

Vinod Dhote1*, KanikaDhote2, Dinesh Kumar Mishra3
1Truba Institute of pharmacy, Bhopal (M.P)
2Ravishankar College of Pharmacy, Bhopal (M.P)
3College of Pharmacy, IPS Academy Indore-452012 (M.P)
*Corresponding author
Vinod Kumar Dhote
Asst. Prof. (Pharmaceutics)
Truba Institute of Pharmacy, Bhopal
Email: vinoddhote@gmail.com, dineshdops@gmail.com
Abstract
Objective: The objective of the current research is to reduce dosing frequency and improve patient compliance by designing and systematically evaluating controlled release microbeads of Rioprostil. Materials and methods: Rioprostil loaded mucoadhesive microbeads were successfully prepared by ionotropic gelation and crosslinking technique by using sodium alginate as the hydrophilic carrier in combination with HPMC and chitosan polymers as drug release modifiers. Formulated microbeads were evaluated for particle size, swelling ratio, drying rate, drug entrapment, bio adhesion study, invitro release, release kinetic and stability study. Particle size distribution of both placebo and drug loaded formulations were measured by an optical microscope and particle size of optimized beads was determined by scanning electron microscopy. Results: Results shows that no significant drug-polymer interactions were observed in FTIR studies. In-vitro drug release profile of Rioprostil microbeads were studied in phosphate buffer pH 6.8 and exhibited zero order kinetic. The in vitro wash-off test indicates microbeads had good mucoadhesive properties. The drug loaded batches were found to be stable when stored at room temperature for 45 days. Conclusion: Hence the formulated HPMC coated Sodium alginate beads can be used as an alternative and cheaper carrier for the oral controlled delivery of Rioprostil, especially for the treatment of ulcer colitis.
Keywords: Rioprostil, microbeads, ionotropic gelation, mucoadhesive, cross linking
Introduction
Controlled drug delivery technique presents front line part of today’s developed technique, in this includes many scientific approaches, serving for individual care (Petersen, 1999). The drug deliverance technique having abundant advantages than existing conventional type of dosage, it involves enhanced effectiveness, minimized poisoning, enhanced consumer conformity also ease (Pillay, 1999; Nagata, 1995). This type of drug deliverance technique utilizes micro molecules, for caring drugs. As the varieties of forms for dosage are invented like microparticle as well as nanoparticles shown more significance (Florence, 1997; Ulrich, 2005).
An ideal and advanced oral drug delivery system is that, which exactly controls speed, time as well as site of release of medicament separately of normal physiological variables such as gastrointestinal tract pH, digestive condition of the gastrointestinal tract, peristalsis movement and circadian rhythm. Advance in polymer science and technology outcome in pick up the pace research and developmental activity in the design of drug delivery devices (Roy, 1989; Dashevsky, 2004).
Rioprostil is a synthetic methylprostaglandin E1 analog that reduces gastric acid secretion and enhances the gastric mucus-bicarbonate barrier. It is effective in the therapy of gastric ulcers and gives significant protection against NSAID-induced gastric mucosal damage. The drug also prevents cyclosporin A-induced damage to endocrine and exocrine pancreatic secretions. It shows a low order of acute toxicity and there is no evidence of embryotoxicity, fetotoxicity, teratogenicity, or mutagenicity in animal studies (Lambert, 2000).
Various agents with different action mechanisms used to treat or ameliorate peptic ulcer or irritation of the gastrointestinal tract. This has included antibiotics to treat helicobacter infections; histamine H2 antagonists to reduce gastric acid secretion; and antacids for symptomatic relief.
The purpose of this research was to develop a controlled delivery system containing drug Rioprostil with different ratio of polymers. Hydrophilic polymers are widely used in the formulation of modified release oral dosage forms (Bechgaard, 1978).Their convenience and ease of manufacture may cut down the cost of the final product. Besides, hydrophilic polymer matrix system offers several additional advantages over other technologies for controlled release drug delivery (Barratt, 2000).
Materials and Methods
Reagents
Rioprostil was obtained as gift sample from Torrent Pharmaceutical private limited, Ahmedabad, India. HPMC was purchased from high media laboratory Pvt Ltd Mumbai. Sodium alginate, chitosan and calcium chloride were purchased from Colorcon, Goa, India. All ingredients used were of analytical grade.
