

Sharba Kausar1, Wajihullah Khan1*, Ameer Azam2
1Section of Parasitology, Department of Zoology, Faculty of Life Sciences, Aligarh Muslim University, Aligarh-202002 (U.P.), India
2Section of Applied Physics, Zakir Hussain College of Engineering and Technology, Aligarh Muslim University, Aligarh-202002 (U.P.), India
*Corresponding author
Wajihullah Khan,
Section of Parasitology, Department of Zoology, Aligarh Muslim University, Aligarh (U.P.), India.
Phone No.: +91 9045493671
Abstract
Background: Setaria cervi which is a cosmopolitan bovine filariid causing peritonitis and lumbar paralysis in domestic and farm animals by circulating microfilariae in blood. Efficacy of Diethyl Carbamazine (DEC), Nitazoxanide (NTZ) and a combination of Nitazoxanide and Silver nanoparticles (NTZ+AgNP) was observed on the enzyme activities of the microfilariae which may help in blocking the transmission to healthy animals. Methods: Effects of DEC, NTZ and NTZ+AgNP were assessed by motility inhibition assay of microfilariae (mf) at different concentrations ranging from 10-100µg/ml at hourly intervals in vitro. Localization of succinate dehydrogenase (SDH), malate dehydrogenase (MDH) and isocitrate dehydrogenase (ICDH) was observed in the mf which were incubated for 6 hrs in 100µg/ml of incubationg medium of aforesaid enzymes. SEM pictures were also taken to see apparent changes on the microfilariae. Results: Highest mortality rate of mf was observed in NTZ+AgNPs compared to NTZ whereas, DEC treated microfilariae showed no significant effect. NTZ+AgNPs treated microfilariae showed maximum inhibition of TCA cycle enzymes. The sheath of microfilariae also gets ruptured when treated with NTZ+AgNP. No such alterations were observed on the surface of microfilariae treated with NTZ and DEC alone. Conclusion: Nitrazoxanide which is a broad-spectrum anthelmintic when combined with AgNPs improved its efficacy against S. cervi mf which may be tried in vivo to check transmission of bovine as well as other filarial infections of man and animals in tropical countries.
Key words: Anthelmintics, nanoparticles, TCA cycle enzymes, microfilariae, Setaria cervi
Introduction
Setaria cervi, a cosmopolitan bovine filariid causing peritonitis and cerebro-setariasis which may also results in lumbar paralysis in cattle (Pachauri, 1972). Fragmentary reports are available regarding the effects of anthelmintics on the longevity and enzyme activities of the adult worms and mf of different species of S. cervi in vitro and in vivo (Ahmad and Srivastava, 2007; Singh and Rathaur 2010; El-Shahawi 2010; Srinivasan et al., 2011). The effect of anthelmintics on a number of enzymes of glycolysis and oxidative pathways have been assessed in the parasites living in the peritoneum, GI tract and tissues of hosts (Anwar et al., 1977; Walter, 1979; Agarwal et al., 1990; Hussain et al., 1990; Ahmad and Srivastava, 2007; Srinivasan et al., 2011; Khan et al., 2012). Widespread anthelmintic resistance has been reported among nematodes of livestock as a consequence of frequent administration of the same class of compounds over long periods (Wolstenholme et al., 2004, Von Samson-Himmelstjerna and Blackhall, 2005) and therefore, combination chemotherapy was considered as a powerful strategy to slow it down. (Barnes et al., 1995; Nyunt and Plowe, 2007). Nitazoxanide, a broad-spectrum thiazolide compound possesses anthelmintic, antiprotozoal and antiviral properties (Rossignol and Stachulski, 1999; Hemphill et al., 2006). Its therapeutic efficacy has been observed against Ascaris lumbricoides and Trichuris trichiura when multiple doses were administered (Fox and Saravolatz, 2005; Anderson and Curran, 2007; Van den Enden, 2009). Recently nanomedicine has been successfully tried against microorganiasms (Dubey, 2006; Patel et al., 2011; Brigger et al., 2002; Merisko-Liversidge et al., 2003; Gherbawy et al., 2013). AgNPs formulation is one of them which demonstrated significant anti-leishmanial effects by inhibiting the proliferation and metabolic activity of promastigotes (Allahverdiyev et al., 2011). SEM pictures revealed cuticular damage in nematode parasites against benzimidazole class of drugs (Hanser et al., 2003; Matadamas-Martínez et al., 2013). Plant extracts and nitazoxanide also showed antiparasitic action against Trichuris muris (Stepek et al., 2006; Tritten et al., 2012).
