polymer chain (Yanchun, 2005). A good organic solvent is used in which the Tp monomer is soluble but the PTp is not soluble. As the chain of Tp monomer length increases, they adhere to the AAO template and grow within the pores of AAO to produce PTp nanowires.
Table 1. Summary of the best electrochemical polymerization parameters for the growth of PTp nanowires
|
Electropolymerization parameters |
Best condition |
|
Polymerization time |
90 minutes |
|
Tp monomer concentration |
0.1 M |
|
Current density |
2 mA/cm2 |
|
Dopant concentration |
0.1 M TBAClO4 |
|
Type of solvent |
10 mL acetonitrile |
|
Polymerization temperature |
0 0C+1 0C |
|
Applied voltage |
1.6 V |
The formation mechanism of PTp nanowire structures could be explain through the process in which the AAO template is prepared by anodic oxidation in a 0.3 M oxalic acid solution, leaving some anionic sites on the pore walls of the AAO template (Park et. al., 2010). These anionic sites on the pore walls of the template and the positively charged Tp monomer can be adsorbed and nucleated preferentially on the pore walls according to the electrostatic interaction (Schonenberger et. al., 2009). On the other hand, these anionic sites can also be viewed as molecular anchor which guide the Tp monomer molecules in depositing on the pore walls of the AAO template. According to this mechanism, the PTp nanowires with different length and diameter can be obtained if the various electrochemical polymerization parameters were changed.
Various experimental parameters that control the diameter and morphology of the PTp nanowires grown have been investigated as presented in Table 1. After the dissolution of the alumina template using 0.4 M NaOH solution for 60 minutes, the wires are freed from the template that can be further investigated. The morphology of PTp nanowires was studied by SEM and was correlated with its electrical properties. Figure 2 shows the SEM micrograph of the formation of PTp nanowires having diameter of ~150 nm.
The electrical conductivity of fabricated PTp nanowires is greatly influenced by the method and conditions used to obtain the nanowires. The measured conductivity of PTp nanowires revealed highest conductivity of 11.30 S/cm at 3 trials with 0.50% RSD as compared to the pristine Al foil and bulk polymerized Al foil which has conductivity values of 2.10x10-3 S/cm and 1.45 S/cm, respectively. The SEM micrograph images could explain the increasing conductivity when the diameter of the PTp nanowires decreases. The conductivity of the polymer nanowires has been studied as a function of the diameter. The confinement of the nanowires in the pore walls of the AAO template, it is assumed that the polymer chains on the surface are aligned giving rise to a large conductivity.

Figure 2. SEM micrograph image of PTp nanowires synthesized using the best electrochemical polymerization parameters at 3500x magnification
For possible application of the fabricated PTp nanowires, in vitro testing of its antimicrobial properties were performed in this study. On the other hand, quantitative determination of the antimicrobial activities of PTp nanowire was examined using S. aureus and E. coli. Three replicates of PTp nanowires, bulk polymerized Al foil and pristine Al foil (served as negative control) were tested after 24 hour incubation period. The quantitative determination of antimicrobial activity of S. aureus and E. coli were investigated in this study. It was observed from the plates after 24 hour incubation that S. aureus did not grow over the PTp nanowires, since there were no colonies that were formed. On the other hand, S. aureus grew over the bulk polymerized Al foil and it showed 40% reduction of colony growth. While the bacteria S. aureus grew readily over the pristine Al foil as presented in Figure 3. The S. aureus streaks on the pristine Al were of the same strengths as those in PTp nanowires. The 100% reduction in colony growth, imply that PTp nanowire exhibited bactericidal property.
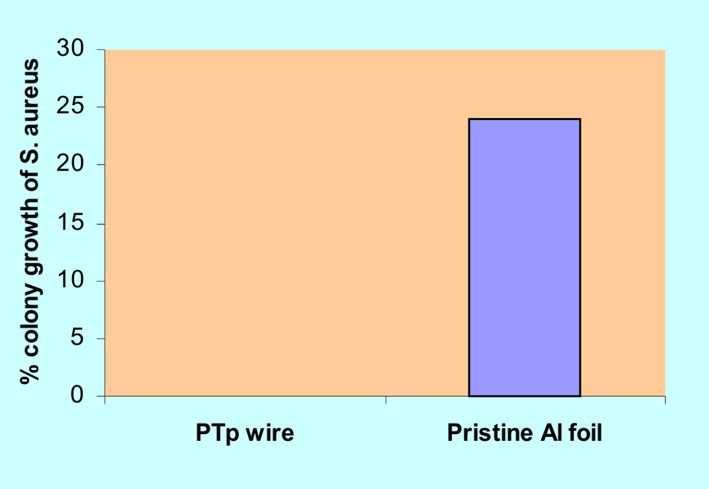 Figure 3. Percent inhibition growth of S. aureus colonies
Figure 3. Percent inhibition growth of S. aureus colonies
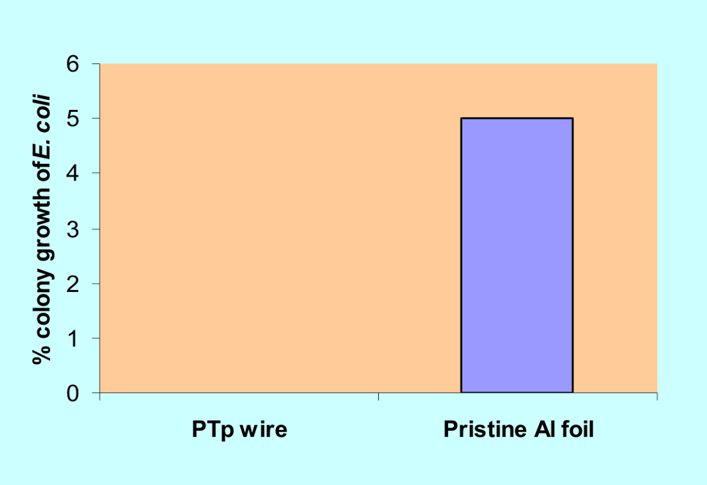
Figure 4. Percent inhibition growth of E. coli colonies
E. coli a Gram-negative bacterium was used in this study. The 3 replicates of pristine Al foil and PTp nanowires were dispersed in the media. Figure 4, demonstrates colony growth of E. coli on the pristine Al after 24 hours of incubation. On the PTp nanowires, no E. coli growth was observed after the 24 hour incubation period. The 100% decrease in colony growth implies that the PTp nanowire exhibited bactericidal property. The results showed that the fabricated PTp nanowires exhibited cidal properties against S. aureus and E. coli bacteria during in vitro testing.
