

Sumanta Mondal1*, Debjit Ghosh2, Seru Ganapaty1, M. Sushrutha1
1GITAM Institute of Pharmacy, GITAM University, Visakhapatnam-530045, Andhra Pradesh, India
2Department of Chemistry, GITAM Institute of Science, GITAM University, Visakhapatnam-530045, Andhra Pradesh, India
*For correspondence:
Dr. Sumanta Mondal
Assistant Professor
GITAM Institute of Pharmacy, GITAM University,
Visakhapatnam-530045, Andhra Pradesh, India.
Mob: +91-9703615761
Abstract
Objectives: The present study deals with the preliminary phytochemical analysis of ethanol extract of Eclipta alba leaves (EEEAL) and also study the effect of the extract on hair growth stimulating activity on Wistar albino rats. Materials and methods: Preliminary phytochemical test were performed according to standard methods to detect the possible presence of different phytochemical groups in the extract. Investigation of hair growth stimulating potential were performed on Wistar albino rats. Primary skin irritation test were performed to detect any possible abnormal effects after shaving the dorsal hairs of the rats. The rats were then divided into three groups with six animals (rats) each where group I received water (control), group II was applied with 2% minoxidil (positive control) and group III was treated with 10% ethanol extract of Eclipta alba leaves. The average length of the hair was also recorded on 10th day, 20th day and 30th day. Results: Preliminary phytochemical screening of the extract revealed the presence of alkaloids, sterols, carbohydrates, saponins, flavonoids, tannins, and triterpenoids. Significant hair growth stimulating activity was revealed in the extract when compared with the control group. Conclusion: The ethanol extract of Eclipta alba leaves possesses potent hair growth stimulating activity which confirms its folkloric use as a hair promoting agent. Thus it can also be used as a potential hair stimulating agent.
Keywords: Eclipta alba L.; Hair growth stimulating activity; Minoxidil; Phytochemical analysis; Primary skin irritation test
Introduction
Herbal medicine is still considered the primary form of the health care mainly in the developing countries which is about 75 - 80% of the world population. There is a general faith that herbal drugs does not possesses any dangerous adverse effects besides being inexpensive and are available easily. Plants have been in use for the treatment of various diseases and disorders since human history and forms the basis of current sophisticated medicine. Many effective drugs have been originated from plant sources such as morphine (Papaver somniferum), aspirin (willow bark), digoxin (Digitalis sp.), and quinine (cinchona bark) (Garg et al., 2016; Tirtha, 1998; Tyler, 1999; Winslow and Kroll, 1998).
Eclipta alba L. also known as Bringaraja and False daisy, is a small branched annual herb which belongs to Asteraceae family (Figure 1). It is commonly found in tropical as well as subtropical areas of the world. The juice extracted from the leaf if taken internally or applied on the scalp it results in blackening of hair (Chopra et al., 1955; Kritikar and Basu, 1975). Several phyto-constituents has been reported from Eclipta alba such as alkaloids, coumestans, glycosides, flavonoids, triterpenoids and polyacetylenes. Wedelolactone,
a-terthienylmethanol, stigmasterol, dimethyl wedelolactone-7-glucoside and dimethyl wedelolactone have been detected from leaves. The roots contain heptacosanol and hentriacontanol. The aerial part is also reported to contain several phyto-compounds such as phytosterol, P-glucoside of phytosterol, P-amyrin, luteolin-7-glucoside, a glucoside of a triterpenic acid and wedelolactone. The seeds contains sterols, and Ecliptalbine (Wagner, 1986; Mithun et al., 2011; Treadway, 1998).
Hair loss is considered as a common emotional and distressing disease in humans, and It is considered that hair loss in both women and men are caused by nutritional deficiency, diseases, aging, stress and hormonal imbalance (Finn et al., 2006; Naito et al., 2008). Currently, there has been a sufficient increase in number of people suffering from alopecia or hair loss diseases (Paik et al., 2001). Hair is a pigmented filament or mini organ composed of dead and terminally differentiated keratinocytes and which grows from follicles present on the skin of humans or other animals. It plays an important role in physical protection with thermo and sensory regulation. Hair growth is a cyclical process which is divided into three phases viz., anagen (growth phase), catagen (regression phase), and telogen (resting phase) (Tiede et al., 2007). Impairment of the hair growth cycle is also connected with the development of certain conditions, such as androgenetic or pattern alopecia. There are two important hair follicle growth regulators viz., β-catenin and Sonic hedgehog (Shh), which are responsible for induction of the transition from telogen to anagen phase, and when there is a decreased level of either protein, severe damage takes place in the growth of hair (Wang et al., 2008; Oro and Higgins, 2003). Thus, the present study deals with the scientific confirmation of the folkloric use of E. alba L. as a hair growth promoter.