Formulation preparation
The microbeads of Rioprostil were prepared by using ionotropic gelation technique. In this method weighed quantity of drug was dispersed uniformly in aqueous mucilage of sodium alginate using mechanical stirrer maintaining the speed at 500-600 rpm. To this dispersion the polymer (HPMC/Chitosan) was mixed in s proportions and the entire mixture was stirred for 30 min (Narkar, 2010; Millili, 1990). The microbeads were formed by dropping the bubble free dispersions through a glass syringe with flat tip needle (20G) into a gently agitated calcium chloride solution (Lim, 1997). The gelled microbeads were cured for 1 hour to complete the gelation reaction before being filtered and washed thoroughly with distilled water and isolated microbeads were dried at room temperature and then in hot air oven at 60°C for 6 h for further drying (Eldem, 1991).
Evaluation of placebo microbeads
Yield of production
The yields of the product for various batches were calculated using the weight of final product after drying respecting the initial total weight of the drug and polymer used for preparation of microbeads and percent production yields were calculated as per the formula mentioned below (Bansal, 1993; Narkar, 2010; Millili, 1990).
Percentage yield = (practical yield/theoretical yield) ×100
Measurement of bead size
The particle sizes of formulations were measured by an optical microscope fitted with an ocular and stage micrometer and particle size distribution was calculated. In all measurements at least 50 beads in five different fields were examined. Each experiment was carried out in triplicate (Ragnarsson, 1992).
Drying rate study of the beads
Prepared beads were placed in open glass bottles and kept in an incubator maintained at 50ºC. Initially, the beads were removed at short intervals of time (5, 10, and 15, up to obtain constant weight). These measurements were continued until attainment of constant mass and note down the temperature and time for the complete drying of the beads (Turkan, 1991).
Evaluation of swelling ratio
Swelling ratio was studied by measuring the percentage water uptake by the beads. About 50 mg of beads from all prepared placebo beads were accurately weighed and placed in 100 ml of phosphate buffer (pH 6.8 and 0.1 N HCl (pH 1.2). Beads were removed from their respective swelling media after 8 h and weighed after drying the surface water using filter paper. The water uptake was calculated as the ratio of the increase in weight of beads after swelling to the dry weight (Tomofumi, 2001; Eldem, 1991).
Swelling ratio = swollen w.t -_ initial w.t X 100
Initial w.t
Estimation of drug content and encapsulation efficiency
Rioprostil content in the microbeads was estimated by a UV-spectrophotometric method. Accurately weighed sample (50mg) of formulation were suspended n 100ml of phosphate buffer pH 6.8.The resulting solution was kept for 24hrs. Next day it was stirred for then solution was filtered and after suitable dilution, drug content in the filtrate was analyzed at 245 nm using Shimadzu 1600 UV-Visible spectrophotometer. The obtained absorbance was plotted on the standard curve to get the exact concentration of the drug. Calculating this concentration with dilution factor we get the percentage of actual drug content and entrapment efficiency. The drug entrapment efficiency was determined using following relationship (Bansal, 1993);
% Drug entrapment efficiency = (Actual drug content/ Theoretical drug content) X 100
Loose surface crystal study (LSC)
The study was conducted to estimate the amount of drug present on the surface of the microbeads which showed immediate release in dissolution media. 100mg of microbeads were suspended in 100ml of phosphate buffer (pH 6.8), simulating the dissolution media. The samples were shaken vigorously for 15min in a mechanical shaker. The amount of drug leached out from the surface was analyzed spectrophotometrically at 245nm. Percentage of drug released with respect to entrapped drug in the sample was recorded (Ragnarsson, 1992).
Scanning electron microscopy (SEM) analysis
The shape and surface characteristics were determined by scanning electron microscopy using gold sputter technique. The particles were Vacuum dried, coated to 200 Ao thicknesses with gold palladium using prior to microscopy. A working distance of 20nm, a tilt of zero-degree and accelerating voltage of 15kv were the operating parameters. Photographs were taken within a range of 50-500 magnifications (Narkar, 2010; Lim, 1997).
In-vitro release studies
In-vitro release studies of prepared microbeads were carried out using phosphate buffer (pH 6.8) using USP- basket type apparatus. Accurately weighed quantity of 250 mg of prepared microbeads put into the basket rotated at a constant speed at 100rpm and maintained temperature 37±5ºC in 900ml of the dissolution medium (phosphate buffer pH6.8).The sample was withdrawn at0.25hrs, 0.5hrs, 1hrs, 2hrs, 3hrs, 4hrs, 5hrs, 6hrs, 7hrs, 8hrs, 9hrs, 10hrs, 11hrs and 12hrs. Each time interval 5 ml of sample was withdrawn, at the same time 5 ml of fresh dissolution media was added to maintain sink condition. The withdrawn samples were suitably diluted and measure the absorbance at 245 nm spectrophotometrically. Then, calculated the cumulative percentage drug release at regular time intervals (Dressman, 2000).
