

Alaa Habib*
Cardiovascular Research Group, School of Biomedical Sciences, University of Nottingham Medical School, Queen's Medical Centre, Nottingham, NG7 2UH UK. King Abdulaziz University, Jeddah, Saudi Arabia
*Address for Corresponding Author
Alaa Habib
Cardiovascular Research Group, School of Biomedical Sciences, University of Nottingham Medical School, Queen's Medical Centre, Nottingham, NG7 2UH UK. King Abdulaziz University, Jeddah, Saudi Arabia
Abstract
Background: Pannexins are newly discovered proteins that were first discovered by Panchin et al. in 2000. This aim of this work was to examine the presence and function of pannexins in the porcine splenic artery (PSA) in which α1A–adrenoceptors is present. Materials and Methods: The involvement of pannexin channels was studied using several pannexin inhibitors, i.e. mefloquine (a non-selective pannexin inhibitor), probenecid (a selective pannexin-1 inhibitor at low concentrations) and carbenoxolone (a selective pannexin-1 inhibitor). Additionally, the involvement of ATP (via activation of P2 purinoceptors) in NA-induced contractile responses as well as sympathetic nerve activation was examined using P2 purinoceptor antagonists (PPADS and suramin). Results: Our data showed that both pannexin-1 and pannexin-2 are present in the PSA. Mefloquine and probenecid reduced the responses to both noradrenaline-induced contractions and the frequency-dependent response curves generated to sympathetic nerve stimulation, whereas carbenoxolone, suramin and PPADS had no effect on responses to neither exogenous NA nor those caused by activating the sympathetic nerves, arguing against the role of ATP in mediating noradrenaline-induced responses in the PSA. This is because mefloquine demonstrated non-selective inhibitory actions on contractile responses since it was also shown to inhibit responses to 5-HT and U46619 (the thromboxane mimetic). Conclusion: The present work therefore suggests evidence for the involvement of pannexin channels in conducting responses to NA-induced α1-adrenoceptor-mediated vasoconstriction in blood vessels in PSA, although great care must be taken in interpreting this data on the basis of a lack of selectivity of the pharmacological agents currently available as pannexin inhibitors.
Keywords: Noradrenaline, pannexins, porcine splenic artery, purinoceptor agonists, vasculature
Introduction
Pannexin channels were first discovered by Panchin et al. (2000) as integral membrane proteins in mammalian genomes (Panchin et al., 2000). The Pannexin family comprises three members, i.e. pannexin-1, pannexin-2, and pannexin-3 (Bond and Naus, 2014). These proteins have some resemblance in their membrane topology to connexins (Ambrosi et al., 2010; Makarenkova et al., 2018). However, the role for Pannexin-1 oligomers (that forms a channel between the cytosol and extracellular space) is different from that of connexon oligomers (that form intercellular gap junctions between adjacent cells). The key dissimilarity is possibly due to the mechanism whereby Pannexin-1 is exceedingly glycosylated on its extracellular loops, which might inhibit docking with Pannexin-1 channels on the adjacent cells (Ohbuchi et al., 2011; Poornima et al., 2012; Sorge et al., 2012; Sumi et al., 2010). Pannexin-1 is ubiquitously present in almost all human tissues including nervous system, muscles and blood vessels (Makarenkova et al., 2018; Baranova et al., 2004; Cea et al., 2014; Shestopalov et al., 2008; Vanden et al., 2006). Whereas the expression of Pannexin-2 is more limited to the central nervous system with lower expression in nonneuronal cells (Dvoriantchikova et al., 2006; Ray et al., 2006). Pannexin-3 is mainly expressed in many tissues including skeletal muscle, sebaceous glands, and the small intestine (Bond et al., 2006; Ishikawa et al., 2011; Turmel et al., 2011; Langlois et al., 2014).
Recently, pannexins have been shown to be expressed in the vasculature, and while their function has yet to be fully determined, research indicated that proposed a mechanism by which the vasoconstriction induced by α1–adrenoceptors in thoracodorsal resistance arteries involved pannexin-1 channels (Billaud et al., 2011; Molica et al., 2018). They found that phenylephrine acting upon the α1D–adrenoceptors caused release of ATP through pannexin channels in mouse thoracodorsal small arteries. ATP then activated P2 receptors on neighbouring smooth muscle cells to enhance vasoconstriction. They concluded that pannexin-1 could contribute to the coordination of the vascular smooth muscle constriction and the regulation of blood pressure as an intermediary for catecholamines released by sympathetic nerves. Although (Billaud et al., 2011) have presented evidence for the involvement of pannexins in controlling the constriction of thoracodorsal resistance arteries, the role of pannexins in mediating responses to NA in other arteries has not been studied. Hence, the aim of this work was to build on the research carried out by Billaud et al. (2011) to provide an overall understanding of the role of pannexin in the regulation of vascular tone. The objectives were to determine (i) the presence of pannexins in PSA by (using Western blot and immunohistochemistry), (ii) the α1–subtype population responsible for mediating NA-induced vasoconstriction in PSA, and (iii) whether pannexins are involved in mediating contractile responses produced by exogenous NA or activation of the sympathetic nerves in PSA.
Materials and Methods
Isometric tension recording
Tissue collection and sample preparation
Porcine tissue was obtained from Wood and Sons Abattoir (Clipston, Nottingham, UK) from breeds of modern hybrid pig, but the sex and exact age of each animal was unknown. Porcine spleens and hearts were isolated at the abattoir and stored in Krebs–Henseleit solution at 4 ˚C during transport to the laboratory. On arrival, gross dissection of splenic arteries were carried out leaving the adipose and connective tissue directly surrounding the vessels intact. Vessels were stored overnight in Krebs–Henseleit solution at 4 ˚C pre-gassed with a 95% O2: 5% CO2 gas mixture. The next day vessels were pinned in a dissection dish and the remaining adipose and connective tissue was removed whilst ensuring vessels were not stretched or damaged. The length of the vessel segments was approximately 5 mm. Tissue was kept in Krebs–Henseleit solution and allowed to normalize to room temperature before being setup in the isometric force transducer. Vessels were attached to two metal hooks placed through the lumen, ensuring that the hooks were not overlapping. One hook was attached to a glass rod and the other was attached to a non-stretchable silk thread. The vessel was then transferred to an organ bath where the glass rod was anchored into the system. The thread attached to one of the hooks was attached to a force transducer (AD Instruments, UK) on a rack and pinion, enabling adjustment of tension. The organ bath contained 20ml Krebs–Henseleit solution and was kept at a constant temperature of 37 ˚C using a thermal circulator pump. The Krebs–Henseleit solution was constantly gassed with the required gas mixture for each experiment. The force transducer was attached to a Bridge Amplifier (ADInstruments, UK) to measure alterations in voltage recorded by the transducer due to changes in the tension applied to the transducer. The Bridge Amplifier was in turn connected to a PowerLab unit (ADInstruments, UK), which connected the transducer to a computer where data was recorded using Lab Chart software (AD Instruments, UK). Transducers were calibrated with a 10 g weight daily to ensure consistency of measurement. Porcine splenic arteries were dissected cut into 4 equal strips approximately 5 mm in length. Four sets of experiments were carried out to investigate the contractile response of porcine splenic arteries to NA in the absence and presence of pannexin inhibitors (mefloquine or probenecid or carbenoxolone) or P2 receptor antagonist (PPADS or suramin).
Sample recordings
As shown previously, segments were exposed to increasing concentrations of NA (10-8 M to 10-4 M) raised in 3–fold increments to generate cumulative concentration curves (CCRC) to NA. The concentration of NA was changed by adding appropriate amounts of NA to the bath as soon as the previous concentration had produced a plateau. After the CCRC was completed, tissues were washed and the whole procedure was repeated again to check the reproducibility of the CCRC curves after a further 30-minute equilibration period. This was compared to CCRC curves to NA generated in the presence of either suramin (10-4M), PPADS (10-5 M), mefloquine (2 × 10-5 M) or probenecid (5 × 10-4 M). Experiments were also carried out on porcine splenic artery using 5HT and U46619 as contractile agents. 5HT was applied at concentrations between 3 × 10-10 M and 10-8 M and U46619 at concentrations from 3 × 10-8 to 10-6 M to produce cumulative concentration curves. The effect of mefloquine 2 × 10-5 M on 5HT and U46619 responses were then determined.
Electrical field stimulation experiment in PSA
Porcine splenic artery ring segments were set up as described previously. Tissues were exposed to NA (10-5 M), followed by a washout using Krebs’-Henseleit solution. Exposure to NA was repeated at least twice before tissues were exposed to electrical field stimulation (EFS). EFS was applied to the artery segments at increasing frequencies (1, 2, 5, 10, 20 Hz, at a voltage of 20 V, pulse width 0.5 ms, for 10 s) at 5 minutes intervals using an SRI electrical stimulator. Frequency-response curves were repeated in the presence and absence of mefloquine (2 × 10-7 M, 2 × 10-6 M and 2 × 10-5 M), probenecid (5 × 10-4 M) carbenoxolone (10-5 M, 2 × 10-5 M and 10-4 M), suramin (10-4 M), and PPADS (10-5 M).