Keeping above findings in view the present study was conducted to observe potential of diethyl carbamazine (DEC) and nitazoxanide (NTZ) as nematocidal drugs. A combination of NTZ with silver nanoparticles (AgNPs) was also applied against the mf. In vitro effects of these drugs were observed on the mf of S. cervi by using the motility inhibition assay. Scanning-electron microscopy and histochemical localizations were done to see the effect of these drugs on the mf of S. cervi in vitro.
Materials and methods
Adult S. cervi worms were collected from freshly slaughtered buffaloes and brought to the laboratory. The worms were washed in normal saline and adult females were dissected to recover mf from the gravid segment of the uterus for in vitro study. Effect of DEC, NTZ and NTZ+AgNPs was studied by using their different concentrations in different rows of the wells of micro titer plates. For each drug 10 concentrations ranging from 10µg/ml to 100µg/ml were used. The effects of these drugs were assessed by motility inhibition assay as earlier described by Sahare and Singh 2013. Three replicates for each drug were run at the concentrations of 10, 20, 30, 40, 50, 60, 70, 80, 90 and 100µg/ml made in Ringer’s solution. Motility inhibition assay was carried out by keeping mf in 200µl of Ringer’s solution in 96-well titer plate. The inhibition of motility/mortality of the mf in the above treatments was used as the criterion for anthelmintic activity. The motility inhibition was observed every hour.
Localization of Succinate, Malate and Isocitrate dehydrogenases
The drug treated and untreated mf were incubated for 1-2 hours at 37 0C in incubating medium consisting of 18 ml incubating solution (0.2 M Tris buffer, pH 7.4; nitro BT; MgCl2; water) and 2 ml substrate of 2.5 M Di Na Succinate for the localization of succinate dehydrogenate (SDH), 1 M Malic acid for malate dehydrogenase (MDH) and 1 M Tri sodium isocitrate for isocitrate dehydogenase (ICDH). 2-4 mg coenzyme NAD was added for MDH and NADP for ICDH enzyme activities. After incubation counterstaining was done by 2% methyl green for 2-5 min. These sections were then washed and mounted in glycerin jelly as earlier described by Bancroft and Gamble (2002).
Scanning electron microscopic study
The effect of DEC, NTZ and NTZ+AgNPs on the sheath of mf of Setaria cervi was observed using scanning electron microscope (SEM). After 6h of incubation in 100µg/ml concentration of the drugs, mf were fixed in 2.5% glutaraldehyde in PBS (pH 7.4) for approximately 24 h at room-temperature. Mf were washed three times in PBS and stored in it at 4 0C until used. Before SEM examination, the samples were dehydrated stepwise for 10 min each in ascending grades of ethanol (30%, 50%, 70%, 80%, 90% and 100%), at room-temperature and kept in 96% ethanol at 4 0C as described by Manneck et al. (2010). Finally, the worms were dried to critical point, fixed on aluminum stubs and sputter coated with 20 nm gold particles. SEM pictures were taken with a high-resolution scanning electron microscope.
Results
In-vitro motility inhibition assay
DEC, NTZ and NTZ+AgNPs were used for screening against microfilariae of S. cervi. 30mg and above concentration of NTZ+AgNPs caused 100% mortality of microfilariae (mf), while 20µg and 10 µg/ml of this formulation immobilized them completely within 2 and 3 hrs, respectively at 37ºC. NTZ alone was less effective as it could immobilize mf after 1 hour at the concentration of 100µg/ml, while it took 6 hours to attenuate the mf at the concentration of 10 µg/ml. DEC treated mf showed no significant effect at all the concentrations when compared to untreated microfilariae which were placed in Ringer’s solution (Table 1). Exposure incubation in fresh medium for 2hrs did not revive the microfilariae, confirming their death. Thus, the results indicated that NTZ+AgNPs was the most effective formulation as inhibition in motility was fastest at low concentration as compared to other two drugs.
