

R. Padmavathi1*, Akula Annapurna2
1G. Pulla Reddy College of Pharmacy, Hyderabad, India
2AU College of Pharmaceutical Sciences, Andhra University, Vishakhapatnam, India
*Address for Corresponding Author
Dr. R. Padmavathi
Associate Professor,
Department of Pharmacology, G.Pulla Reddy College of Pharmacy, Hyderabad-500028 Telangana – India
Abstract
Objective: The present study was carried out to evaluate cerebro-protective potential of Urtica dioica against ischemia-reperfusion induced cerebral damage and neurobehavioral alterations in rats. Material and methods: In order to evaluate the cerebroprotective activity, we have chosen in vivo bilateral common carotid artery occlusion induced cerebral ischemia-reperfusion injury model (30 min ischemia and 24 h reperfusion) and Urtica dioica (0.2, 0.4, 0.8 ml/kg) treatment was given for seven days prior to induction of ischemia and reperfusion. Cerebral damage was determined using TTC staining technique. We employed Elevated plus maze test to assess memory and Locomotor activity, Inclined beam walking test (neurological score) and Hanging wire test for evaluation of motor co-ordination. Results: Severe cerebral damage was observed in rats subjected to ischemia-reperfusion. Urtica dioica treatment dose dependently offered cerebro-protection by significantly reducing the infarct size. Marked impairment of memory and motor co-ordination in parallel to the significant increase in the infarction size was observed in rats subjected to ischemia-reperfusion injury. Pretreatment with Urtica dioica significantly attenuated ischemia-reperfusion induced neurobehavioral alterations in the rats. Conclusion: This work suggests that Urtica dioica may improve the therapeutic outcome of stroke, if administered in conjunction with thrombolytic therapy
Keywords: Urtica dioica, cerebro-protective, ischemia-reperfusion, neurobehavioral
Introduction
Stroke and cardiac arrest are major causes of mortality and disability worldwide, affecting millions of people and accounting for the highest health-care expenses of all diseases. Stroke is a cerebrovascular disorder characterized by the sudden disruption of blood supply to the brain, which, if not resolved quickly, results in severely ischemic brain tissue, eventually leading to cell death (Beal, 2010). According to the World Health Organization’s most recent statistics, around 15 million individuals worldwide suffer from a stroke. One-third of these people died (3 million women and 2.5 million men) and one-third are permanently disabled (Thrift et al., 2017).
Part of the ischemic tissue in which the cells are irreversibly damaged is called “infarction”. The severity of stroke outcome depends on the extent of infarct size (Lipton, 1999). Reperfusion therapy, such as thrombolytics or bypass surgery, can help to reduce the pathological consequences of a stroke. However, several animal and clinical investigations have demonstrated that overproduction of reactive oxygen species and excessive inflammatory response occurs in the early reperfusion period, which potentiates the ischemic damage (Leira et al., 2006; Pan et al., 2007). As a result, oxidative stress and inflammation are thought to be important contributors to cerebral ischemia-reperfusion injury. The inhibition of reactive oxygen species formation, inflammatory cell activation, pro-inflammatory cytokine release and apoptotic gene expression has shown to protect against cerebral ischemia-reperfusion injury (Lin et al., 2016).
Ischemia-reperfusion induced brain damage is linked to a wide range of neuro-psychiatric problems, including motor, behavioural and cognitive disorders (Jenkins et al., 1981). Memory deficits and sensorimotor abnormalities have been observed in global cerebral ischemia subjects in preclinical and clinical studies (Yan et al., 2007; Gaur et al., 2009). At a four-year follow-up, around one-third of stroke survivors screened during hospitalization were found to fulfil dementia criteria and about 80% of patients with acute stroke present with focal weakness or paralysis (Chemerinski and Robinson, 2000).
In diverse experimental settings, a number of traditional medicines with potent antioxidant and anti-inflammatory activities have been shown to reduce brain damage caused by ischemia-reperfusion. Plants, such as Nigella sativa, Ginkgo biloba, garlic, Tanshinone, Panax ginseng, Bacopa monnieri, Withania somnifera, Centella asiatica, Curcumin, and Ocimum sanctum been shown to protect against ischemia-reperfusion induced brain damage in various experimental stroke models (Wu et al., 2010).
Urtica dioica (UD) is a well-known ethnomedicinal plant, is also known as common stinging nettle. It has a long history of use in Indian traditional folk medicine (Mamta and Preeti, 2014). In several experimental animal models and clinical studies, it has been shown to have potent antioxidant and anti-inflammatory properties (Upton, 2013). However, no research on neuroprotective effect of Urtica dioica on ischemia-reperfusion brain injury has ever been done. Thus, it was considered worthwhile to investigate the potential role of Urtica dioica on ischemia-reperfusion induced cerebral injury in rats. Therefore, in the present investigation we have planned to explore cerebro-protective role of Urtica dioica against ischemia-reperfusion induced cerebral damage and, neurobehavioral alterations.