Western blots
Porcine spleens were collected from the abattoir. Following fine dissection, segments were homogenized in lysis buffer [20 mM Tris, 1 mM EGTA, 0.1% (v/v) Triton X-100, 1 mM NaF, 10 mM β-glycerophosphate, pH 7.6], containing protease inhibitor cocktail tablets, EDTA free. Samples with solubilization buffer (6 × SB [24% (w/v) SDS, 30% (v/v) glycerol, 5% (v/v) β-mercaptoethanol, 2.5% (v/v) bromophenol blue, 1.5 M Tris–HCl, pH 6.8) were heated at 95°C for 5 min. Subsequently, 5µg of the porcine tissue were run on 4-20 % Tri Glycine (PAGE) Gold Precast Gels (Bio Rad, Hercules, CA, USA) in electrophoresis buffer (0.19 M Tris hydrochloride, 1.9 M glycine and 35 mM SDS). Electrophoresis was performed at 175V for 35 minutes. The gel was blotted onto nitrocellulose membranes. Following this, the nitrocellulose membrane was blocked in 5% (w/v) milk solution in tris-buffered saline (25mM Tris, 12mM NaCL, 10l dH2O, 10ml Tween 20, pH 7.6), Tween® 20 detergent (TBST) for 1 hour at room temperature. After that the nitrocellulose membrane was incubated overnight at 4oC with the primary antibodies Rabbit anti- pannexin-1 (Abcam cat No. ab124969) and Rabbit anti- pannexin-2 (Abcam cat No. ab55917) at 1:1000 concentration in 5% (w/v) milk solution. Mouse monoclonal anti-GAPDH antibody (G8795 Sigma, UK) (1:40 000) was simultaneously used as housekeeping and loading control. All antibodies where sealed in a plastic bag with the nitrocellulose and incubated over night at 4oC. The following day the primary antibodies were removed by washing with TBST, then blots were probed with corresponding secondary antibodies: IRDye® 680LT Goat anti-mouse IgG (1:10 000) (LI-COR Biosciences, Cambridge, UK) and IRDye® 800CW Goat anti-rabbit IgG (1:10 000) (LI-COR Biosciences, Cambridge, UK) for 1 hour at room temperature. After that the secondary antibodies were removed and the nitrocellulose was washed as before and analysed using Li-Cor Odyssey Imager Scanner and densities of the bands were determined using Odyssey (Application Software Version 3.0LI-COR Biosciences, Cambridge, UK).
Immunohistochemistry
Porcine splenic arteries were dissected and placed in a sponged petri dish full of Krebs–Henseleit solution, where they were finely dissected to remove the connective tissue, then cut to 1 cm in length, immediately placed in 4% formaldehyde and incubated overnight at 4 ºC. The next day, tissues were processed using a Leica bio-system tissue processor 1020 for the tissue to dehydrate. A gentle exchange was achieved by passing the tissue through increasing concentrations of ethyl alcohol (50, 70, 90, 96 & 100%). Once the water had been replaced by alcohol, the alcohol was then replaced with xylene, which is a transitional solvent which is miscible with both the alcohol and molten wax. The final step in processing was to infiltrate the tissue with molten paraffin wax. The following day, the tissue is arranged in a steel mould in the correct orientation for example the porcine arteries were arranged transversely and embedded in molten wax. They were then left to cool and harden prior to sectioning. 5µm sections were cut using a Leica RM2125 microtome (Leica), floated in a water bath set to 42˚ C and transferred to 3-Aminopropyltriethoxysilane (APES) coated slides, which is a chemical solution used to add a charge to the slides, thus helping sections to adhere to the slides better. The slides were then left to dry.
The primary antibodies were diluted in (PBS 0.1%, 0.1% Tween-20, 1%BSA) and incubated overnight, while the secondary antibody, goat anti-rabbit FITC, was diluted in (PBS 0.1%, 0.1% Tween-20, 1%BSA) to a dilution of 1:500, and applied to the sections for 1 h. Following this they were incubated with DAPI (1in 500 dilution) for 5 min in the dark. Coverslips were placed on microscope slides using mounting medium (1% (v/v) 1, 4-diazobicyclo-[2, 2, 2,]-octane (DABCO) DABCO fluorescence mounting medium-SS04. The slides and sections were examined at X20 and X40 using a light microscope and a wide field Fluorescence microscope to assess pannexin location. The following filters were used to visualise the immunostains. DAPI - Excitation 350/50nm, emission 460/50 nm, FITC - Excitation 480/40nm, emission 535/50 nm to visualise the Goat anti rabbit Antibody.
Statistical analysis
Data analysis was carried out using Prism (Graph Pad Software). Data was expressed as mean ± standard error of the mean (s.e.m). Where the N represents the number of animals used in every experiment. The difference between means was considered statistically significant at a value of P <0.05. Statistical significance was determined using two-tailed unpaired Students t-test between two data sets or one-way ANOVA when comparing multiple data-sets. One-way ANOVA was followed by a Bonferroni post hoc test carried out as appropriate (Prism 6.0 software (San Diego, CA)).
Contractions to all agonists were measured in grams and expressed as a percentage of the KCl-induced contraction in porcine tissue. In Electrical field stimulation experiment the responses were measured in grams and expressed as a percentage of NA-induced contractions. The contractions to 5HT and U46619 were expressed as a percentage of their own maximum response in porcine tissue. All data are presented as mean +/- standard error of the mean (s.e.m). In some experiments, the log concentration-response curves were used to determine potency (log EC50 values) and maximum response (Rmax) values. EC50 is the concentration of a drug that produces a response equal to 50% of the maximum response (GraphPad Prism software).
Results
Determination of pannexin expression in PSA
Western blot analysis was carried out to determine pannexin-1 and pannexin-2 expression in PSA using anti-pannexin-1 and 2 specific antibodies. In the absence of any primary antibody we detected no bands on our blot indicating the specificity of our Western blot technique (Supplementary Figure 1d). The expression of pannexin-1 was shown as a 50 kDa band in all samples. The lanes show splenic artery samples from 4 different pigs. The 50 kDa bands observed during western blotting correspond to pannexin-1 protein (Figure 1). The expressing of pannexin-1 was further confirmed using immunolabelling. Immunolabelling of pannexin-1 using anti-pannexin-1 specific antibody was carried out in cross sections and showed endothelium and smooth muscle cells of PSA labeled for presence of pannexin-1 under light and fluorescence microscope at ×20 and ×40. The image shows expression of pannexin-1 in the endothelium and the smooth muscle cells of porcine splenic artery (Figure 2). From Western blot analysis, the expression of pannexin-2 was shown as a 100 kDa band in all lanes corresponding to the pannexin-2 protein (Figure 1).
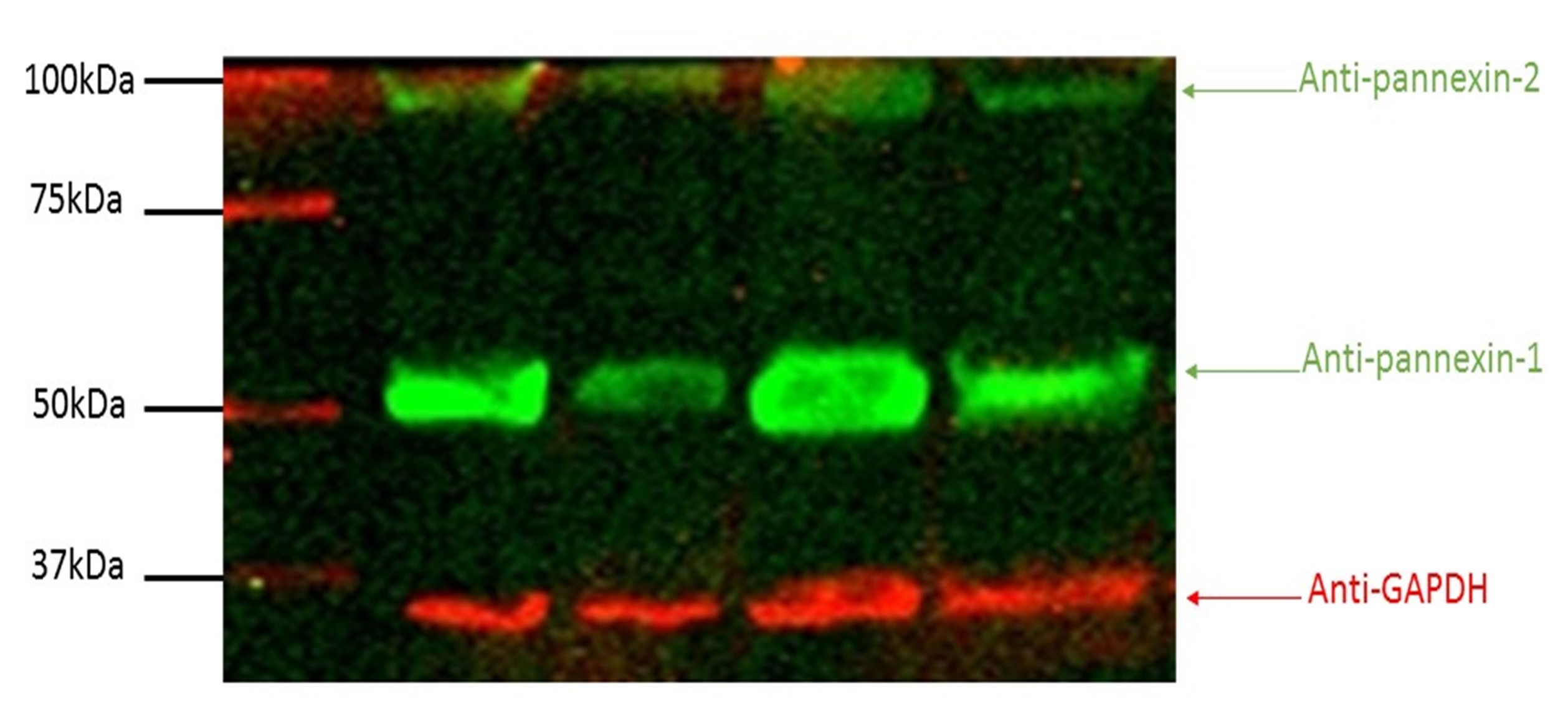 Figure 1. Western blot analysis to determine pannexin-1 and pannexin-2 expression in PSA tissue (N = 4).