Histochemical localization of TCA cycle enzymes (Succinate, Malate and Isocitrate dehydrogenase)
Localization of TCA cycle enzymes are shown in Tables 2-4. Succinate dehydrogenase activity was moderate throughout the body in DEC, mild in NTZ and negligible in NTZ+AgNPs treated microfilariae when compared with untreated control where intense activity of this enzyme was observed. In DEC and NTZ treated mf SDH activity in excretory pore and anal pore was moderate to intense, while it was mild in NTZ+AgNPs treated mf (Figure 1). Malate dehydrogenase activity was moderate throughout the body except cephalic cells, excretory pore and anal pore where the reaction was intense in untreated mf. Enzyme activity was mild except in excretory and anal pores where high enzyme activity was observed in the mf treated with DEC. Low activity of MDH was observed in anal pore which was very feeble on rest of the body of mf treated with NTZ. In NTZ+AgNPs treated mf no enzyme activity was seen. Isocitrate dehydrogenase activity was mild throughout the body in normal as well as DEC treated microfilariae, while it was mild and very feeble in the mf treated with the combination of NTZ+AgNPs. In untreated mf intense activity of isocitrate dehydrogenase was observed in the cephalic cells, excretory and anal pore which was moderate in excretory and anal pore of DEC and NTZ treated MF. However no activity of this enzyme was observed in mf treated with NTZ+AgNPs (Figiure 3).
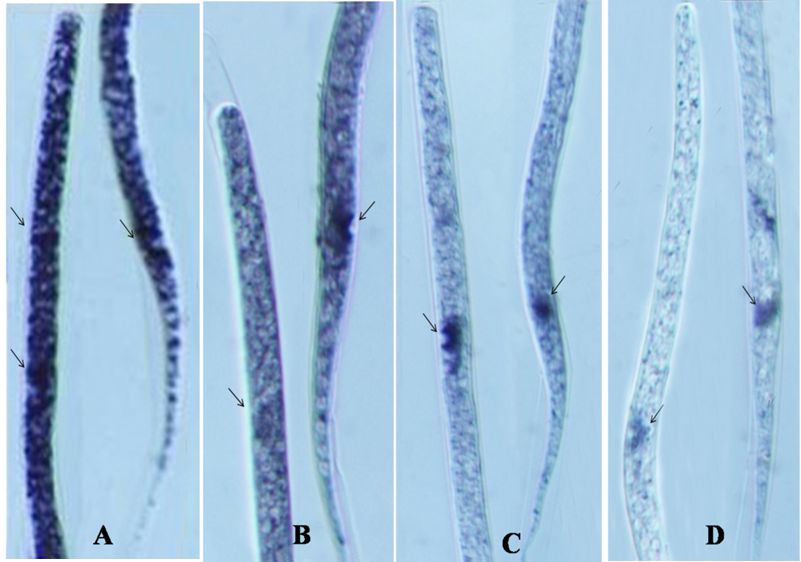
Figure 1. Localization of succinate dehydrogenase in the control (A), DEC (B), NTZ (C) and NTZ+AgNP (D) treated microfilariae of Setaria cervi
Figure 2. Localization of malate dehydrogenase in the control (A), DEC (B), NTZ (C) and NTZ+AgNP (D) treated microfilariae of Setaria cervi

Figure 3. Localization of malate dehydrogenase in the control (A), DEC (B), NTZ (C) and NTZ+AgNP (D) treated microfilariae of Setaria cervi

Figure 4. SEM pictures of Setaria cervi microfilariae incubated in Ringer’s solution for 6 hrs

Figure 5. SEM pictures of Setaria cervi microfilariae incubated in DEC at 100µg/ml Ringer’s solution for 6 hrs

Figure 6. SEM pictures of Setaria cervi microfilariae incubated in NTZ at 100µg/ml Ringer’s solution for 6 hrs

Figure 7. SEM pictures of Setaria cervi microfilariae incubated in NTZ+AgNP at 100µg/ml Ringer’s solution for 6hrs
No significant effect could be observed in SEM pictures of mf treated with 100µg/ml of DEC and NTZ when compared with control, except shrinkage of the sheath (Figures 4, 5 and 6). NTZ+AgNPs treated mf showed eroded sheath at few places along with nanoparticles which were sticking to it (Figure 7).