Materials and methods
Plant extract
Ethanolic extract of UD in the form of homeopathic mother tincture was purchased from a local medical store (manufactured by ‘Bhandaris Pvt. Ltd’, Batch no. M2/07, manufacturing date 11/2012, with 48% ethanol as vehicle .drug strength 1/10).
UD is prescribed as a mother tincture in homeopathy. The whole plant is used to make a homoeopathic. A mother tincture is essentially an alcoholic extract prepared according to the procedure described in Homoeopathic Pharmacopoeia (Sinha and Saha, 1980). Both the US Homoeopathic Pharmacopoeia and the German Homoeopathic Pharmacopoeia cover it. A mother tincture, despite being a homoeopathic formulation, is equally effective as an alcoholic extract often used by researchers in phytochemical or biological studies (Kumar and Sharma, 2005; Nimgulkar et al., 2011; Khuda-Bukhsh et al., 2011; Ghosh et al., 2013).
Dose Selection
In this study, 3 doses (0.2, 0.4, 0.8 ml/kg) of Urtica dioica were used. The doses were chosen based on previous experimental studies in animals with Urtica dioica and extrapolation from the doses used for clinical use. The use of UD has been reported safe in acute and chronic toxicity studies in mice, at the dose levels of 250, 500, 1000, 2000 mg/kg body weight. Typical daily dosages of UD for clinical use include, 360 mg aqueous extract, 460 mg dried extract, and 600 mg freeze-dried preparation. Tincture of the plant is taken at a dose of 1/2-1 tsp (2-5 mL) three times daily (Mamta and Preeti, 2014).
Animals
Wistar male albino rats (230–300g) were obtained from Srinivasa Enterprises, Hyderabad, Telangana, India. The animals were kept under standard laboratory conditions, maintained on 12 h light/dark cycle, had free access to food and water. Animals were acclimatized to laboratory conditions before the test. Each animal was used once in the experiment. Experimental protocol was approved by the Institutional Animal Ethics Committee and was conducted according to the CPCSEA guidelines for the use and care of experimental animals (Regd. No. 516/PO/C/01/CPCSEA).
Experimental Protocol
The rats were divided into seven groups of 6 rats each and following experimental protocol was used. Group 1:Normal control; Group 2: Sham control, received surgery, without BCCA occlusion; Group 3: I/R control, received 30 min BCCA occlusion and 24 hours reperfusion; Group 4: Vehicle control, received 0.8 ml/kg of 24% ethanol (double dilution of tincture) for 7 days prior to induction of ischemia and reperfusion; Group 5: Urtica dioica (0.2 ml/kg) Group 6: Urtica dioica (0.4 ml/kg) Group 7 : Urtica dioica (0.8 ml/kg). Prior to induction of ischemia and reperfusion, the rats in the treatment groups (Group-5, 6 and 7) received respectively 3 doses of Urtica dioica (0.2, 0.4, 0.8 ml/kg) intraperitonealy daily for seven days.
At the end of experimental procedure, all the rats were assessed for neurobehavioral outcome. Then the rats were sacrificed, % cerebral infarction was measured.
Induction of Cerebral Ischemia-Reperfusion (I/ R) injury in rats
Induction of cerebral ischemia-reperfusion injury was carried as modified method of Jingtao (Jingtao et al., 1999). Animals were anesthetized by giving thiopentone sodium (45 mg/kg, i.p). Both common carotid arteries were exposed over a midline incision, and a dissection was made between the sternocleidomastoid and the sternohyoid muscles parallel to the trachea. Each carotid artery was freed from its adventitial sheath and vagus nerve, which was carefully separated and maintained. A cotton thread was passed below each carotid artery. The induction of ischemia was performed by occluding bilateral common carotid arteries (BCCAO) for 30 min. After 30 min of ischemia arteries were freed from occlusion by removing threads. Then it was confirmed for reflow of blood. The skin was closed with stitches using waxed silk suture. Reperfusion was allowed for 24 hours. During the BCCAO, animals were observed for the following criteria: maintenance of dilated pupils, absence of a corneal reflex when exposed to strong light stimulation, and maintenance of rectal temperature at (37°C). Animals which did not match these criteria and showed seizures were excluded from the study. Sham control animals received surgery, without BCCAO.
Measurement of percentage cerebral infarction
Animals were sacrificed by cervical dislocation and the brain was removed immediately. Then it was washed with ice cold saline. The brain was wrapped in aluminium foil and kept at −4°C. Frozen brain was sliced into uniform sections of 2 mm thickness. The slices were incubated in 1% 2,3,5-Triphenyl tetrazolium chloride (TTC), purchased from Sigma Aldrich, India, dissolved in phosphate-buffered saline (pH 7.4) at 370C for 30 min. TTC is converted to red formazone pigment by nicotinamide adenine dinucleotide (NAD) and dehydrogenase present in living cells. Hence viable cells were stained deep red. The infarcted cells loose these enzymes and, thus remained unstained 110 (Bederson et al., 1986). Pale, necrotic infarcted tissue was separated and weighed. Percentage cerebral infarction was calculated.