Figure 1. Western blot analysis to determine pannexin-1 and pannexin-2 expression in PSA tissue (N = 4).
Isometric tension recording
Effect of NA on tension of PSA
To test whether the response to NA was reproducible in PSA, consecutive concentration-response curves were obtained. The maximum responses did not significantly alter between the two curves; Rmax values were 230.3% ± 27.58% and 221.4% ± 0.08% of the KCl response for curves one and two, respectively (Supplementary Figure 1a). Similarly, the sensitivity to NA did not change between the curves; Log EC50 values were -6.04 ± 0.27 and -6.33 ± 0.28 for curves one and two, respectively (P >0.05, Student’s t-test, n = 8).
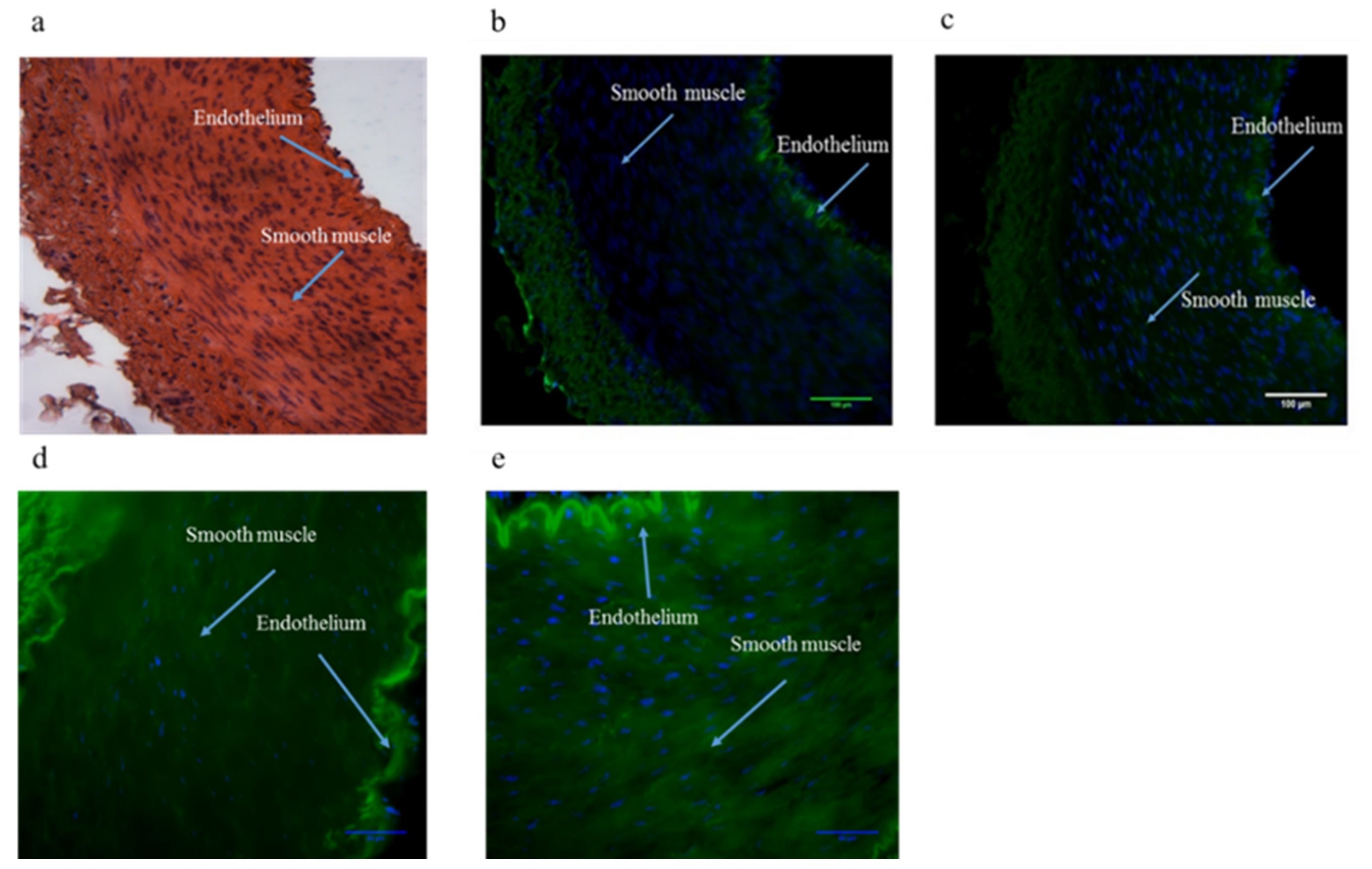
Figure 2. Light microscopic images of porcine splenic artery at X20 (a) stained without primary antibody. Expression of Pannexin-1 shown by immunohistochemistry in the absence (b) and presence (c) of anti pannexin-1 specific antibody at X20. Expression of Pannexin-1 shown by immunohistochemistry in the absence (d) and presence (e) of anti pannexin-1 specific antibody at X40. Expression was evident in the endothelium and the smooth muscle cells of porcine splenic artery. White arrows represent the areas of pannexin-1 expression(N = 6).
Effect of pannexin inhibitors on NA induced contraction in PSA
Mefloquine at 2 ×10-5 M reduced the magnitude of NA-induced contraction in PSA. The maximum responses to NA were 231%.2 ± 8.87% and 118.3% ± 9.44% in the absence and presence of mefloquine respectively (P <0.0001, n = 7) (Figure 3a). However, mefloquine did not alter the sensitivity of responses to NA in PSA; log EC50 values were -5.69 ± 0.08 and -5.62 ± 0.16 in the absence and presence of mefloquine respectively (P > 0.05) (Figure 3a).
Probenecid at 2 ×10-3 M significantly reduced the response to NA in the PSA. The responses to NA (3 × 10-4 M) were reduced from 158.83% ± 11.14% to 96.64% ± 14.14% in the absence and presence of probenecid respectively (P <0.001, n = 8) (Figure 3b). However, similar to mefloquine, probenecid did not alter the sensitivity of responses to NA in PSA (P >0.05) (Figure 3b).
Carbenoxolone, used at three different concentrations of 10-5 M, 2 × 10-5 M and 10 -4 M, did not significantly alter the magnitude of NA-induced contraction in PSA (Figure 3c to 3e). At a concentration of 10-5 M, the maximum response to NA were 149.9% ± 11.31% and 145.8% ± 7.84% in the absence and presence of carbenoxolone respectively (P >0.05, n = 5). The sensitivity (Log EC50) values were -6.25 ± 0.13 and -6.75 ± 0.13 in the absence and presence of carbenoxolone (10-5 M) respectively (P >0.05) (Figure 3c). At a concentration of 2 × 10-5 M, the maximum responses to NA were 175.7% ± 24.86% and 143.7% ± 17.17% in the absence and presence of carbenoxolone respectively (P >0.05, n = 5), whereas the sensitivity values were -5.84 ± 0.27 and -5.65 ± 0.14 in the absence and presence of carbenoxolone respectively (P >0.05) (Figure 3d). At a concentration of 10-4 M, the maximum responses to NA were 170.9% ± 34.65% and 156.5% ± 16.07% in the absence and presence of carbenoxolone respectively (P >0.05, n = 5), whereas the sensitivity values were -6.07 ± 0.21 and -5.80 ± 0.13 in the absence and presence of carbenoxolone respectively (P >0.05) (Figure 3e).