Materials and methods
Chemicals and reagents
Veet hair removal cream was purchased commercially (Reckitt Benckiser). Minoxidil which was used as the reference standard were obtained from Dr. Reddy’s, (Hyderabad, India). Ethanol (purity 99.9%) used for the extraction was obtained from Changshu Hongsheng Fine Chemicals Co., Ltd. (China). All other solvents, reagents and chemicals used for the analysis and activity studies were of analytical grade.
Collection and authentication of plant materials
The fresh leaves of Eclipta alba (Linn.) were collected in the month of September, 2012 from matured plants. The plant materials were then authenticated by Dr. M. Venkaiah, Professor (Retd.), Department of Botany, Andhra University, Visakhapatnam, Andhra Pradesh, India. A voucher specimen of the plant has been deposited in our research laboratory for future reference.
 Figure 1. Voucher specimen of Eclipta alba (L.)
Figure 1. Voucher specimen of Eclipta alba (L.)
Preparation of extracts
The collected leaves were gently washed in tap water to remove dirt and then they were shade dried in the laboratory under room temperature (24±2°C) for 3-4 weeks. After complete drying of the leaves they were grinded by using a mechanical grinder and then followed by sieving to obtain a coarse powder. Firstly, about 500 g of the coarsely powdered plant material were defatted using petroleum ether (60-80°C) in a soxhlet extractor. The following marc was then air dried for complete evaporation of petroleum ether and then extracted with ethanol (90%). The excess solvents were removed using rotary vacuum evaporator (Evator, Media Instrument Mfg. Co., Mumbai, India) and concentrated by heating the extract using a hot plate to obtain a dark greenish residue. The percentage yield of the crude extract was calculated according to the following formula.
Preliminary phytochemical tests
Preliminary phytochemical analysis of the ethanol extract of Eclipta alba leaves (EEEAL) were carried out for the detection of various phytoconstituents like alkaloids, carbohydrate, proteins, tannins, sterols, triterpenoids, saponins, and flavonoids (Harborne, 1984; Balachandran et al., 2016).
Test for alkaloids
The dry crude extract was dissolved in 2 N Hydrochloric acid. The following mixture was filtered and the resultant filtrate was divided into three equal portions. Mayer’s test: The first test portion was treated with a few drops of Mayer’s reagent, appearance of buff coloured precipitate proves the presence of alkaloids in the extract. Dragondorff’s test: Few drops of Dragondorff’s reagent was put in to the second portion where appearance of orange brown precipitate confirms that alkaloids are present in the extract. Wagner’s test: Few drops of Wagner’s reagent was mixed with the third portion of the test extract, formation of a reddish brown precipitate indicates the presence of respective alkaloids.
Test for carbohydrates
The test extracts were divided into three portions. Molisch’s test: To the first portion of the test extract, 10% alcoholic solution of a-napthol were added. The resultant mixture was shaken and few drops of Conc. H2SO4 were added along the side of the test tube. Appearance of a violet coloured ring at the junction of the two liquids confirms the presence of carbohydrates in the test extract. Fehling’s test: The second portion was treated with 2 ml of Fehling’s solution A and 2 ml of Fehling’s solution B and boiled. Formation of brick red precipitate confirms presence of reducing sugars. Benedict’s test: The third portion was treated with 5 ml of Benedict’s reagent and boiled on a water bath. Formation of brick red precipitate at the bottom of the test tube shows the presence of monosaccharide.