Assessment of neurobehavioral parameters
Locomotor activity
The Locomotor activity (ambulatory activity) was recorded by using actophotometer. Before Locomotor task, the animal was placed individually in the activity meter for 3 min for habituation. Thereafter, Locomotor activity was recorded using actophotometer for a period of 5 min. It was recorded and expressed in terms of total photo beam count per 5 min (Reddy and Kulkarni, 1998).
Hanging wire test
This task was used to measure gripping and forelimb strength of the rats after ischemiareperfusion induced brain injury. In this test, animals were suspended by the forelimbs on a wire stretched between 2 posts 60 cm above a foam pillow. The time (in seconds) until the animal fell was recorded. The cut off time was taken as 90 Sec (Hunter et al., 2000).
Inclined beam walking test (Neurological Score)
The inclined beam walking test was used to evaluate fore and hind limb motor coordination. Each animal was individually placed on a wooden bar, inclined at an angle of 60° from the platform. The motor performance of rats was scored on a scale ranging from 0 to 4. A score of 0 was assigned to an animal that could readily traverse the beam. Score 1, 2 and 3 were given to animals demonstrating mild, moderate and severe impairment, respectively. Score 4 was assigned to the animals completely unable to walk on the beam (Feeney et al., 1981).
Elevated plus maze test for memory
Elevated plus maze was used to assess memory dysfunction. It consists of two opposite open arms (50×10 cm), crossed with two closed arms of the same dimensions with 40 cm high walls. The arms are connected with the central square (10×10 cm). Acquisition of memory was assessed on day 6th, day just before the surgery. The rat was placed individually at one end of an open arm facing away from the central square. The time taken by animal to move from open arm and enter into one of the closed arms was recorded as initial transfer latency (ITL). The rat was allowed to explore the maze for 30 s after recording ITL and returned to its home cage (Hock, 2015).