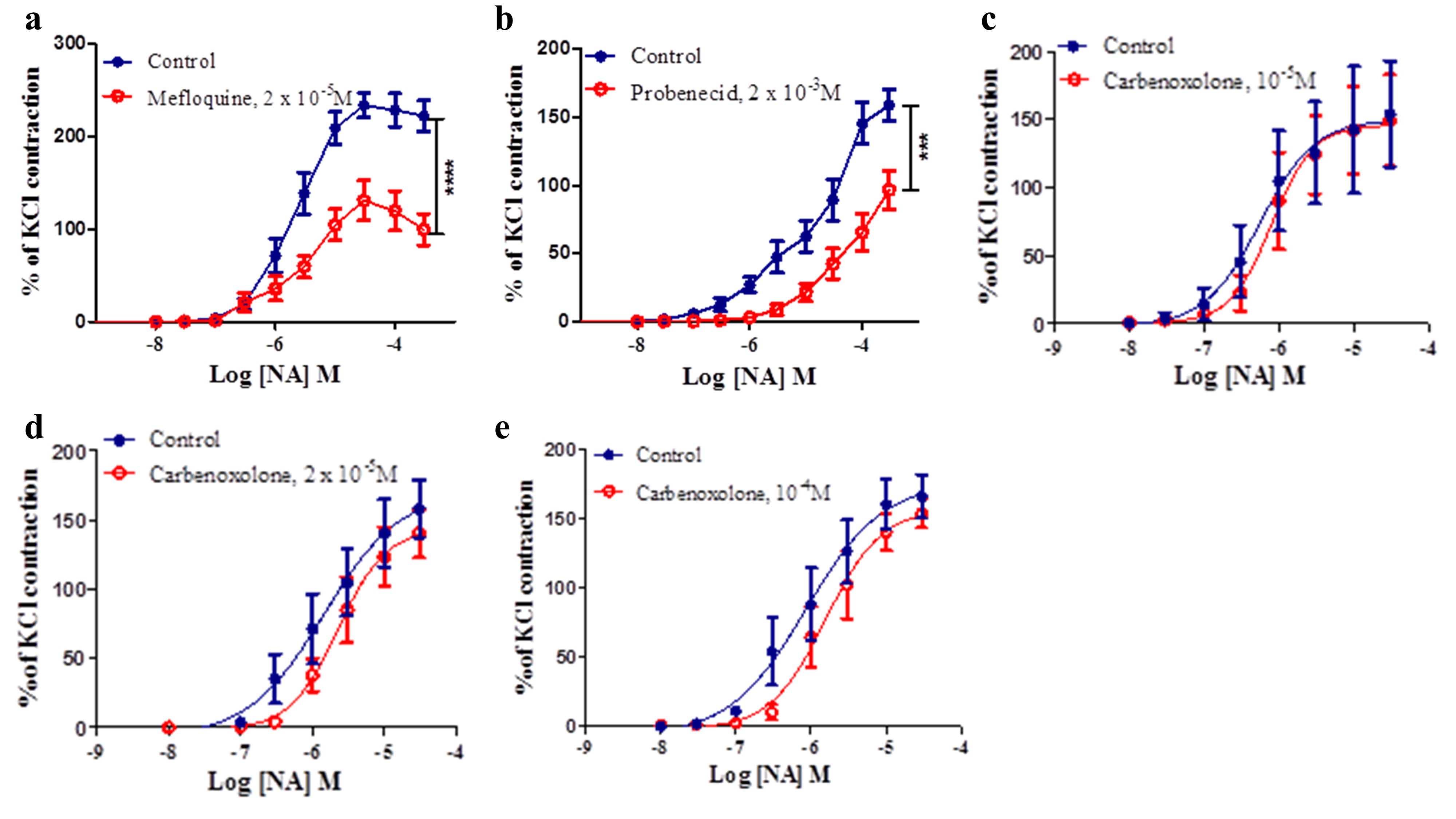
Figure 3. (a) Effect of mefloquine (2 x10-5M) on the response to NA in the PSA. (b) Effect of probenecid (2x10-3M) on the response to NA in the PSA. Effect of carbenoxolone (10-5 (c), 2x10-5 (d) and10-4M (e) on the response to NA in the PSA. The contractions caused by NA were expressed as a percentage of the contractions produced by KCl. Data are presented as the mean value ± s.e.m. Differences between Rmax values are shown by *** P < 0.001**** P < 0.0001 (Student’s paired t-test, N=5-8).
Effect of P2 purinoceptor antagonists on NA induced contraction in PSA
Suramin at 10-4M did not reduce the magnitude of NA-induced contraction in PSA. The maximum responses to NA were 186.2% ± 76.80% and 132.9% ± 19.18% in the absence and presence of suramin (P >0.05, n = 8) (Figure 4a). Suramin also did not alter the sensitivity of responses to NA in PSA; log EC50 values were -5.99 ± 0.54 and -6.41 ± 0.19 in the absence and presence of suramin respectively (P >0.05).
Similar to suramin, PPADS at 10-5 M did not alter the magnitude of NA-induced contraction in PSA. The maximum responses to NA were 175.0% ± 8.48% and 188.7% ± 14.21% in the absence and presence of PPADS (P >0.05, n =8) (Figure 4b). PPADS also did not alter the sensitivity to NA; log EC50values were -5.92 ± 0.10 and-5.79 ± 0.15 in the absence and in the presence of PPADS respectively (P >0.05).
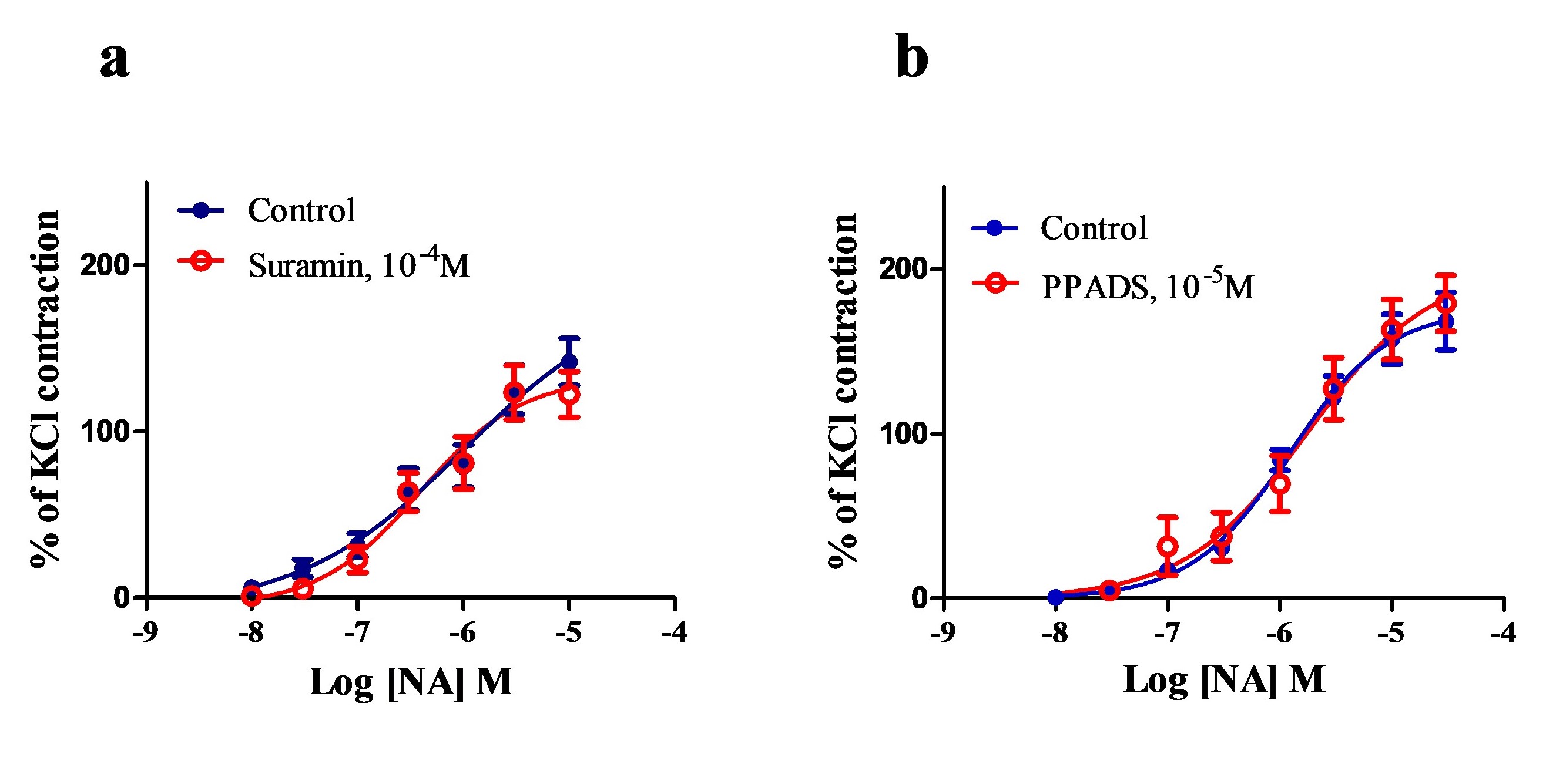
Figure 4. Effect of suramin (10-4M) (a) and PPADS (10 -5M) (b) on the response to NA in the PSA. The contractions caused by NA were expressed as a percentage of the contractions caused by KCl. Data are presented as the mean value ± s.e.m. Suramin (a) and PPADS (b) did not change the contraction to NA in the PSA (P>0.05, Student’s t-test, N =8).