Release kinetic study
In order to understand the mechanism and kinetics of drug release, the drug release data of the in-vitro dissolution study was analyzed with various kinetic equations like zero-order, first order and korsmeyer and Peppas equation. Coefficient of correlation (r) values were calculated for the linear curves obtained by regression analysis of the above plots (Dressman, 2000; Korsmeyer 1981).
Mucoadhesion testing by in vitro wash-off method
The mucoadhesive properties of drug loaded sodium alginate beads were evaluated by the in vitro wash-off method. Freshly excised pieces of goat intestinal mucosa (1 cm × 1 cm, collected from a slaughter house) were mounted on a glass slide (7.5 cm×2.5 cm) using thread. About 50 beads were spread out on each piece of mucosa and then hung from the arm of the tablet disintegration test apparatus. The tissue specimen was given a regular up and down movement in a vessel containing 900 ml of phosphate buffer (pH 6.8) maintained at 37 ± 0.5 °C. The adherence of beads was regularly observed. The beads that remained adhered to the mucosa were counted at regular intervals for up to 6h (Lim, 1997).
% Mucoadhesion = (Number of adhered microbeads/ Total number of applied microbeads) x100
Results and Discussion
The aim of the study is to investigate possibility of using sodium alginate microbeads coated with Chitosan and HPMC as drug release retardants to form sustained release system. Formulated microbeads containing Rioprostil by ionotropic gelation and cross linking method and studied the effects of various factors (concentration of sodium alginate, concentration of coating polymers like chitosan, HPMC and concentration of drug.
Production yield
The yield of all the formulations was found to be in range of 74.5 ± 3.01 to 92.2 ± 2.55. The values of production yield are given in Table-1.
Table 1. Composition and characteristics of drug loaded microbeads
|
Formulation Code |
Drug in mg |
Sodium alginate |
HPMC (%) |
Chitosan (%) |
% yield |
Beads size in µm |
Swelling ratio at pH 1.2 |
Swelling ratio at pH 6.8 |
|
F1 |
5 |
1% |
1 |
- |
73 |
1123±2.23 |
220 |
1722 |
|
F2 |
10 |
1.25% |
0.75 |
- |
76 |
1143.4±1.42 |
234 |
1729 |
|
F3 |
20 |
1.5% |
0.5 |
- |
80 |
1172.5±1.3 |
244 |
1734 |
|
F4 |
30 |
1.75% |
0.25 |
- |
83 |
1223.2±0.9 |
242 |
1755 |
|
F5 |
5 |
1% |
- |
1 |
70 |
1153±2.8 |
260 |
1900 |
|
F6 |
10 |
1.25% |
- |
0.75 |
75 |
1228.6±1.7 |
265 |
2176 |
|
F7 |
20 |
1.5% |
- |
0.5 |
79 |
1276±2.35 |
272 |
2372 |
|
F8 |
30 |
1.75% |
- |
0.25 |
81 |
1353.8±1.8 |
293 |
2532 |
Particle size and entrapment efficiency
Particle size analysis of different formulations of Rioprostil microbeads was carried out using digital micrometer. The values are given in Table-2 and 3.
Table 2. Characteristic of Drug Loaded Beads
|
Formulation code |
Drug content |
Entrapment efficiency (%) |
LSC (%) |
|
F1 |
4.0436+0.03 |
83.23+1.84 |
1.949 |
|
F2 |
8.844+0.016 |
86.97+1.2 |
1.113 |
|
F3 |
18.09+0.02 |
90.57+0.58 |
4.300 |
|
F4 |
27.08+0.02 |
90.76+0.33 |
4.34 |
|
F5 |
4.774+0.03 |
95.49+1.05 |
0.14 |
|
F6 |
8.718+0.14 |
87.18+0.1 |
0.159 |
|
F7 |
16.54+0.04 |
82.73+0.43 |
0.213 |
|
F8 |
24.44+0.37 |
81.46+0.31 |
0.35 |
Table 3. Data for release kinetics for microbeads
|
Formulation code |
Zero order (R2) |
Higuchi matrix (R2) |
Korsmeyer peppas ( R2) |
‘n’ values |
|
F1 F2 F3 F4 F5 F6 F7 F8 |
0.9475 0.9423 0.939 0.957 0.9263 0.9638 0.9202 0.9766 |
0.9454 0.9289 0.9752 0.9471 0.9627 0.9547 0.9179 0.9487 |
0.932 0.938 0.932 0.962 0.935 0.967 0.945 0.962 |
1.423 1.981 1.437 1..648 2.26 2.21 2.05 1.58 |
Table 4. Percentage mucoadhesion of selected formulation (F3&F7)
|
Formulation code |
% Mucoadhesion |
|
F3 |
92 |
|
F7 |
90 |
Swelling Study
Swelling study analysis shows that microbeads prepared with highest alginate concentration 1.75% shows highest swelling rate followed by 1.5%, 1.25% and finally 1% formulation. The probable reason behind this may be due to the percent water uptake of alginate increases with increase in the concentration. The swelling behavior of sodium alginate microbeads coated with different polymers, were observed, by increasing the concentration of coating polymer ratio which enables decreasing of the swelling properties of microbeads. Swelling of dry microbeads is mainly attributed to the hydration of the hydrophilic groups of alginate. On comparing the swelling ratio of all formulations; the lower values obtains at pH 1.2, whereas the highest at increased pH level of the medium. These results suggest that the dried beads swell slightly in the stomach, when they are subsequently transferred to upper intestine.