Statistical analysis
All the values are expressed as mean ± S.E.M. Wilcoxon rank sum test was used for the neurological scores. The data of all the other experiments were analyzed using One way analysis of variance (ANOVA) followed by Tukey's post hoc test. In all the tests, criterion for statistical significance was P <0.05.
Results
Effect of Urtica dioica on percentage cerebral infarction
Percentage cerebral infarction in vehicle control rats was 62.95 ± 2.460. It was significantly reduced by 24%, 56%, and 62% respectively with the treatment of 0.2, 0.4, 0.8 ml/kg doses of Urtica dioica for 7 days prior to induction of ischemia and reperfusion, indicating the cerebro-protective action of Urtica dioica (Figure 1, Figure 2 and Table 1).
Table 1. Effect of Urtica dioica on Percentage Cerebral Infarction
|
Groups (n=6) |
% Cerebral Infarction |
% Reduction of Infarction |
|
Normal |
1.690 ± 0.335 |
|
|
Sham control |
3.192 ± 0.375 |
|
|
I/R control |
62.47 ± 2.340### |
|
|
Vehicle control |
62.95 ± 2.460 |
|
|
Urtica dioica (0.2 ml/kg) |
47.88 ± 2.561*** |
24 |
|
Urtica dioica (0.4 ml/kg) |
35.20 ± 1.337*** |
56 |
|
Urtica dioica (0.8 ml/kg) |
23.87 ± 1.335*** |
62 |
I/R (ischemia/reperfusion). Results were represented as mean ± S.E.M. (n = 6). ANOVA and Tukey's post hoc test was used for statistical analysis of data. ### Significantly different from Sham control group at P < 0.001. *** Significantly different from I/R control group at P < 0.001
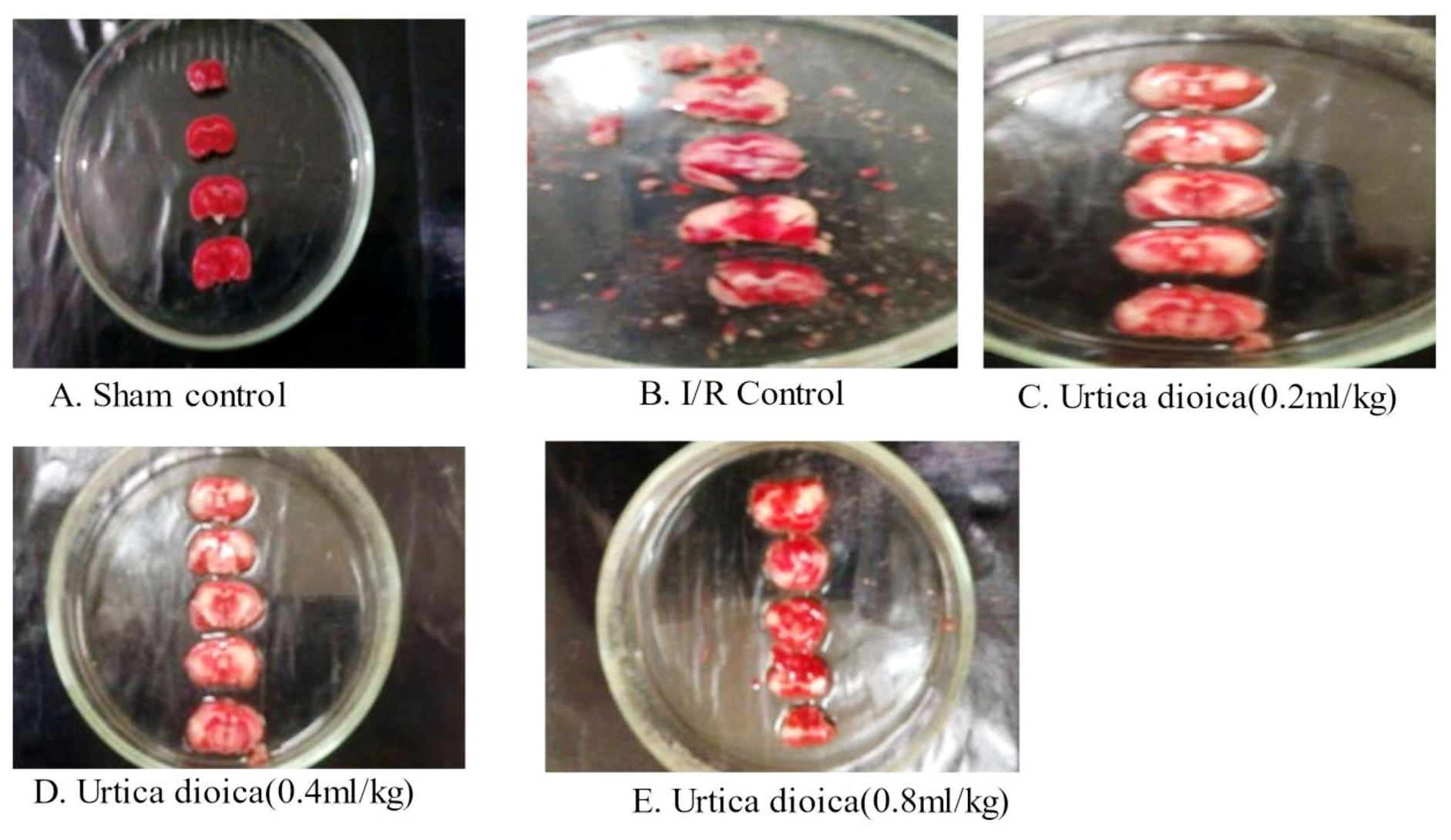
Figure 1. Photograph of brain sections stained with TTC Pale, unstained section indicates infarcted tissue and darkly stained section indicates viable (non-necrotic) tissue
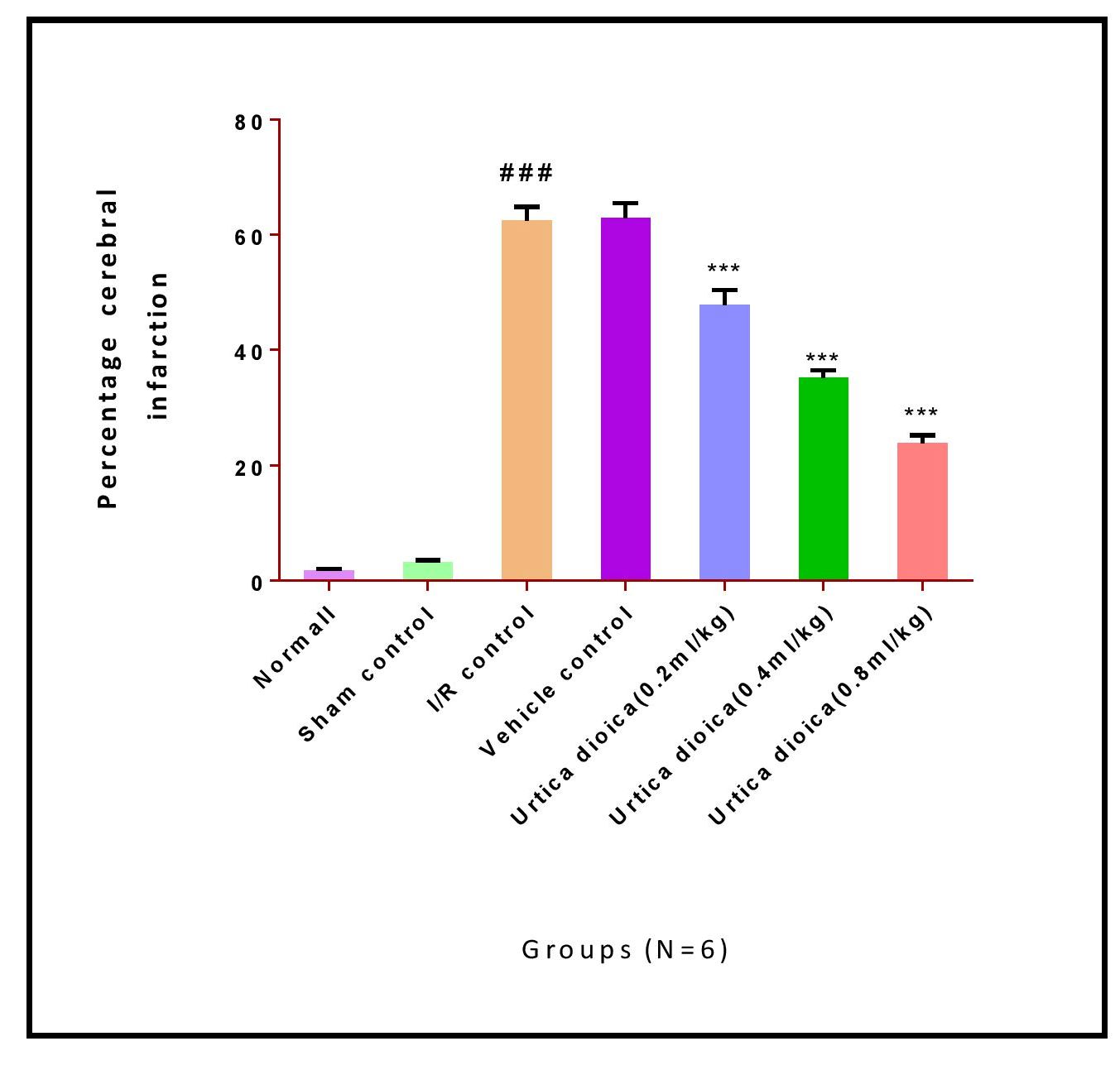
Figure 2. Effect of Urtica dioica on brain infarction volume in rats exposed to cerebral ischemia/reperfusion I/R (ischemia/reperfusion). Results were represented as mean ± S.E.M. (n = 6). ANOVA, and Tukey’s post hoc test was used for statistical analysis of data. ### Significantly different from Sham control group at P < 0.001. ***Significantly different from I/R control group at P < 0.001.