Table 1: In vitro antifilarial activity of DEC, NTZ and NTZ+AgNPs against microfilariae of
Setaria cervi in terms of motility inhibition
|
NTZ Concentration (µg/ml) |
NTZ+AgNP Concentration (µg/ml) |
Incubation time (Hours) |
Microfilariae motility (Test) |
Microfilariae motility (control and DEC) |
|
10 µg/ml |
- |
6 |
100 |
0 |
|
20 µg/ml |
- |
5 |
100 |
0 |
|
30 µg/ml |
- |
4 |
100 |
0 |
|
40-60 µg/ml |
10 µg/ml |
3 |
100 |
0 |
|
70-90 µg/ml |
20 µg/ml |
2 |
100 |
0 |
|
100 µg/ml |
30-100 µg/ml |
1 |
100 |
0 |
Table 2: Succinate dehydrogenase localization in the treated and untreated microfilariare of Setaria cervi
|
Parameters |
Control |
DEC |
NTZ |
NTZ+AgNP |
|
Nerve ring |
+++ |
++ |
+ |
- |
|
Nuclear column |
+++ |
++ |
+ |
- |
|
Cephalic cells |
+++ |
++ |
+ |
- |
|
Muscles cells |
+++ |
++ |
+ |
± |
|
G-cells |
+++ |
++ |
+ |
+ |
|
Innenkorper thread |
+++ |
++ |
+ |
+ |
|
Excretory pore |
+++ |
++ |
++ |
++ |
|
Anal pore |
+++ |
+++ |
++ |
++ |
(Strong) +++, (Moderate) ++, (weak) +, (Slight) ±, (Absent) –
Table 3. Malate dehydrogenase localization in the treated and untreated microfilariae of Setaria cervi
|
Parameters |
Control |
DEC |
NTZ |
NTZ+AgNP |
|
Nerve ring |
++ |
+ |
- |
- |
|
Nuclear column |
+++ |
+ |
- |
- |
|
Cephalic cells |
+++ |
+ |
- |
- |
|
Muscles cells |
+++ |
++ |
- |
- |
|
G-cells |
+++ |
++ |
- |
- |
|
Innenkorper thread |
+++ |
+ |
- |
- |
|
Excretory pore |
+++ |
++ |
- |
- |
|
Anal pore |
+++ |
++ |
+ |
- |
(Strong) +++, (Moderate) ++, (weak) +, (Slight) ±, (Absent) –
Table 4. Isocitrate dehydrogenase localization in the treated and untreated microfilariae of Setaria cervi
|
Parameters |
Control |
DEC |
NTZ |
NTZ+AgNP |
|
Nerve ring |
- |
- |
- |
- |
|
Nuclear column |
+ |
± |
± |
- |
|
Cephalic cells |
+++ |
+ |
- |
- |
|
Muscles cells |
++ |
+ |
± |
- |
|
G-cells |
+ |
+ |
- |
- |
|
Innenkorper thread |
- |
- |
- |
- |
|
Excretory pore |
+++ |
++ |
+ |
- |
|
Anal pore |
+++ |
++ |
+ |
- |
(Strong) +++, (Moderate) ++, (weak) +, (Slight) ±, (Absent) –
Discussion
During the present study efficacies of DEC, NTZ and NTZ+AgNPs were observed in vitro. NTZ+AgNPs was the most effective drug which killed the mf at the concentration of 30µg/ml within 1 hour of incubation which might be due to the induction of mitochondrial apoptotic pathway by the increased production of ROS. The increased level of ROS creates redox imbalance and promotes apoptosis through up regulating pro-apoptotic gene expression as earlier observed by green silver nanoparticles from Acacia auriculiformis at a very low dose (Saini et al., 2015). No significant effect was observed on the microfilariae treated with DEC in vitro. The probable reason might be lack of body factor specially opsonizing effect by the immune bodies as earlier indicated by Misra et al., (1990). Similar in vitro results in DEC treated Brugia malayi was obtained by Murthy and Chatterjee, (1999). NTZ was effective against the mf in the present study where marked motility was noticed. Similar mortality was observed in the larvae and adult worms of Trichuris muris and Ancylostoma ceylanicum using the motility assay (Stepek et al., 2006, Kopp et al., 2008, Silbereisen et al., 2011). Earlier workers indicated that NTZ and TZ targets energy metabolism by inhibition of pyruvate ferredoxin oxidoreductase (PFOR) and protein disulfide isomerase (PDI) enzyme activities to attenuate and kill of the parasite (Hemphill et al., 2006).