Test for proteins and amino acids
The test extracts were divided into four portions and kept in a test tube. Biuret test: The first portion was treated with 2 ml of 10% NaOH solution and then 2 to 3 drops of 1% CuSO4 solution were added. The following solution was mixed thoroughly, appearance of purple or violet colouration in the solution indicates the presence of proteins. Ninhydrin test: The second portion was treated with 0.5 ml of ninhydrin solution and warmed for 2 mins and cooled. The presence of proteins in the test extract would be confirmed by the appearance of blue colour. Xanthoproteic test: To the third portion, 1 ml of concentrated nitric acid was added, then boiled and cooled. To the resultant mixture a solution of 40% sodium hydroxide was added drop by drop and then mixed thoroughly. Appearance of a coloured solution indicates presence of proteins. Millon’s test: The fourth portion was treated with 2 ml of Millon’s reagent, then warmed and cooled. To the mixture few drops of NaNO2 solution were added. Formation of red colour or precipitate indicates presence of proteins.
Test for tannins and phenolic compounds
Ferric chloride (FeCl3) test: The test extract was treated with 1% w/w solution of FeCl3. Formation of blue/green/brown colour indicates the presence of tannins and phenolic compounds in the test extract.
Test for steroids and sterols
The test extracts were divided into two portions and kept in test tubes. Liberman Burchard test: The first portion (2 ml test extract solution in chloroform) was treated with 3-4 drops of acetic anhydride and mixed well. 1 ml of Conc. H2SO4 was added along the side of the test tube. Formation of a reddish brown ring at the junction of the two layers confirms the presence of sterols and steroids. Salkowski’s test: The second portion (5 ml test extract solution in chloroform) was treated with an equal volume of Conc. H2SO4 was added gently along the sides of the test tube. The lower acid layer and the upper chloroform layer were observed. Appearance of a yellow colour with a green fluorescence in the lower acid layer and a play of colours first from bluish red to gradually violet red in the upper chloroform layer proves the presence of sterols and steroids.
Test for triterpenoids
Sulphuric acid test: About 5 ml of chloroform was added to 300 mg of extract and then the following mixture was boiled for 30 minutes. Then few drops of Conc. H2SO4 was added to the chloroform solution and mixed thoroughly. Formation of a red colour confirms the presence of triterpenes.
Test for saponins
Foam test: About 300 mg of the test extract was warmed with 5 ml of distilled water for about 2 minutes. Then the following mixture was cooled and shaken vigorously and left idle for few minutes. Appearance of foam on the upper layer of the test solution confirms the presence of saponins.
Test for flavonoids
The test extracts were divided into three portions and kept in a test tube. Shinoda test: To the first portion a piece of magnesium ribbon was added and 3-4 drops of Conc. HCl were poured along the side of the test tube. A pink/magenta colour develops which indicates presence of flavonoids. Ferric chloride test: Few drops of neutral FeCl3 solution was added to the second portion of the test extract solution. Appearance of a blackish green colour confirmes the presence of flavonoids. Lead acetate test: The third portion was treated with few drops of 10% lead acetate solution. Appearance of yellow precipitate proves the presence of flavonoids in extract.
Experimental animals and housing conditions
Healthy Wistar albino rats, both male and female weighing between 150–250 g and of approximately the same age were procured from the central animal house of the institute and was used for the activity study. All the animals were acclimatized for at least one week in the laboratory animal room prior to experimentation and maintained in clean polypropylene cages with optimum light, temperature and humidity (light/dark cycles (12/12h), Temp: 25 ± 2ºC, and 75% relative humidity) and fed with commercially pelleted rat diet (M/s Hindustan Lever Ltd., Mumbai) and water ad libitum.
Ethical approval
All the experimental procedures complied with the guide for the care and use of laboratory animals, and the permission to carry out the animal study and protocols related to the experiments were approved by the Institutional Animal Ethics Committee (IAEC) of GITAM Institute of Pharmacy, Visakhapatnam, Andhra Pradesh, India (Registration No.1287/ac/09/CPCSEA and Protocol No: IAEC/GIP-1287/Bpharm/IP/SM-NS/11/2012-13).
Hair growth stimulating activity
Primary skin irritation test
For the evaluation of primary skin irritation test, the dorsal hairs of the rats were shaved with the help of electrical shavers and hair clippers. Veet, a commercially available hair removal cream was used for the complete removal of hair from 3 cm2 area (Roy et al., 2007). Surgical spirit was used for the cleaning of the shaved dorsal area of the rats. After completion of the cleaning of the shaved area, 10% w/w EEEAL was applied and the rats were observed for 48 hrs post application for any possible toxic effects.