Effect of Urtica dioica on neurological score (Inclined beam walking test)
This test was used to evaluate fore and hind limb motor co-ordination. In the rats subjected to 30 min of BCCA occlusion followed by 24 hours of reperfusion there was significant elevation of neurological score, indicating impairment in motor co-ordination as compared to the sham control rats. Neurological Score was not significantly influenced with Urtica dioica (0.2 142 ml/kg) pretreatment as compared to the vehicle control rats. On the contrary, Urtica dioica (0.4, 0.8 ml/kg) significantly and dose dependently improved neurological score, the motor impairment as compared to the vehicle control rats (Table 2).
Table 2. Effect of Urtica dioica on Neurobehavioral Parameters
|
Groups (n=6) |
Neurological score |
Locomotor Activity (count/5 min) |
Hanging wire Latency time (sec) |
Transfer latency time (sec) |
|
Normal |
0.33 ± 0.211 |
253.8 ± 2.496 |
71.50 ± 2.405 |
36.67 ± 1.308 |
|
Sham control |
0.33 ± 0.211 |
251.5± 3.253 |
69.50 ± 2.579 |
38.33 ± 1.542 |
|
I/R control |
2.83 ± 0.307### |
146.3 ± 2.418### |
24.00 ± 1.949### |
77.00 ± 1.414### |
|
Vehicle control |
2.67 ± 0.211 |
150.5± 2.432 |
23.50± 2.487 |
78.67 ± 1.706 |
|
Urtica dioica (0.2 ml/kg) |
2.33 ± 0.211 |
167.0± 2.708** |
32.33 ± 1.892 |
66.83 ± 1.600** |
|
Urtica dioica (0.4 ml/kg) |
1.67 ± 0.211* |
208.7± 3.029*** |
43.17 ± 1.701*** |
56.83 ± 1.621*** |
|
Urtica dioica (0.8 ml/kg) |
0.83 ± 0.307*** |
235.7 ± 2.963*** |
56.17 ± 2.151*** |
49.83 ± 1.302*** |
I/R (ischemia/reperfusion). Results were represented as mean ± S.E.M. (n = 6). Results were analyzed by one way ANOVA, followed by Tukey’s multiple comparison test. ### Significantly different from Sham control group at P < 0.001. ** Significantly different from I/R control group at p < 0.01. *** Significantly different from I/R control group at P < 0.001. * Significantly different from I/R control group at P < 0.05.