Role of α1-adrenoceptors and/or P2X receptors in mediating electrically-evoked vasocontractile responses in PSA
Under basal tone conditions, EFS elicited frequency-dependent vasoconstrictor responses in the PSA (20 V, pulse width 0.5 msec) at frequencies 1, 2, 5, 10 and 20 Hz were applied for 10 s. The results showed that guanethidine (5 × 10-6 M), a sympatholytic compound, abolished vasoconstriction response evoked by EFS in the PSA at 10 Hz and 20 Hz (P <0.05, P <0.0001, Two-way ANOVA) (Supplementary Figure 1b). We also examined whether the vasoconstriction response to EFS was reproducible in PSA, consecutive responses to EFS were obtained. EFS-induced contraction of PSA was reproducible as there were no significant differences between the responses (Supplementary Figure 1c) (P > 0.05, Two-way ANOVA, n = 6).
Effect of pannexin inhibitors on responses to EFS in the PSA
Mefloquine at 2 × 10-6 M and 2 × 10-7 M did not alter the contraction-induced by EFS (P >0.05, Two-way ANOVA); however, mefloquine at a higher concentration (2 × 10-5 M) significantly reduced the vasoconstriction response evoked by EFS in the PSA at 20 Hz (P <0.0001, Two-way ANOVA, n = 8) (Figure 5a,b and c). Probenecid (2 × 10-3 M) significantly reduced the contraction response evoked by EFS in the PSA at 10 Hz and 20 Hz (P <0.001, Two-way ANOVA, n = 8) (Figure 5d). Carbenoxolone (10-4 M) did not significantly (P >0.05, Two-way ANOVA, n = 4) alter the contraction to EFS in PSA (Figure 5e).
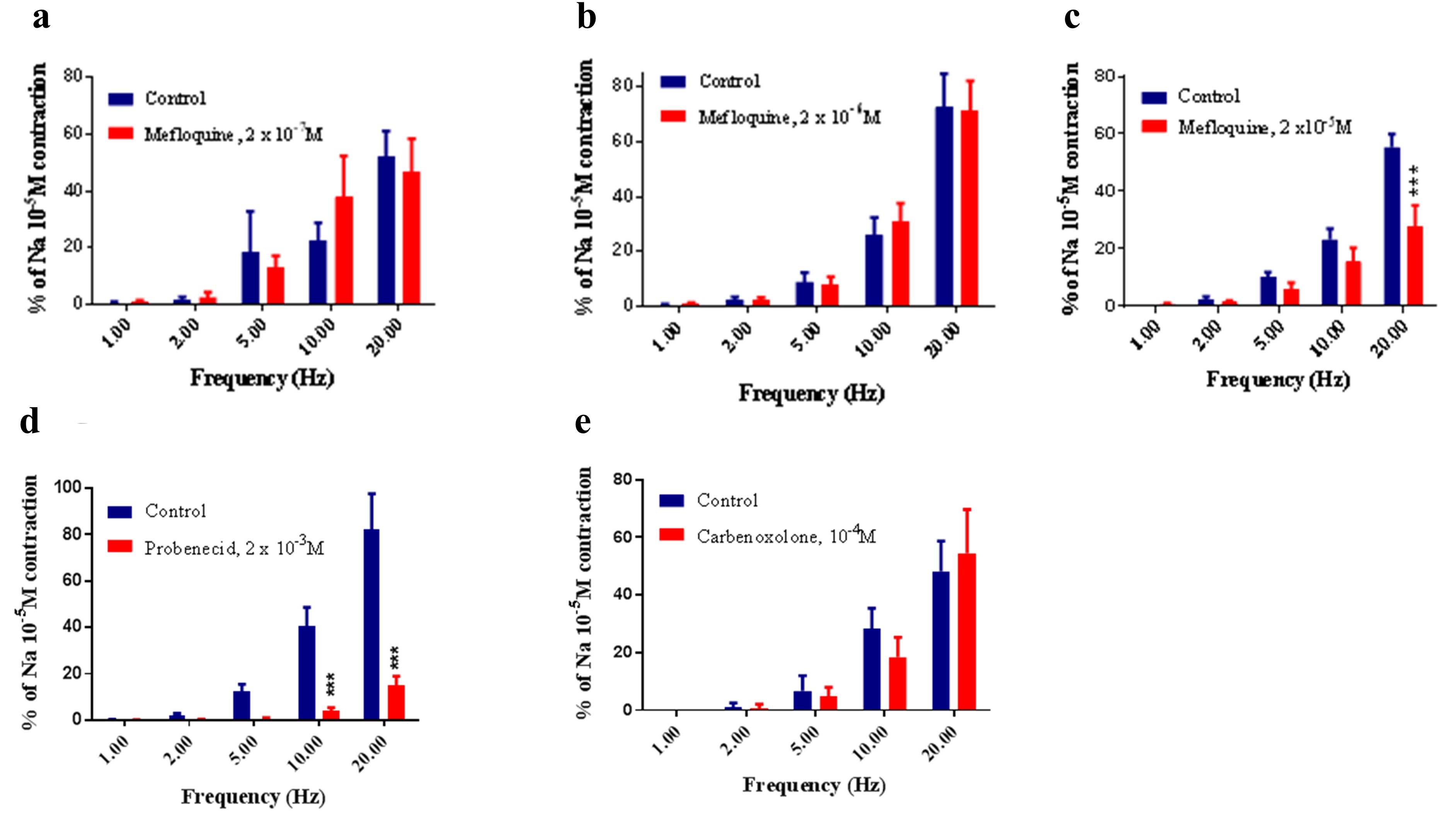
Figure 5. Effect of mefloquine ((a) 2x10 -7, (b) 2x10 -6, (c) 2x10-5M) on the response to EFS in the PSA. Responses to nerve stimulation were expressed as a percentage of the response produced by NA (10-5M). Mefloquine at 2x10-7 (a) and 2x10-6M (b) did not alter the contraction-induced by EFS (P > 0.05, Two-way ANOVA, N = 8). Mefloquine at 2x10-5M (c) significantly reduced responses to EFS in PSA at the highest frequency applied (20Hz). (d) Effect of probenecid (2x10-3M) on the response to EFS in the PSA. Probenecid decreased the contraction to EFS at 10Hz and 20 Hz (***P < 0.001, Two-way ANOVA, N = 8). (e) Effect of carbenoxolone (10-4M) on the response to EFS in the PSA. Carbenoxolone had no significant effect on the contraction to EFS. Each column represents the mean ± s.e.m. (***P < 0.0001, Two-way ANOVA, N = 8).
Effect of P2 purinoceptor agonists on responses to EFS in PSA
Suramin (3 × 10-4 M) had no significant effect on the contraction to ESF in PSA (P >0.05, Two-way ANOVA, n = 5) (Figure 6a). Similarity to suramin (3 × 10-4 M), PPADS (10-5 M) did not significantly alter the contraction to EFS in PSA (P >0.05, Two-way ANOVA, n = 8) (Figure 6b).