Conclusion
This study presents an easy way of preparing porous AAO template used to fabricate PTp nanowires. The diameter of PTp nanowires are closed to the pore diameter of the fabricated AAO template (~150 nm). The mechanism of PTp nanowires growth is due to the electrostatic interaction of Tp monomer molecules and the pore walls of the AAO template, which led to the adsorption, nucleation and polymerization of Tp monomer, preferentially on the pore walls of the AAO template. Polythiophene nanowires supported on porous alumina template gave high conductivity of 11.30 Scm-1. The morphologies correlated well with the electrical property of the PTp nanowires and a higher conductivity in the smaller nanowires diameter is observed. This is due to the presence of a highly ordered layer of PTp along the pore wall of the template. In this case, it is assumed that the polymer chains increased and are aligned, giving rise to a higher conductivity of the fabricated PTp nanowires. The results presented in this study bring new dimensions that could contribute to a better characterization of PTp nanowires. Filling the pores of the AAO template, PTp nanowires are obtained reproducibly and economically. The method used in this study offers the possibility to fabricate PTp nanowires with an engineered diameter and length. This point is very interesting for the development of different applications based on PTp nanowires as the process is not limited to the production of polymeric nanowires but could also be used for the preparation of other nanomaterials. The results presented in this study bring new dimensions that could contribute to a better characterization of PTp nanowires. Filling the pores of the AAO template, PTp nanowires are obtained reproducibly and economically. The method used in this study offers the possibility to fabricate PTp nanowire with an engineered pore diameter and length. This point is very interesting for the development of different applications based on PTp nanowires as the process is not limited to the production of polymeric nanowires but could also be used for the preparation of other nanomaterials. With its cidal properties, the fabricated PTp nanowires could be apply as an effective biocidal surface for its possible application in medical devices.
Acknowledgement
One of the authors (K.M.C.L.), would like to thank the Research Office, University of Eastern Philippines, Catarman N. Samar for the support.
References
Joo J, Park K, Kim B, Lee S. 2003. Conducting Polymer Nanotube and Nanowire Synthesized by Using Nanoporous Template: Synthesis, Characteristics, and Applications. Journal of American Chemical Society, 9(7): 135-136.
Kim BH, Park DH, Gu YK, Joo J. 2004. Electrical, Magnetic, and Optical Properties of p-Conjugated Polymer Nanotubes and Nanowires. Journal of Nonlinear Optical Physics and Materials, 13 (3): 547-551.
Mativetsky JM, Datars W. 2002. Morphology and electrical properties of template-synthesized polypyrrole nanocylinders. Physica, 324: 191-204.
Maynor B. 2001. Direct Writing of Polymer Nanostructures: Polythiophene Nanowires on Semiconducting and Insulating Surfaces. Journal of American Chemical Society, 124 (4): 522-523.
Park DH, Kim BH, Jang MK, Bae KY, Lee SJ, Joo J. 2010. Synthesis and Characterization of Polythiophene and Poly (3-methylthiophene) Nanotubes and Nanowires. Synthetic Metals, 153: 341-344.
Popat K, Gopa C, Craig C, Tejal R. 2004. Polyethylene glycol grafted Nanoporous Alumina Membrane. Journal of Membrane Science, 243: 97-106.
Schonenberger C, Avan der Zande B, Henny M, Schmid C. 2009. Template Synthesis of Nanowires in Porous Polycarbonate Membranes; Electrochemistry and Morphology. Journal of American Chemical Society, 101(18): 5497-5505.
Yanchun Z, Chen M, Zhang Y, Xu T, Liu W. 2005. A facile approach to formation of through–hole porous anodic aluminum oxide film. Materials Letters, 59: 40-43.