Glycolytic and TCA cycle enzymes have been observed in adult filarial worms, but only a few fragmentary reports are available regarding their localization and distribution in microfilariae Anthelmintics are known to inhibit a variety of enzymes in adult nematodes. We observed NTZ+AgNPs as most potent inhibitors of succinate, malate and isocitrate dehydrogenase as compared to DEC and NTZ. Localization of succinate dehydrogenase almost on the entire body of microfilariae indicates the presence of PEP-succinate pathway in the microfilariae of S. cervi. Strong MDH activity on entire body of mf except nerve ring indicated its role in the energy metabolism. Similar intense localization of this enzyme was reported by earlier workers in S. digitata and B. malayi (Banu et al., 1991, Bhandary et al., 2006). This enzyme catalyzes one of the steps of TCA cycle by oxidizing malate to oxaloacetate and vice versa. It has been observed that the oxaloacetate reduction reaction was more active than their respective opposite reactions in S. digitata, suggesting that the pathway proceeds in the direction of malate formation which seems true in our study also where localization of MDH was intense throughout the body of mf. It has been reported by many workers that the malate which formed as a result of cytosolic MDH activity enters into the mitochondria for further catabolic processes affect the parasite survival adversely (Barrett, 1981, Saz, 1981, Ward, 1982). Thus decrease in SDH and MDH activities in treated mf point towards the blockage of PEP-succinate pathway and a shift towards homolactate fermentation as earlier reported by Rathaur, (1982). In our study we observed low activity of ICDH enzyme throughout the body except cephalic cells and anal pore where activity was more pronounced indicating its pronounced activity in the parts related to secretion and excretion. Localization of complete sequence of the TCA-cycle enzymes was reported in many filarial nematodes including, Onchocerca volvulus, O. gibsoni, O. gutturosa, O. lienalis, B. pahangi and Dirofilaria immitis.Though the enzymes involved in the initial steps of the cycle showed low activity in most of the species of Onchocerca except in O. faciata which showed strong ICDH activity pointing towards an additional pathway (pyruvate-succinate) of glucose metabolism via a reverse sequence of the TCA cycle in the parasite (Middleton and Saz, 1979, Omar et al., 1996, Walter and Albiez, 1986, Dunn et al., 1988).
In the present study we observed NTZ+AgNPs as most effective inhibitor of TCA cycle enzyme as it slowed down SDH to large extent and completely hinder MDH and ICDH activities in treated mf. This indicates that it affects the PEP-succinate pathway due to the synergistic effect of AgNPs with nitazoxanide in which NTZ inhibits the energy metabolism and AgNPs regulates mitochondrial apoptotic pathway in microfilariae as earlier indicated by Hemphill et al., (2006) and Saini et al., (2016).
DEC was less effective as moderate SDH and comparatively low MDH and ICDH activities were observed in the mf after its treatment, indicating its action as inhibitor of glucose uptake and glycogen synthesis in the microfilariae which is in agreement with the earlier findings where similar effects were observed in the mf of S. cervi and Litomosoides carinii (Anwar et al., 1978, Rathaur et al., 1980, Zakai and Khan, 2015). Thus decrease in SDH, MDH and ICDH in DEC treated microfilariae point towards the blockage of PEP-succinate pathway and a shift towards homolactate fermentation as earlier observed by Rathaur et al., (1982).
SEM did not show any marked effect of DEC and NTZ (100µg/ml) on the microfilariae of S. cervi when compared with control except shrinkage in the sheath. On contrary to this Peixoto et al., (2003) reported sheath loss and wrinkled appearance of the microfilariae of W. bancrofti when treated with DEC. Chandrashekar et al., (1984) observed changes on the surface of microfilariae of L. sigmodontis and B. pahangi and concluded that these changes help in exposing the main body of the microfilariae to the antigenic determinants which in turn triggers secondary immunological damage. Therefore, DEC seems to have different pharmacological mechanism of action for different filarial species and various development stages for the same species as earlier indicated by Ottesen, (1984). Recent studies by SEM with the third stage of subperiodic B. malayi showed no morphological changes with DEC or ALB alone or in combination (Tippawangkosol et al., 2004). Effect of NTZ and TZ was also reported in B. malayi, Echinococcus granulosus and Hymenolepis diminuta where these drugs target and cause some structural and functional changes in mitochondria (Walker et al., 2004, Hemphill et al., 2006, Rao et al., 2009).
We have studied the effect of NTZ on microfilariae of S. cervi using SEM and could not find any marked effect which is in corroboration of earlier studies against Trichuris muris and Ancylostoma ceylanicum where no morphological damage was noticed by applying 200 µg/ml of Nitazoxanide (Tritten et al., 2012). NTZ+AgNPs treated microfilariae showed brocken sheath along with attached nanoparticles on the whole body. The sheath was eroded at the anterior and posterior regions of the microfilariae. Our result is in agreement with earlier studies where similar breaks on the cuticle were observed by AgNPs treatment in Panagrellus redivivus and Caenorhabditis elegans (Kim et al., 2012, Mahmoud et al., 2016).