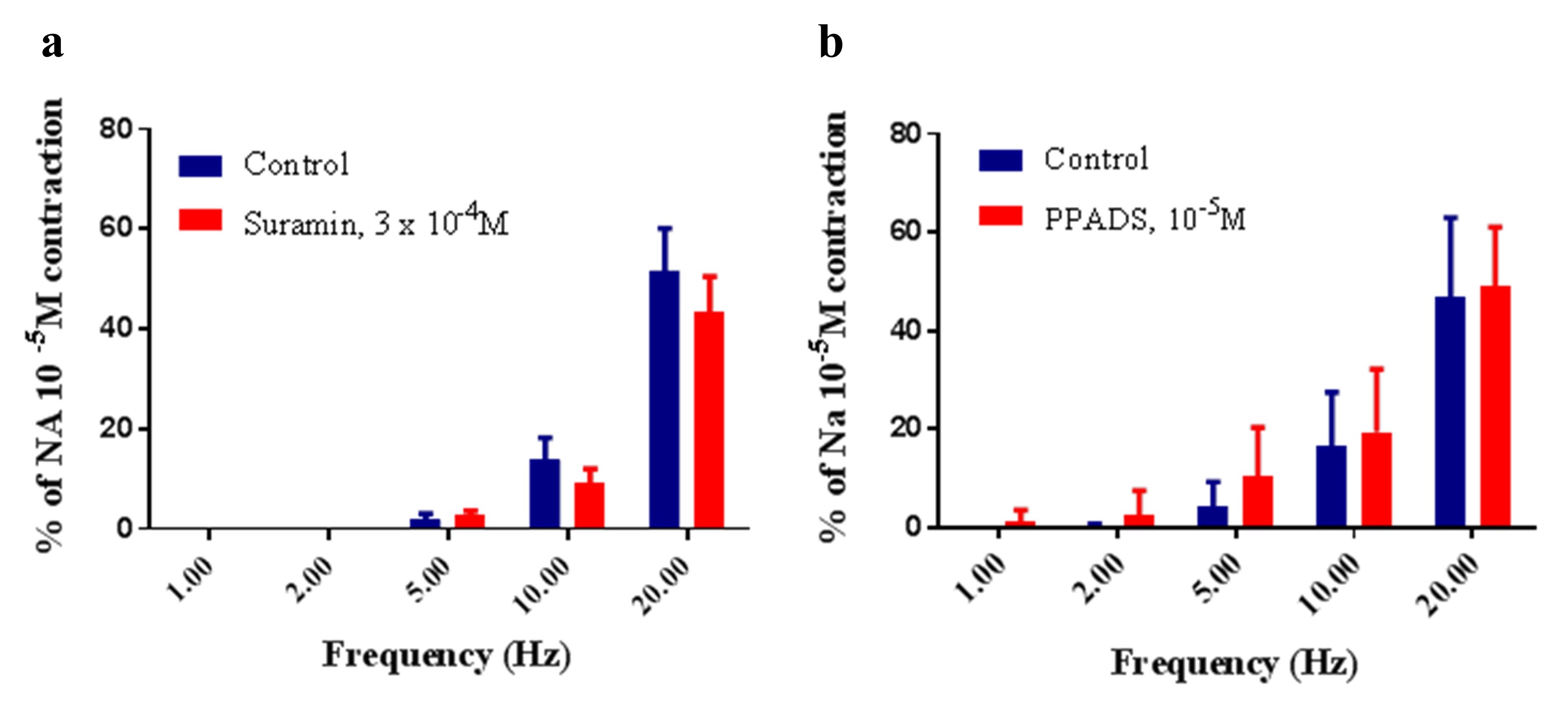
Figure 6. Effect of (a) suramin (3x10-4M) and (b) PPADS (10-5M) on the response to EFS in the PSA. Responses to nerve stimulation were expressed as a percentage of the response produced by NA (10-5M). Each column represents the mean ± s.e.m. (a) Suramin and (b) PPADS had no significant effect on the contraction to ESF (P > 0.05, Two-way ANOVA, N = 5-8).
Effect of mefloquine to 5-HT and U46619 in the PSA
In order to investigate the specificity of mefloquine, responses to 5-HT and U46619 were obtained in the absence and presence of mefloquine (2 × 10-5 M). Mefloquine significantly reduced the maximum response to both 5-HT and U46619 in the PSA. The maximum recorded responses to 5-HT were 125.78% ± 28.1% and 18.86% ± 8.47% in the absence and presence of mefloquine respectively (P <0.001, Student’s paired t-test; n = 4) (Figure 7a). The maximum responses to U46619 were reduced from 117.7% ± 9.057% in the control to 125.7% ± 6.2% in the presence of mefloquine (P <0.001, Student t-test, n = 4) (Figure 7b).
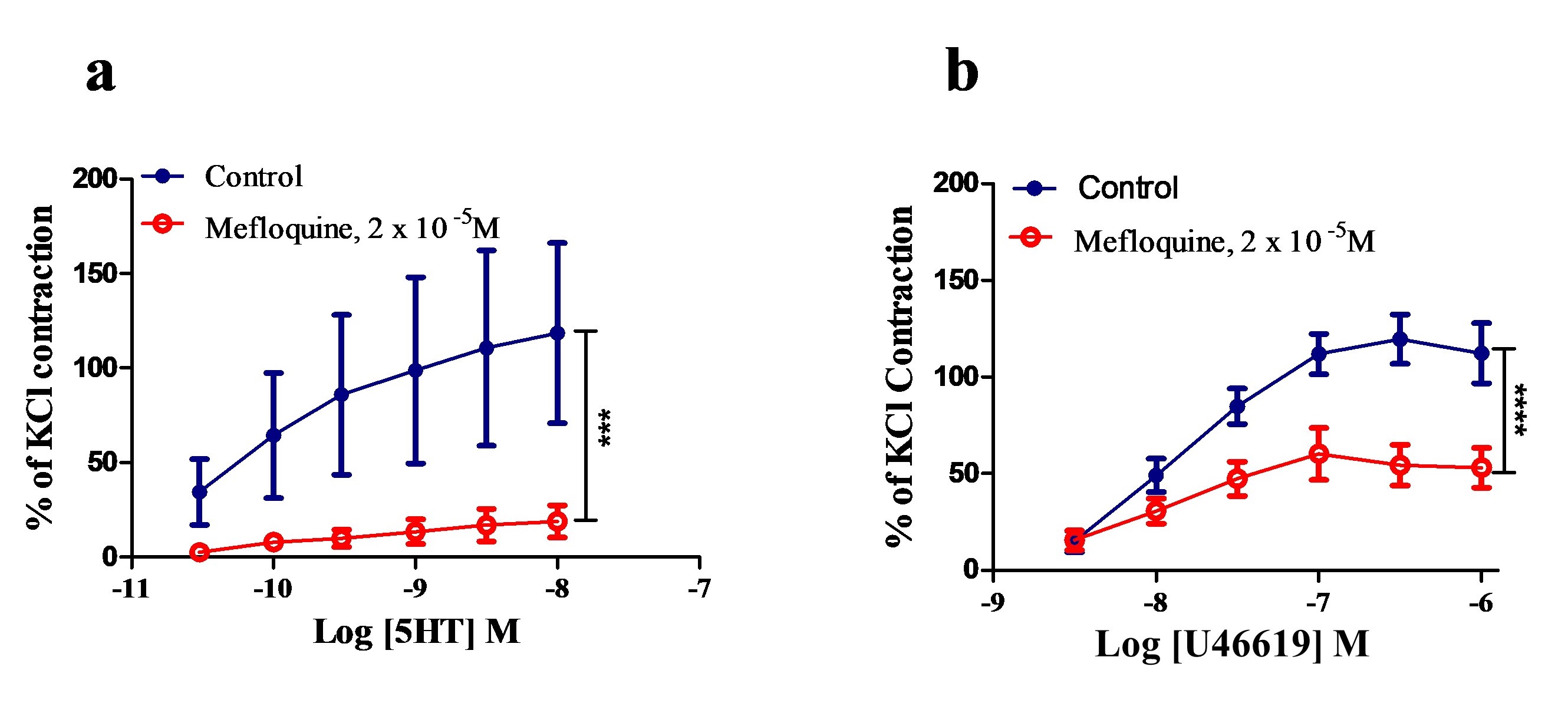
Figure 7. Effect of mefloquine (2x10-5M) on the responses to 5-HT (a) and U46619 (b) in the PSA. The contractions caused by 5-HT and U46619 are represented as a percentage of the contractions caused by KCl (60mM). Data are presented as the mean value ± s.e.m. Differences between Rmax values are shown by ***P < 0.001, (Student’s paired t-test, N = 4).
Discussion
Expression of pannexin in PSA
The initial aim of this work was to determine whether pannexins were present in the porcine splenic artery (PSA) and to examine their possible function. Western blot analysis was carried out using anti-pannexin-1 and anti-pannexin-2 antibodies. Both pannexin-1 and pannexin-2 expression were present in PSA tissues. Both pannexin-1 and pannexin-2 were detected in PSA at 50 kDa and 100 kDa respectively. The primary antibody used was rabbit anti-pannexin antibody while the goat anti-rabbit was used as a secondary antibody. Burns et al. (2012) have shown the presence of pannexin-1, pannexin-2 and pannexin-3 in rat middle cerebral artery (Burns et al., 2012). Immunolabelling of pannexin-1 in PSA showed pannexin-1 expression in both endothelial and smooth muscle cells. This was in agreement with previous studies carried by (Billaud et al., 2011; Lohman et al., 2012), who showed pannexin-1 to be observed in both the endothelium and smooth muscle of smaller arteries and arterioles in rat aorta and femoral artery. This was also in agreement with Burns et al et al. (2012) who showed pannexin-1 and pannexin-2 to be present in the smooth muscle cells of the rat middle cerebral artery (Burns et al., 2012). Pannexin-1 expression has also been reported in human erythrocytes (Locovei et al., 2006), although a recent effort to detect the protein in these cells with different antibodies was unsuccessful (Melhorn et al., 2013). Pannexin-3 has been previously reported in small arteries of less than 100μm in diameter, particularly in the kidney (Lohman et al., 2012).
Effect of pannexin inhibition on responses to NA and EFS in the PSA
The effect of various pannexin inhibitors (mefloquine, probenecid and carbenoxolone) on NA induced contraction as well as responses to EFS in the PSA were investigated. At the concentrations used in this study, both mefloquine and probenecid significantly reduced the response of PSA to both (i) NA-induced contractions and (ii) frequency-dependant response curves caused by activating the sympathetic nerves, but just when higher frequencies were applied i.e. 10 and 20Hz. Such data, in addition to the evidence provided on the expression of pannexin in the PSA, could suggest that pannexin channels are involved in conducting responses to NA in the PSA blood vessels. This was similar to the observations reported by Billaud et al. (2011) in thoracodorsal resistance arteries (TDA) from mice where 10μM and 20μM of mefloquine and 500μM of probenecid produced a considerable inhibitory effect on the response of TDA of mice to phenylephrine (a selective α1–adrenoreceptor agonist).