Experimental grouping and dosing for hair growth stimulating activity
Eighteen animals were divided into three groups with six animals each. Hairs from a 3 cm2 area were shaved from the dorsal portion of all rats using electric shavers and a commercial hair removal was used to completely remove hair from the skin. Group I received water (control), group II was applied with 2% minoxidil (positive control) and group III received 10% ethanol extract of Eclipta alba leaves. The drug was applied on the depilated area twice daily for 30 days and the pattern of hair growth was observed and recorded.
Hair length determination
Hair was randomly plucked using sterile forceps from the particular study area on the dorsal side of the rats on every ten days. Evaluation of the average length of hair was calculated taking 25 hairs. The completion of hair growth was considered based on the average length of hair. The growth of hair in the shaved area was observed and the time to cover the bald patch was noted (Suraj et al., 2009). The average length of the hair was recorded and noted on 10th day, 20th day and 30th day.
Statistical analysis
The results obtained from the experiments were subjected to one way of analysis of variance (ANOVA). The significance between groups was analyzed using Dunnet’s t-test. A p-value less than <0.05 were considered to be significant. All the values were expressed as
mean ± S.E.M. (n = 6 in each group).
Results
Percentage yield and phytochemical analysis of EEEAL
After complete drying the ethanol extract of Eclipta alba leaves (EEEAL) yielded about 10.5% w/w of extract. Preliminary phytochemical screening of EEEAL for detection of different secondary metabolites revealed the presence of alkaloids, carbohydrates, sterols, flavonoids, saponins, tannins, and triterpenoids in the test extract (Table 1).
Table 1. Preliminary phytochemical screening of ethanol extract of E. alba leaves (EEEAL)
|
S. No. |
Phytochemicals |
Tests performed |
Inference |
|
1. |
Alkaloids |
Mayer’s test |
+ |
|
Dragondorff’s test |
+ |
||
|
Wagner’s test |
+ |
||
|
2. |
Carbohydrates |
Molisch’s test |
+ |
|
Fehling’s test |
+ |
||
|
Benedict’s test |
+ |
||
|
3. |
Proteins and amino acids |
Biuret test |
- |
|
Ninhydrin test |
- |
||
|
Xanthoproteic test |
- |
||
|
Millon’s test |
- |
||
|
4. |
Tannins and phenolic compounds |
Ferric chloride test |
+ |
|
5. |
Steroids and sterols |
Liberman Burchard test |
+ |
|
Salkowski’s test |
+ |
||
|
6. |
Triterpenoids |
Sulphuric acid test |
+ |
|
7. |
Saponins |
Foam test |
+ |
|
8. |
Flavonoids |
Shinoda test |
+ |
|
Ferric chloride test |
+ |
||
|
Lead acetate test |
+ |
Effect of EEEAL on hair growth stimulating activity
No abnormal effects were seen on the shaved area of the rats in the primary skin irritation test (Figure 2). Growth of hair from the shaved dorsal area was observed and the number of days taken for the initiation of hair growth was noted. The time taken for complete hair growth was 19.01 ± 0.51 days and 16.05±0.41 days in EEEAL (10% w/w) and 2% Minoxidil treated groups, respectively which was significant (P<0.01) when compared with the control
(Figure 3). The increase in length of the hair from the denuded area were observed at the end of the 10th day, 20th day and 30th day where it was 8.91±0.03 mm, 16.01±0.01 mm and 21.08±0.03 mm respectively in EEEAL (10% w/w) treated group. In 2% Minoxidil treated group the increase in length of the hair from the denuded area on the 10th day, 20th day and 30th day were 9.23±0.01 mm, 17.63±0.02 mm and 22.13±0.04 mm, respectively. The increase in hair length in both the extract and reference standard treated groups were significant (P<0.05) when compared with the control group (Figure 4). Thus the ethanol extract of Eclipta alba leaves (EEEAL) possesses potent hair growth stimulating activities as evidenced by complete hair growth on the 30th day as shown in Figure 5.