Conclusion
Effect of Diethyl carbamazine (DEC), Nitazoxanide (NTZ) and a combination of NTZ and silver nanoparticles (AgNPs) was observed by motility inhibition assay and highest mortality rate of mf was observed in NTZ+AgNPs. It is therefore concluded that NTZ+AgNPs was the most effective synergistic combination against the TCA cycle enzymes which blocked the ICDH and MDH completely and SDH to large extent in the mf of S. cervi. It seems that AgNPs ruptured the sheath which made NTZ accessible to the main body of the mf and produce maximum effect by penetrating through the body wall and acting on the TCA cycle enzymes which plays a vital role in the energy metabolism and survival of mf. This formulation may be used to control the transmission of bovine and other filariids in tropical countries.
Acknowledgement
Financial support by Maulana Azad National Fellowship of University Grants Commission, New Delhi, India to the first author is gratefully acknowledged.
References
Agarwal A, Tekwani BL, Shukla OP, Ghatak S. 1990. Effect of anthelmintics and adenosine 5'- triphosphatases of filarial parasite Setaria cervi. Indian Journal of Experimental Biology 28: 245-248.
Ahmad R, Srivastava AK. 2007. Biochemical composition and metabolic pathways of filarial worms Setaria cervi: search for new antifilarial agents. Journal of Helminthology 81: 261-80.
Allahverdiyev AM, Abamor ES, Bagirova M, Ustundag CB, Kaya C, Kaya F, Rafailovich M. 2011. Antileishmanial effect of silver nanoparticles and their enhanced antiparasitic activity under ultraviolet light. International Journal of Nanomedicine 6: 2705-2714.
Anderson VR, Curran MP. 2007. Nitazoxanide: a review of its use in the treatment of gastrointestinal infections. Drugs 67: 1947-1967.
Anwar N, Ansari AA, Ghatak S, Krishnamurti S. 1977. Setaria cervi: Enzymes of glycolysis and PEP-succinate pathway. Zeitschrift Fur Parasitenkunde 51: 275-283.
Anwar N, Srivastava AK, Ghatak S. 1978.Effect of antifilarials on the metabolic activity of Setaria cervi. Indian Journal of Parasitology 2: 101-105.
Bancroft JD, Gamble M (Eds). 2002. Theory and Practice of Histological Techniques. 5th. Churchill Livingstone. pp. 608-610.
Banu MJ, Nellaiappan K, Dhandayuthapani S. 1991. Intermediary carbohydrate metabolism in the filarial worm Setaria digitata. International Journal of Parasitology 21: 795-799.
Barnes EH, Dobson RJ, Barger IA. 1995. Worm control and anthelmintic resistance: adventures with a model. Parasitology Today 11: 56-63.
Barrett J (Ed). 1981. Biochemistry of parasitic helminths. Macmillan Press, London. pp. 308.
Bhandary YP, Krithika KN,Kulkarni S, Reddy MVR,Harinath BC. 2006. Detection of enzymes dehydrogenases and proteases in Brugia malayi filarial parasites. Indian Journal of Clinical Biochemistry 21(1): 1-7.
Brigger I, Dubernet C, Couvreur P. 2002. Nanoparticles in cancer therapy and diagnosis. Advances in Drug Delivery Review 54: 631-651.
Chandrashekar R, Rao UR, Subrahmanyam D. 1984. Effect of diethylcarbamazine on serum-dependent cell-mediated immune reactions to microfilariae in vitro.Tropenmedizin und Parasitologie 35: 177-182.
Dubey R. 2006. Impact of Nanosuspension technology on drug discovery and development. Drug Delivery Technology 6: 65-67.
Dunn TS, Raines PS, Barrett J, Butterworth PE. 1988. Carbohydrate metabolism in Onchocerca gutturosa and Onchocerca lienalis (Nematoda: Filarioidea). International Journal of Parasitology 18: 21-26.
El-Shahawi GA, Abdel-Latif M, Saad AH, Bahgat M. 2010. Setaria equina: in vivo effect of diethylcarmazine citrate on microfilariae in albino rats. Experimental Parasitology 126: 603-610.
Fox LM, Saravolatz LD.2005. Nitazoxanide: a new thiazolideantiparasitic agent. Clinically Infectious Diseases 40: 1173-1180.
Gherbawy YA, Shalaby IM, El-Sadek MSA, Elhariry HM, Banaja AA. 2013. The Anti-Fasciolasis Properties of Silver Nanoparticles Produced by Trichoderma harzianum and Their Improvement of the Anti-Fasciolasis Drug Triclabendazole. International Journal of Molecular Science 14: 21887-21898.