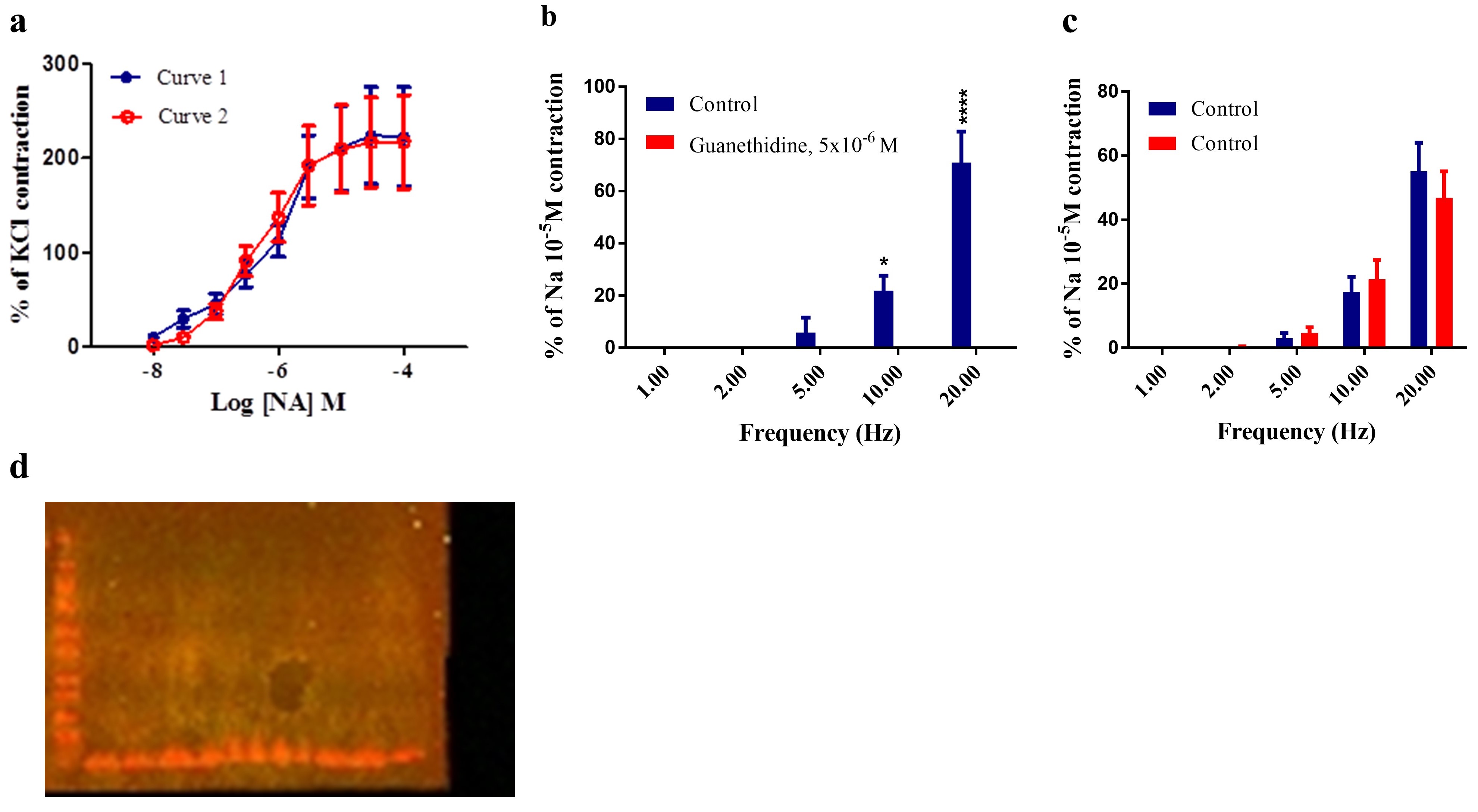
Supplementary Figure 1 (a) Responses to NA in the PSA. (b) Electrical field stimulation responses to 5 different frequencies (1, 2, 5, 10, 20Hz, 90 V, 5ms, 10s) were expressed as a percentage of the response produced by NA (10-5M). Each column represents the mean ± s.e.m. Contractile responses were abolished in the presence of guanethidine (5x10-6) (*P < 0.05, ****P < 0.0001, Two-way ANOVA, N = 4). (c) EFS responses to 5 different frequencies (1, 2, 5, 10, 20Hz, 90 V, 5ms, 10s), the contraction was expressed as a percentage of the response produced by NA (10 -5M). Each Colum represents the mean ± s.e.m. There was no significant difference between the responses (P > 0.05, Two-way ANOVA, N = 6). (d) Western blot analysis of porcine splenic artery in the absence of any primary antibody.
Billaud et al. (2011) suggested that pannexin-1 contributes to α1-adrenoreceptor-mediated vasoconstriction in thoracodorsal resistance arteries isolated from mice Billaud et al. (2011). Both pannexin-1 inhibitors (i.e. mefloquine and probenecid) and purinergic receptor antagonists (i.e. suramin and reactive blue-2) were shown to reduce PE-induced vasoconstriction. Therefore, it has been speculated that PE binds to the α1D-adrenoreceptor, causing pannexin-1 to open to release ATP, which then acts as an intercellular messenger to cause P2 receptor-mediated contractions. However, our study provided an evidence against a role for ATP released from inside cells via pannexin channels in the PSA due to the lack of significant effect of carbenoxolone (100µM), one of the first agents shown to directly inhibit pannexin-1 currents (Bruzzone et al., 2005), on the responses to neither NA exogenous nor nerve activation in the PSA. Carbenoxolone has been shown to interact with pannexins. For example, Sridharan et al. (2010) showed carbenoxolone (100µM) to inhibit the hypnotically induced ATP release from human erythrocytes (Sridharan et al., 2010). Similarly, Angus et al. (2015) showed that carbenoxolone had no effect on agonist or nerve-induced activation of alpha- adrenoceptors in rat small mesenteric resistance arteries (Angus et al., 2015).
The effects of two P2 purinoceptor antagonists (suramin and PPAD) on NA induced contraction as well as the responses to EFS in PSA were investigated. Suramin (100 µM to 300 µM) has been shown to inhibit P2 receptors, leading to decreased contractility of TDA induced by phenylephrine in rate (Billaud et al., 2011). However, in the current study, neither suramin (100 µM) nor PPADS (10 µM) had significant effects on responses to neither exogenous NA not nerve simulation in the PSA, providing further evidence against a role for ATP-activation of P2 receptors in transducing an α1-adrenoceptor-mediated response in the PSA. Additionally, neither P2 receptor antagonists affected responses to nerve stimulation conforming that ATP is not a functional sympathetic neurotransmitter in this preparation, in contrast to some other blood vessels including rat mesenteric arteries (Pakdeechote et al., 2007; Rummery et al., 2007) and porcine mesenteric arteries (Shatarat et al., 2014), although in the latter preparation, some pharmacological manipulation was required before a nerve-mediated purinergic response was observed.
The present data do not support the hypothesis proposed by (Billaud et al., 2011), who suggested alpha-adrenoceptor stimulation involved the release of ATP through pannexin channels to invoke a multicellular contraction by activating P2 purinoceptors. There are justifiable grounds to question whether mefloquine is acting selectively, which could provide an explanation for the apparent disagreement on the involvement of ATP in α1-adrenoreceptor-mediated vasoconstriction. The present study showed mefloquine to inhibit responses to both 5-HT and U46619 (a thromboxane mimetic), indicating that mefloquine is not a selective pannexin inhibitor at the concentrations used in this study. Such argument was supported by a recent study by Angus et al. (2015), who recently showed mefloquine (10μM to 20 μM) to demonstrate a wide range of inhibitory actions (Angus et al., 2015). For example, mefloquine (3μM to 10μM) inhibited the contractions mediated by vasopressin, phenylephrine, endothelin-1, U46619, sympathetic nerve stimulation and K+ (40 mM) in rat and mouse small mesenteric arteries. It is possible that pannexins are involved in transducing responses to any G-protein coupled receptor. For example, mefloquine (10μM) has been shown to inhibit L-type Ca2+ currents in single ventricular myocytes (Coker et al., 2000). Although the mechanism by which mefloquine and probenecid bind to pannexin channels remain speculative, one theory proposed that probenecid (a blocker of organic anion transporters) may either access pannexin channels through the lipid bilayer or interact either with hydrophilic aspects of the protein (Silverman et al., 2008). Probenecid is known to inhibit pannexin-1 channels with an IC50 of ~150μM, whereas it does not influence connexin-based channels regardless of its concentration (Silverman et al., 2008).