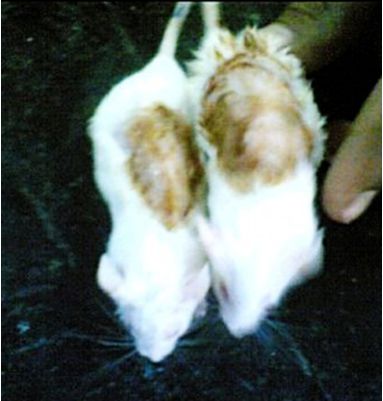
Figure 2. Dorsal side of rats shaved during evaluation of primary skin irritation test
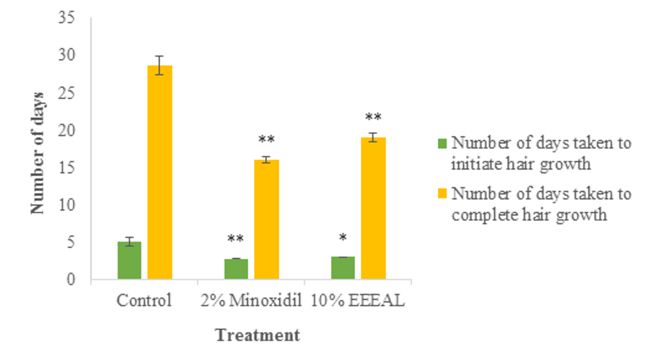
Figure 3. Qualitative observation of hair growth for ethanol extract of E. alba leaves (EEEAL). Values are expressed as mean ± S.E. (n = 6). All columns are significant using ANOVA. * P<0.05, ** P<0.01 when compared to control; Dunnet’s t-test.
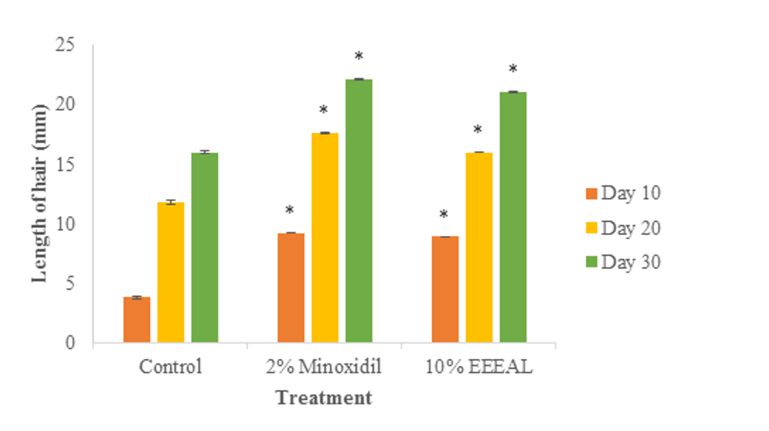
Figure 4. Effect of ethanol extract of E. alba leaves (EEEAL) on hair length of Wistar rats. Values are expressed as mean ± S.E. (n = 6). All columns are significant using ANOVA. * P<0.05, ** P<0.01 when compared to control; Dunnet’s t-test.
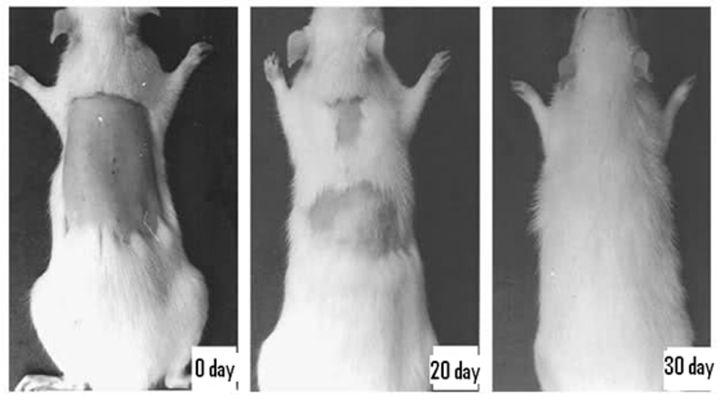
Figure 5. Photographs showing hair growth stimulating activity of ethanol extract of E. alba leaves (EEEAL) studied on Wistar albino rats taken on initial day, 20th day and 30th day
Discussion
Plant kingdom harbours an almost inexhaustible source of active phytoconstituents responsible for the treatment and management of various diseases. The preliminary phytochemical test are used to find and locate phyto constituents which are source of pharmacologically active principles (Sivagnanam and Chandra, 2016). In our study the ethanol extract of Eclipta alba leaves (EEEAL) revealed the presence of flavonoids, alkaloids, saponins, carbohydrates, tannins and triterpenoids which may be responsible for different pharmacological activities.