Effect of Urtica dioica on locomotor activity
Challenging the animals with 30 min of BCCA occlusion followed by 24 h of reperfusion significantly impaired locomotor activity as compared to the sham control rats. Pretreatment with Urtica dioica (0.2, 0.4, 0.8 ml/kg) significantly improved the locomotor activity in a dose dependent manner as compared to vehicle control rats (Table 2).
Effect of Urtica dioica on hanging wire latency time
This task was used to measure gripping and forelimb strength of the rats. In the rats subjected to 30 min of BCCA occlusion followed by 24 hours of reperfusion there was significant decrease in hanging wire latency time, indicating impairment in grip strength as compared to the sham control rats. Urtica dioica (0.4, 0.8 ml/kg) pretreatment showed significant and dose dependent increase in the hanging wire latency time as compared to the vehicle control rats. However, Urtica dioica (0.2 ml/kg) did not influence hanging wire latency time significantly as compared to the vehicle control rats (Table 2).
Effect of Urtica dioica on transfer latency time (Elevated plus maze test)
This test was used to assess memory function in rats. Challenging the animals with 30 min of BCCA occlusion followed by 24 h of reperfusion resulted in significant increase in the transfer latency time, indicating memory impairment as compared to the sham control rats. Pretreatment with Urtica dioica (0.2, 0.4, 0.8 ml/kg) significantly attenuated the memory impairment in a dose dependent manner as compared to the vehicle control rats (Table 2).
Discussion
Many animal models have been developed to induce cerebral ischemia-reperfusion injury. A model of incomplete global cerebral ischemia, achieved by BCCA occlusion of rats for 30 min, followed by reperfusion for 24 hours was used in this study. Such induced partial ischemia, without affecting the collateral circulation, reflects the events occurring during transient ischemic attacks and clinical cerebral infarction (Jingtao et al., 1999).
We have measured extent of ischemia-reperfusion induced cerebral damage in terms of percentage cerebral infarction. TTC staining was used to differentiate infarcted tissue from non-necrotic tissue (Benedek et al., 2006).
We found that sections in the sham control rats were uniformly and darkly stained, indicating non-necrotic and viable tissue. Whereas, in vehicle control rats, a part of the tissue was found pale and unstained, indicating necrosis of brain tissue due to ischemia and reperfusion. Therefore, we confirmed that BCCA occlusion model was established successfully.
Infarction volume in the brain is an important determinant in assessing the consequences of cerebral ischemia-reperfusion injury. Therefore, we have measured cerebral damage in terms of percentage cerebral infarction. We found that BCCA occlusion of rats for 30 min, followed by reperfusion for 24 hours (vehicle control group) produced a percentage infarction of 62.95 ± 2.460. Pretreatment with Urtica dioica (0.2, 0.4, 0.8 ml/kg) for 7 days prior to induction of ischemia and reperfusion reduced the percentage cerebral infarction significantly and dose dependently.
Our results are in accordance with earlier studies carried out in the same experimental models (Gaur et al., 2009; Raghavendra et al., 2009). Moreover, our findings are in harmony 137 with previous studies demonstrating that natural herbal medicines could protect the brain from damage caused by ischemia and reperfusion (Hatware et al., 1999; Jiang et al., 2007).
Cerebroprotective action of Urtica dioica observed in our study is strongly supported by previous studies conducted on Urtica dioica. Vafaee et al. (2012) investigated the neuroprotective effect of the immunomodulary drug Setarud, which is composed of herbal extracts including Rosa canina, Urtica dioica and Tanacetum vulgare, supplemented with selenium against focal ischemia-reperfusion injury. Therefore, the results in the present study propose that Urtica dioica has significant cerebroprotective action as shown by a significant decrease in percentage cerebral infarction.
Global cerebral ischemia and reperfusion induced behavioral alterations in experimental animals are similar to clinical symptoms in stroke patients (Dobkin, 1991). Similarly, global cerebral ischemia has been demonstrated to cause a marked decrease in grip and muscle strength of the limbs and memory impairment in several experimental models (Mukherjee et al., 2007; Rehni and Singh, 2007).
In the present study, BCCA occlusion for 30 min followed by 24 hours of reperfusion showed significant impairment of memory, demonstrated by increased transfer latency time 146 (Elevated plus maze test) and a marked decrease in muscle and grip strength (Locomotor activity, Hanging wire test and Inclined beam walking test), which were significantly attenuated by Urtica dioica pretreatment, suggesting the therapeutic potential of Urtica dioica against ischemia-reperfusion injury.
These findings are similar to earlier studies, which also reported motor and memory deficits in the animals following BCCA occlusion and reperfusion injury (Rehni and Singh, 2007; Zarruk et al., 2011). In support, several studies have reported the protective effect of natural compounds against ischemic-reperfusion induced neurobehavioral alterations. Our findings are also supported by the previous studies, where natural compounds reversed the ischemia-reperfusion injury induced neurobehavioral alterations, due to their antioxidant and anti-inflammatory potential (Choi et al., 2004; Saleem et al., 2006; Jiang et al., 2007).