The current data show the first demonstration of the expression of pannexin-1 and pannexin-2 receptors in the muscle and endothelial cells of PSA. Pannexin-1 channels may contribute to α1-adrenoreceptor-evoked vasoconstriction in PSA, as the inhibition of pannexin leads to a reduction in the contraction of NA in PSA. However, as mefloquine was shown to be not selective in our model our data must be taken with caution on the basis of a lack of selectivity of the pharmacological agents currently available as pannexin inhibitors. On the other hand, our data argue against the involvement of ATP-activation of P2 receptors in NA-induced contraction in PSA. Future studies should be carried out to examine whether pannexins involve in mediating responses to NA in a preparation that contained α1D-adrenoceptors, namely the rat thoracic aorta.
References
Ambrosi C, Gassmann O, Pranskevich JN, Boassa D, Smock A, Wang J, Dahl G, Steinem C, Sosinsky GE. 2010. Pannexin-1 and Pannexin-2 channels show quaternary similarities to connexons and different oligomerization numbers from each other. The Journal of biological chemistry 285(32): 24420-24431.
Angus JA, Betrie AH, Wright CE. 2015. Pannexin-1 channels do not regulate α1-adrenoceptor-mediated vasoconstriction in resistance arteries. European Journal of Pharmacology 750:43-51.
Baranova A, Ivanov D, Petrash N, Pestova A, Skoblov M, Kelmanson I, Shagin D, Nazarenko S, Geraymovych E, Litvin O, Tiunova A, Born TL, Usman N, Staroverov D, Lukyanov S, Panchin Y. 2004. The mammalian pannexin family is homologous to the invertebrate innexin gap junction proteins. Genomics 83(4):706-716.
Billaud M, Lohman AW, Straub AC, Looft-Wilson R, Johnstone SR, Araj CA, Best AK, Chekeni FB, Ravichandran KS, Penuela S, Laird DW, Isakson BE. 2011. Pannexin-1 regulates α1-adrenergic receptor-mediated vasoconstriction. Circulation Research 109(1):80-85.
Bond SR, Lau A, Penuela S, Sampaio AV, Underhill TM, Laird DW, Naus CC. 2011. Pannexin-3 is a novel target for Runx2, expressed by osteoblasts and mature growth plate chondrocytes. Journal of Bone and Mineral Research 26(12):2911-2922.
Bond SR, Naus CC. The pannexins: past and present. Frontiers in physiology 2014; 5: p. 58-58.
Bruzzone R, Barbe MT, Jakob NJ, Monyer H. 2005. Pharmacological properties of homomeric and heteromeric pannexin hemichannels expressed in Xenopus oocytes. Journal of Neurochemistry 92(5):1033-1043.
Burns AR, Phillips SC, Sokoya EM. 2012. Pannexin protein expression in the rat middle cerebral artery. Journal of Vascular Research 49(2):101-110.
Cea LA, Riquelme MA, Vargas AA, Urrutia C, Sáez JC. 2014. Pannexin-1 channels in skeletal muscles. Frontiers in Physiology 5(139).
Coker SJ, Batey AJ, Lightbown ID, Díaz ME, Eisner DA. 2000. Effects of mefloquine on cardiac contractility and electrical activity in vivo, in isolated cardiac preparations, and in single ventricular myocytes. British Journal of Pharmacology 129(2):323-330.
Dvoriantchikova G, Ivanov D, Panchin Y, Shestopalov VI. 2006. Expression of pannexin family of proteins in the retina. FEBS Letters 580(9):2178-2182.
Ishikawa M, Iwamoto T, Nakamura T, Doyle A, Fukumoto S, Yamada Y. 2011. Pannexin-3 functions as an ER Ca2+ channel, hemichannel, and gap junction to promote osteoblast differentiation. The Journal of cell biology 193(7):1257-1274.
Langlois S, Xiang X, Young K, Cowan BJ, Penuela S, Cowan KN. 2014. Pannexin-1 and pannexin-3 channels regulate skeletal muscle myoblast proliferation and differentiation. The Journal of Biological Chemistry 289(44): 30717-30731.
Locovei S, Wang J, Dahl G. 2006. Activation of pannexin-1 channels by ATP through P2Y receptors and by cytoplasmic calcium. FEBS Letters 580(1):239-244.
Lohman AW, Billaud M, Straub AC, Johnstone SR, Best AK, Lee M, Barr K, Penuela S, Laird DW, Isakson BE. 2012. Expression of pannexin isoforms in the systemic murine arterial network. Journal of Vascular Research 49(5):405-416.
Makarenkova HP, Shah SB, Shestopalov VI. 2018. The two faces of pannexins: new roles in inflammation and repair. Inflammation Research Journal 11: p. 273-288.
Melhorn MI, Brodsky AS, Estanislau J, Khoory JA, Illigens B, Hamachi I, Kurishita Y, Fraser AD, Nicholson-Weller A, Dolmatova E, Duffy HS, Ghiran IC. 2013. CR1-mediated ATP release by human red blood cells promotes CR1 clustering and modulates the immune transfer process. The Journal of Biological Chemistry 288(43):31139-31153.
Molica F, Figueroa XF, Kwak BR, Isakson BE, Gibbins JM. 2018. Connexins and Pannexins in Vascular Function and Disease. International Journal of Molecular Sciences 19(6): 1663.
Ohbuchi T, Yokoyama T, Saito T, Ohkubo J, Suzuki H, Ishikura T, Katoh A, Fujihara H, Hashimoto H, Suzuki H, Ueta Y. 2011. Possible contribution of pannexin channel to ATP-induced currents in vitro in vasopressin neurons isolated from the rat supraoptic nucleus. Brain Research 1394:71-78.
Pakdeechote P, Rummery NM, Ralevic V, Dunn WR. 2007. Raised tone reveals purinergic-mediated responses to sympathetic nerve stimulation in the rat perfused mesenteric vascular bed. European Journal of Pharmacology 563(1):180-186.
Panchin Y, Kelmanson I, Matz M, Lukyanov K, Usman N, Lukyanov S. 2000. A ubiquitous family of putative gap junction molecules. Current Biology 10(13): p. R473-R474.
Poornima V, Madhupriya M, Kootar S, Sujatha G, Kumar A, Bera AK. 2012. P2X7 Receptor–Pannexin-1 Hemichannel Association: Effect of extracellular calcium on membrane permeabilization. Journal of Molecular Neuroscience 46(3):585-594.
Ray A, Zoidl G, Wahle P, Dermietzel R. 2006. Pannexin expression in the cerebellum. The Cerebellum 5(3):189-192.
Rummery NM, Brock JA, Pakdeechote P, Ralevic V, Dunn WR. 2007. ATP is the predominant sympathetic neurotransmitter in rat mesenteric arteries at high pressure. The Journal of Physiology 582(Pt 2):745-754.
Shatarat A, Dunn WR, Ralevic V. 2014. Raised tone reveals ATP as a sympathetic neurotransmitter in the porcine mesenteric arterial bed. Purinergic signalling 10(4):639-649.
Shestopalov VI, Panchin Y. 2008. Pannexins and gap junction protein diversity. Cellular and Molecular Life Sciences 65(3):376-394.
Silverman W, Locovei S, Dahl G. 2008. Probenecid a gout remedy, inhibits pannexin-1 channels. American Journal of Physiology Cell physiology 295(3):C761-C767.
Sorge RE, Trang T, Dorfman R, Smith SB, Beggs S, Ritchie J, Austin JS, Zaykin DV, Vander Meulen H, Costigan M, Herbert TA, Yarkoni-Abitbul M, Tichauer D, Livneh J, Gershon E, Zheng M, Tan K, John SL, Slade GD, Jordan J, Woolf CJ, Peltz G, Maixner W, Diatchenko L, Seltzer ZE, Salter MW, Mogil JS. 2012. Genetically determined P2X7 receptor pore formation regulates variability in chronic pain sensitivity. Nature medicine 18(4):595-599.
Sridharan M, Adderley SP, Bowles EA, Egan TM, Stephenson AH, Ellsworth ML, Sprague RS. 2010. Pannexin-1 is the conduit for low oxygen tension-induced ATP release from human erythrocytes. American Journal of Physiology. Heart and circulatory physiology 299(4):H1146-H1152.
Sumi Y, Woehrle T, Chen Y, Yao Y, Li A, Junger WG. 2010. Adrenergic receptor activation involves ATP release and feedback through purinergic receptors. American Journal of Physiology Cell Physiology 299(5):C1118-C1126.
Turmel P, Dufresne J, Hermo L, Smith CE, Penuela S, Laird DW, Cyr DG. 2011. Characterization of pannexin-1 and pannexin-3 and their regulation by androgens in the male reproductive tract of the adult rat. Molecular Reproduction and Development 78(2):124-138.
Vanden Abeele F, Bidaux G, Gordienko D, Beck B, Panchin YV, Baranova AV, Ivanov DV, Skryma R, Prevarskaya N. 2006. Functional implications of calcium permeability of the channel formed by pannexin-1. The Journal of Cell Biology 174(4):535-546.