Hair loss is considered as one of the most common dermatological disorders spread throughout the world to human races and usually is of great concern for decades. Thus it is essential for identification of new hair growth stimulating agents as there are only few drugs such as minoxidil, finasteride etc., which are approved by the Food and Drug Administration (FDA) for the treatment of alopecia. Upjohn Company conducted a large randomized placebo controlled trials for Minoxidil, a potassium channel opener on humans which proved to be effective in 54% of the treated subjects as opposed to 34% in placebo control group. There are several adverse effects caused by Minoxidil such as dryness, scaling, pruritis, dermatitis and irritation (De Villez, 1990).
For the treatment of Androgenetic alopecia (male pattern baldness), Finasteride, an oral 5 alpha reductase inhibitor is used to promote hair growth. Merck research laboratories conducted large clinical trials (MRL clinical study report: 087-03, 089-03, 092, 1996) and reported that hair regrowth of 48% was observed in finasteride treated subjects in one year. Finasteride is usually well tolerated but few patients have withdrawn from the treatment due to sexual disorders related to the drug. The drug is not suggested for use in women (McClellan and Markham, 1999). Hair loss disorders are not considered life threatening, but according to American hair loss association hair loss is a very emotionally distressing disease which makes afflicted patients vulnerable (The Washington post, 2006).
Thus observing and studying these factors, development of novel drugs that revitalises the hair growth pattern and appearance with minimal adverse effects is important and so investigation of medicinal plants as a potential hair growth promoter was brought into the focus of the current research. In our study the ethanol extract of Eclipta alba leaves (EEEAL) was able to promote hair growth on the denuded dorsal area of albino rats and induce the anagen phase in telogen phase of hair follicles. This pattern of hair growth may be taking place due to the premature shifting of hair follicles from the telogen to anagen phase (Philpot et al., 1992). The prolonged anagen phase, conversion of short hairs to long terminal hairs, and follicular size enlargement is mainly due to the increased rate of proliferation of cells (Uno and Kurata, 1993). In treated groups, the transformation of hair follicles from telogen phase to anagen phase may be taking place due to the epithelial cell proliferation near the base of follicles which induce vasodilation of blood vessels in the scalp (Savin and Atton, 1993). Our study proved that the ethanol extract of Eclipta alba leaves (EEEAL) possesses hair growth stimulating activity as there was dense hair growth in the extract treated group.
Conclusion
In conclusion, our study showed that the ethanol extract of Eclipta alba leaves (EEEAL) possesses potent hair growth stimulating activity and thus can be used as a potential hair stimulating agent. This study can also be taken as a benchmark for further investigation on this plant to identify the chemical entity and characterization of the active constituent responsible for the hair growth stimulating activity. The mechanism of action of the extract or bioactive compounds on hair growth stimulating activity must also be studied to help improve more efficient and safe hair promoting drugs.
Acknowledgements
The authors are thankful to GITAM University, Visakhapatnam, Andhra Pradesh, India for providing facilities and support to organize and conduct this research.
Conflicts of interest
The authors declare that they have no conflicts of interest.
References
Balachandran P, Maroky AS, Ajay Kumar TV, Parthasarathy V. 2016. Preliminary phytochemical analysis of the ethanolic extract of brown Seaweed Sargassum wightii. International Journal of Research in Pharmaceutical Sciences, 7(2): 154-156.
Chopra RN, Nayar SL, Chopra IC. 1955. Glossary of Indian Medicinal plants C.S.I.R., New Delhi.
De Villez RL. 1990. The therapeutic use of topical minoxidil. Dermatologic Clinics, 8(2): 367–375.
Finn DA, Beadles-Bohling AS, Beckley EH, Ford MM, Gililland KR, Gorin-Meyer RE, Wiren KM. 2006. A new look at the 5 alpha-reductase inhibitor finasteride. CNS Drug Reviews, 12(1): 53-76.
Garg A, Shukla A, Pandey P, Dev S. 2016. Inhibitory effect of alcoholic extract of Tulsi (Ocimum sanctum) on calcium oxalate crystals: An in-vitro study. Asian Journal of Pharmacy and Pharmacology. 3(2): 77-80.