Cerebroprotective action of Urtica dioica observed in our study is strongly supported by previous studies conducted on Urtica dioica. Vafaee et al. (2012) reported the protective effect of the immunomodulary drug Setarud, containing extracts of Rosa canina, Urtica dioica and Tanacetum vulgare, supplemented with selenium against ischemic cerebral damage. It has been shown to reduce cerebral infarct volume, and improve the motor function of rats with cerebral ischemia (Vafaee et al., 2012).
Conclusion
Urtica dioica showed cerebroprotective potential by limiting the infarct size in ischemia reperfusion injured rats. In this experiment we found that Urtica dioica showed dose dependent cerebroprotective effect with the selected doses of 0.2, 0.8 ml/kg. Marked impairment of memory and motor co-ordination in parallel to the significant increase in the infarction size was observed after BCCA occlusion for 30 min followed by 24 hours reperfusion in rats. Pretreatment with Urtica dioica significantly and dose dependently attenuated ischemia-reperfusion induced neurobehavioral alterations in the rats. Present study suggests the protective effect of Urtica dioica and its therapeutic potential against ischemia-reperfusion induced neurobehavioral alterations in rats.
Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Acknowledgments
G. Pulla Reddy College of Pharmacy, Hyderabad for supporting and permitting to do this study.
Financial support
The authors declared no financial support.
References
Beal CC. 2010. Gender and stroke symptoms: A review of the current literature. Journal of Neuroscience Nursing, 42(2):80–87.
Bederson JB, Pitts LH, Tsuji M, Nishimura MC, Davis RL, Bartkowski H. 1986. Rat middle cerebral artery occlusion: Evaluation of the model and development of a neurologic examination. Stroke, 17(3):472–476.
Benedek A, Móricz K, Jurányi Z, Gigler G, Lévay G, Hársing LG, Mátyus P, Szénási G, Albert M. 2006. Use of TTC staining for the evaluation of tissue injury in the early phases of reperfusion after focal cerebral ischemia in rats. Brain Research, 1116(1):159–165.
Chandrakant Nimgulkar C, Dattatray Patil S, Dinesh Kumar B. 2011. Anti-asthmatic and anti-anaphylactic activities of Blatta orientalis mother tincture. Homeopathy, 100(03):138–143.
Chemerinski E, Robinson RG. 2000. The neuropsychiatry of stroke. Psychosomatics, 41(1):5–14.
Choi Y Bin, Kim YI, Lee KS, Kim BS, Kim DJ. 2004. Protective effect of epigallocatechin gallate on brain damage after transient middle cerebral artery occlusion in rats. Brain Research, 1019(1-2):47–54.
Dobkin BH. 1991. The rehabilitation of elderly stroke patients. Clinics in Geriatric Medicine, 7(3):507–523.
Feeney DM, Boyeson MG, Linn RT, Murray HM, Dail WG. 1981. Responses to cortical injury: I. Methodology and local effects of contusions in the rat. Brain Research, 211(1):67–77.
Gaur V, Aggarwal A, Kumar A. 2009. Protective effect of naringin against ischemic reperfusion cerebral injury: Possible neurobehavioral, biochemical and cellular alterations in rat brain. European Journal of Pharmacology, 616(1-3):147–154.
Ghosh S, Bishayee K, Paul A, Mukherjee A, Sikdar S, Chakraborty D, Boujedaini N, Khuda-Bukhsh AR. 2013. Homeopathic mother tincture of Phytolacca decandra induces apoptosis in skin melanoma cells by activating caspase-mediated signaling via reactive oxygen species elevation. Journal of Chinese Integrative Medicine, 11(2):116–124.
Hatware KV, Annapurna A, Sharma S, Karri S. 1999. Studies on cerebroprotective activity and establishment of probable mechanism of action of dimethyl fumarate (dmf) against the ischemia and reperfusion induced cerebral injury in wistar rats. Injury 7:9.
Hock FJ. 2015. Drug discovery and evaluation: Pharmacological assays, fourth edition.
Hunter AJ, Hatcher J, Virley D, Nelson P, Irving E, Hadingham SJ, Parsons AA. 2000. Functional assessments in mice and rats after focal stroke. Neuropharmacology, 39(5):806–816.
Jenkins LW, Povlishock JT, Lewelt W, Miller JD, Becker DP. 1981. The role of postischemic recirculation in the development of ischemic neuronal injury following complete cerebral ischemia. Acta Neuropathologica, 55(3):205–220.
Jiang J, Wang W, Sun YJ, Hu M, Li F, Zhu DY. 2007. Neuroprotective effect of curcumin on focal cerebral ischemic rats by preventing blood-brain barrier damage. European Journal of Pharmacology, 561(1-3):54–62.