Hanser E, Mehlhorn H, Hoeben D, Vlaminck K.2003. In vitro studies on the effects of flubendazole against Toxocara canis and Ascaris suum. Parasitology Research 89: 63-74.
Hemphill A, Mueller J, Esposito M. 2006. Nitazoxanide, a broad-spectrum thiazolide anti-infective agent for the treatment of gastrointestinal infections. Expert Opinion Pharmacotherapy 7: 953-964.
Hussain H, Shukla OP, Ghatak S, Kaushal NA. 1990. Enzymes of PEP –succinate pathway in Setaria cervi and effect of anthelmintic drugs. Indian Journal of Experimental Biology 28: 871-875.
Khan W, Umm-E-AsmaTaufiq F. 2012.Effect of anthelmintics on the enzyme activities of Setaria cervi implanted intraperitoneally in rabbits. Journal of Veterinary Parasitology 26(1): 48-52.
Kim SW, Nam SH, An YJ. 2012. Interaction of silver nanoparticles with biological surfaces of Caenorhabditis elegans. Ecotoxicology and Environmental Safety 77: 64-70.
Kopp SR, Coleman GT, McCarthy JS, Kotze AC. 2008. Phenotypic characterization of two Ancylostoma caninum isolates with different susceptibilities to the anthelmintic pyrantel. Antimicrobial Agents and Chemotherapy52: 3980-3986.
Mahmoud WM, Abdelmoneim TS, Elazzazy AM. 2016. The impact of silver nanoparticles produced by Bacillus pumilus as antimicrobial and nematicide. Frontiers in Microbiology 7: 1-9.
Manneck T, Haggenmuller Y, Keiser J. 2010. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes of Schistosoma mansoni. Parasitology137: 85-98.
Matadamas-Martínez F, Nogueda-Torres B, Hernández-Campos A, Hernández-Luis F, Castillo R, Mendoza, G., Ambrosio, J.R., Andrés-Antonio, G., Yépez-Mulia, L., 2013. Analysis of the effect of a 2-(trifluoromethyl)-1H-benzimidazole derivative on Trichinella spiralis muscle larvae. Veterinary Parasitology194: 193-197.
Merisko-Liversidge E, Liversidge GG, Cooper ER.2003.Nanosizing: A formulation approach for poorly-watersoluble compounds. European Journal of Pharmaceutical Sciences 18: 113-120.
Middleton KR, Saz HJ. 1979. Comparative utilization of pyruvate by Brugia pahangi, Dipetalonema viteae, and Litomosoides carinii. Journal of Parasitology 65(1): 1-7.
Misra S, Singh DP, Murthy PK, Chatterjee RK. 1990. Mode of action of antifilarials: Modulation of immune adherence to microfilariae in vitro. Tropical Medicine32: 33-43.
Murthy PK, Chatterjee RK. 1999. Evaluation of two in vitro test systems employing Brugia malayi parasite for prescreening of potential antifilarials. Current Science 1999; 77(8): 1084-1089.
Nyunt MM, Plowe CV. 2007. Pharmacologic advances in the global control and treatment of malaria: combination therapy and resistance. Clinical Pharmacology and Therapeutics 82: 601-605.
Omar MS, Raoof A, Ai-Amari OM. 1996.Onchocerca fasciata: enzyme histochemistry and tissue distribution of various dehydrogenases in the adult female worm. Parasitology Research 82: 32-37.
Ottesen EA. 1984. The action of diethylcarbamazine on adult worms of the lymphatic-dwelling filariaeWuchereria bancrofti, Brugia malayi and Brugia timori in man, WHO/ FIL/84. 174: 1-24.
Pachauri SP. 1972. Cerebrospinal nematodiasis in a buffalo.A case report. Indian Journal of Animal Research 6: 17.
Patel M, Shah A, Patel NM, Patel MR, Patel KR. 2011. Nano suspension: a novel approch for drug delivery system. Journal of Pharmaceutical Science and Bioscientific research 1(1): 1-10.
Peixoto CA, Alvesa LC, Braynera FA, Floreˆnciob MS. 2003. Diethylcarbamazine induces loss of microfilarial sheath of Wuchereria bancrofti. Micron 34: 381-385.