Harborne JB. 1984. Phytochemical Methods: A Guide to Modern techniques of Plant analysis. Chapman and Hall, New York.
Kritikar KR, Basu, BD. 1975. Chronica Botanica Indian Medicinal plants: NewDelhi.
McClellan KJ, Markham A. 1999. Finesteride. A review of its use in male pattern hair loss. Drugs, 57(1): 111–126.
Mithun NM, Shashidhara S, Kumar VR. 2011. Eclipta alba (L.) A Review on its Phytochemical and Pharmacological Profile. Pharmacologyonline, 1: 345-357.
MRL clinical study report, multicenter study: a double blind randomized placebo controlled, multicenter study to determine the effect of finasteride on hair loss inmenwith androgenetic alopecia. Merck Research Laboratories 1996: (protocol 087-03).
MRL clinical study report, multicenter study: a double blind randomized placebo controlled, multicenter study to determine the effect of finasteride on hair loss inmenwith androgenetic alopecia. Merck Research Laboratories 1996: (protocol 089-03).
MRL clinical study report, multicenter study: a double blind randomized placebo controlled, multicenter study to determine the effect of finasteride on hair loss inmenwith androgenetic alopecia. Merck Research Laboratories 1996: (protocol 092).
Naito A, Sato T, Matsumoto T, Takeyama K, Yoshino T, Kato S, Ohdera, M. 2008. Dihydrotestosterone inhibits murine hair growth via the androgen receptor. British Journal of Dermatology, 159(2): 300–305.
Oro AE, Higgins K. 2003. Hair cycle regulation of Hedgehog signal reception. Developmental Biology, 255(2): 238–248.
Paik JH, Yoon JB, Sim WY, Kim BS, Kim NI. 2001. The prevalence and types of androgenetic alopecia in Korean men and women. British Journal of Dermatology, 145(1): 95–99.
Philpot MP, Green MR, Kealey T. 1992. Rat hair follicle growth in vitro. British Journal of Dermatology, 127(6): 600-607.
Roy RK, Mayank T, Dixit VK. 2007. Effect of Citrullus coclocynthis Schrad. on hair growth activity of albino rats. Australian Journal of Medical Herbalism, 6(2): 108-112.
Savin RC, Atton AV. 1993. Minoxidil: update on its clinical role. Dermatologic Clinics, 11(1): 55-64.
Sivagnanam S, Chandra AP. 2016. Preliminary Phytochemical Analysis of Amarathus viridis. Research Journal of Pharmaceutical, Biological and Chemical Sciences, 7(1): 1-7.
Suraj R, Rejitha G, Anbu J, Promwichita P. 2009. In vivo hair growth activity of Prunus dulcis seeds in rats. Biology and Medicine, 1(4): 34-38.
The Washington post, 2006. American hair loss association.
Tiede S, Kloepper JE, Whiting DA, Paus R. 2007. The ‘follicular trochanter’: an epithelial compartment of the human hair follicle bulge region in need of further characterization. British Journal of Dermatology, 157(5): 1013–1016.
Tirtha SSS. 1998. Overview of Ayurveda. In the Ayurveda Encyclopedia: Natural Secrets to healing, prevention and longevity. Amrit Kaur Khalsa and Rob Paon Satyaguru Publications.
Treadway S. 1998. An ayurvedic herbal approach to a Healthy Liver. Clinical nutrition insights, 6(16): 1-3.
Tyler VE. 1999. Phytomedicine: Back to the Future. Journal of Natural Products, 62: 1589-1592.
Uno H, Kurata S. 1993. Chemical agents and peptides affect hair growth. Journal of Investigative Dermatology, 101: 143-147.
Wagner H. 1986. Coumestans as the main active principles of the liver drug Eclipta alba and Wedelia Calendulaceae. Planta Medica, 5: 370-374.
Wang X, Zhao L, Han T, Chen S, Wang J. 2008. Protective effects of 2,3,5,40-tetrahydroxystilbene-2-O-beta-d-glucoside, an active component of Polygonum multiflorum Thunb, on experimental colitis in mice. European Journal of Pharmacology, 578(2–3): 339–348.
Winslow LC, Kroll DJ. 1998. Herbs as medicine. Archives of Internal Medicine, 158: 21-92.