Jingtao J, Sato S, Yamanaka N. 1999. Changes in cerebral blood flow and blood brain barrier in the gerbil hippocampal CA1 region following repeated brief cerebral ischemia. Medical Electron Microscopy, 32(3):175–183.
Khuda-Bukhsh AR, Paul S, Bhattacharyya SS, Boujedaini N. 2011. Anticancer potentials of root extract of Polygala senega and its PLGA nanoparticles-encapsulated form. Evidence-based Complementary and Alternative Medicine,
Kumar S, Sharma A. 2005. Anti-anxiety activity studies on homoeopathic formulations of Turnera aphrodisiaca Ward. Evidence-based Complementary and Alternative Medicine, 2(1):117–119.
Leira R, Blanco M, Rodríguez-Yáñez M, Flores J, García-García J. 2006. Non-pharmacological neuroprotection: Role of emergency stroke management. Cerebrovascular Diseases, 21(Suppl. 2):89–98.
Lin L, Wang X, Yu Z. 2016. Ischemia-reperfusion Injury in the Brain: Mechanisms and Potential Therapeutic Strategies. Biochemistry & Pharmacology: Open Access, 5(4).
Lipton P. 1999. Ischemic cell death in brain neurons. Physiological Reviews, 79(4):1431–1568.
Mamta S, Preeti K. 2014. Urtica Dioica (Stinging Nettle): a Review of Its Chemical, Pharmacological, Toxicological and Ethnomedical Properties. International Journal of Pharmacy, 4(1):270–277.
Mukherjee PK, Ahamed KFHN, Kumar V, Mukherjee K, Houghton PJ. 2007. Protective effect of biflavones from Araucaria bidwillii Hook in rat cerebral ischemia/reperfusion induced oxidative stress. Behavioural Brain Research, 178(2): 221–228.
Pan J, Konstas AA, Bateman B, Ortolano GA, Pile-Spellman J. 2007. Reperfusion injury following cerebral ischemia: Pathophysiology, MR imaging, and potential therapies. Neuroradiology, 49(2):93–102.
Raghavendra M, Maiti R, Kumar S, Trigunayat A, Mitra S, Acharya S. 2009. Role of Centella asiatica on cerebral post-ischemic reperfusion and long-term hypoperfusion in rats. International Journal of Green Pharmacy, 3(2):88–96.
Reddy DS, Kulkarni SK. 1998. Possible role of nitric oxide in the nootropic and antiamnesic effects of neurosteroids on aging- and dizocilpine-induced learning impairment. Brain Research, 799(2):215–229.
Rehni AK, Singh N. 2007. Role of phosphoinositide 3-kinase in ischemic postconditioning-induced attenuation of cerebral ischemia-evoked behavioral deficits in mice. Pharmacological Reports, 59(2):192–198.
Saleem S, Ahmad M, Ahmad AS, Yousuf S, Ansari MA, Khan MB, Ishrat T, Islam F. 2006. Behavioral and histologic neuroprotection of aqueous garlic extract after reversible focal cerebral ischemia. Journal of Medicinal Food, 9(4):537–544.
Sinha N. 1994. A treatise on Homoeopathic Pharmacy. In A treatise on homoeopathic pharmacy. B. Jain Publishers.
Thrift AG, Thayabaranathan T, Howard G, Howard VJ, Rothwell PM, Feigin VL, Norrving B, Donnan GA, Cadilhac DA. 2017. Global stroke statistics. International Journal of Stroke, 12(1):13–32.
Upton R. 2013. Stinging nettles leaf (Urtica dioica L.): Extraordinary vegetable medicine. Journal of Herbal Medicine, 3(1):9–38.
Vafaee F, Zangiabadi N, Pour FM, Dehghanian F, Asadi-Shekaari M. 2012. Neuroprotective effects of the immunomodulatory drug Setarud on cerebral ischemia in male rats. Neural Regeneration Research, 7(27):2085–2091.
Wu PF, Zhang Z, Wang F, Chen JG. 2010. Natural compounds from traditional medicinal herbs in the treatment of cerebral ischemia/reperfusion injury. Acta Pharmacologica Sinica, 31(12):1523–1531.
Yan B, Bi X, He J, Zhang Y, Thakur S, Xu H, Gendron A, Kong J, Li XM. 2007. Quetiapine attenuates spatial memory impairment and hippocampal neurodegeneration induced by bilateral common carotid artery occlusion in mice. Life Sciences, 81(5):353–361.
Zarruk JG, García-Yébenes I, Romera VG, Ballesteros I, Moraga A, Cuartero MI, Hurtado O, Sobrado M, Pradillo JM, Fernández-López D, Serena J. 2011. Neurological tests for functional outcome assessment in rodent models of ischaemic stroke. Revista de Neurologia, 53(10):607–618.