Rao RU, Huang Y, Fischer K, Fischer PU, Weil GJ. 2009. Brugia malayi: Effects of nitazoxanide and tizoxanide on adult worms and microfilariae of filarial nematodes. Experimental Parasitology 121: 38-45.
Rathaur S, Awar N, Chatterjee RK, Ghatak S. 1980. Metabolic pattern of the microfilariae of Setaria cervi and Litomosoides carinii. Indian Journal of Parasitology 4: 67-69.
Rathaur S, Anwar N, Saxena JK, Ghatak S. 1982. Setaria cervi: enzymes in microfilariae and in vitro action of antifilarials. Zeitschrift Fur Parasitenkunde68(3): 331-338.
Rossignol JF, Stachulski AV. 1999. Syntheses and antibacterial activities of tizoxanide, an N-(nitrothiazolyl) salicyclamide, and its O-aryl glucuronide. Journal of Chemical Research (S): 44-45.
ahare KN, Singh V. 2013. In-vitro antifilarial activity of methanolic extract of Aegle marmelos Corr. Leaves.Indo American Journal of Pharmaceutical Sciences 3(6): 4567-4573.
aini P, Saha SK, Roy P, Chowdhury P, Santi P, Babu S. 2016. Evidence of reactive oxygen species (ROS) mediated apoptosis in Setaria cervi induced by green silver nanoparticles from Acacia auriculiformis at a very low dose. Experimental Parasitology 160: 39-48.
Saz ÇJ. 1981. Energy metabolism of parasitic helminths. Annual Review of Physiology 43: 323-341.
Silbereisen A, Tritten L, Keiser J. 2011. Exploration of novel in vitro assays to study drugs against Trichuris spp. Journal of Microbiological Methods 87: 169-175.
Singh A, Rathaur S. 2010. Combination of DEC plus aspirin induced mitochondrial mediated apoptosis in filarial parasite Setaria cervi.Biochimie 92: 894-900.
Srinivasan L, Mathew N, Karunan T, Muthuswamy K. 2011. Biochemical studies on glutathione S-transferase from the bovine filarial worm Setaria digitata. Parasitology Research 109: 213-219.
Stepek G, Lowe AE, Buttle DJ, Duce IR, Behnke JM. 2006. In vitro and in vivo anthelmintic efficacy of plant cysteine proteinases against the rodent gastrointestinal nematode, Trichuris muris. Parasitology132: 681-689.
Tippawangkoso P, Choochote W, Na-Bangchang K, Jitpakdi A, Pitasawat B, Riyong D. 2004. The in vitro effect of albendazole, ivermectin, diethylcarbamazine, and their combinations against infective third-stage larvae of nocturnally subperiodic Brugia malayi (Narathiwat strain): scanning electron microscopy. Journal of Vector Ecology 29(1): 101-108.
Tritten, L, Silbereisen A, Keiser J. 2012. Nitazoxanide: In vitro and in vivo drug effects against Trichuris muris and Ancylostoma ceylanicum, alone or in combination. International Journal of Parasitology Drugs and Drug Resistance 2: 98-105.
Van Den Enden E. 2009. Pharmacotherapy of helminth infection. Expert Opinion Pharmacotherapy 10: 435-451.
Von Samson-Himmelstjerna G, Blackhall W. 2005. Will technology provide solutions for drug resistance in veterinary helminths? Journal of Veterinary Parasitology 132: 223-229.
Walker M, Rossignol JF, Torgerson P, Hemphill A. 2004. In vitro effects of nitazoxanide on Echinococcus granulosus protoscoleces and metacestodes. Journal of Antimicrobial Chemotherapy 54: 609-616.
Walter RD, Albiez EJ. 1986. Phosphoenolpyruvatecarboxykinase from Onchocerca volvulus and O. gibsoni. Tropical Medicine and Parasitology 37: 356-358.
Walter RD. 1979. Inhibition of lactate dehydrogenase activity of Dirofilaria immitis by suramin.Zeitschrift Fur Tropenmedizin Und Parasitologie 30: 463-465.
Ward PFV.1982. Aspects of helminth metabolism. Parasitology 84: 177-194.
Wolstenholme AJ, Fairweather I, Prichard R, Von-Samson-Himmelstjerna G, Sangster NC. 2004. Drug resistance in veterinary helminth. Trends in Parasitology 20: 469-476.
Zakai HA, Khan W. 2015. Effects of filaricidal drugs on longevity and enzyme activities of the microfilariae of Setaria cervi in white rats. Asian Pacific Journal of Tropical Biomedicine 5(9): 714-719.