Drug content and entrapment efficiency
The result of drug content indicated that drug is uniformly dispersed in formulation. It was observed that, due to water insoluble nature of Rioprostil almost all the drug is entrapped in the polymer matrix resulted in higher drug content and encapsulation efficiency. Percentage drug loaded and entrapment given in the table 2, Figure 1.
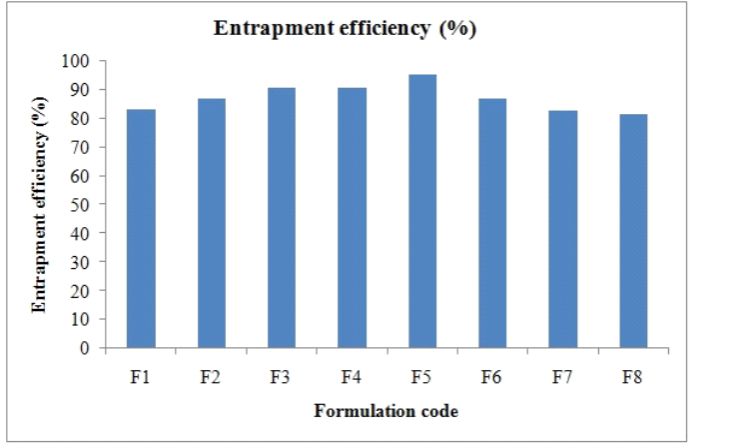 Figure 1. Entrapment efficiency of formulated microbeds
Figure 1. Entrapment efficiency of formulated microbeds
Loose surface crystal study
Loose surface crystal (LSC) study was an important parameter giving an indication of the amount of drug on the surface of the microbeads without proper entrapment. This study indicated that amount of drug released at 15 minute for the immediate action. Data are given in table 4.
Scanning electron microscopy (SEM)
The morphological evaluation of the optimized microbeads formulation (sodium alginate microbeads coated with HPMC) was done by scanning electron microscopy. SEM study revealed that the microspheres were almost spherical in shape with rough outer surface (Figure 2).
 Figure 2. SEM images of optimized batch F3
Figure 2. SEM images of optimized batch F3
In vitro drug release study
The In-Vitro drug release studies of the different formulations cumulative percentage drug release was observed in the range of 84.54 – 97.74. The formulations F1- F8 containing 1.75% of sodium alginate and 0.25 % of coating polymers like HPMC and Chitosan showed a release range of 92.38-99.96 . This indicates that the release rate is further retarded due to addition of coating polymer (Figure 3).
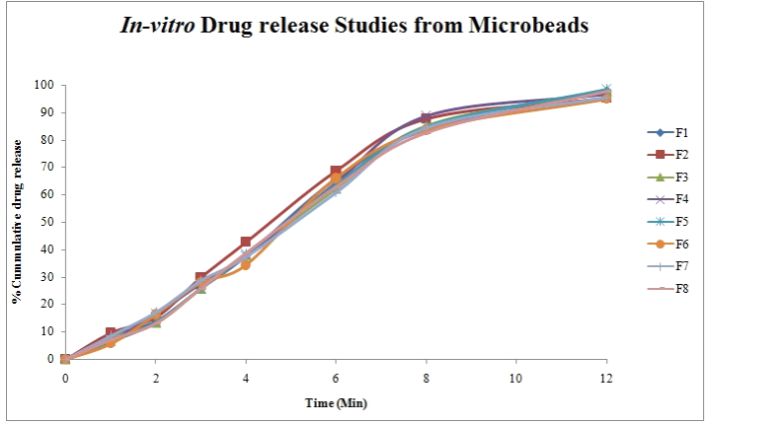 Figure 3. In-vitro drug release from microbeads
Figure 3. In-vitro drug release from microbeads
Release kinetic study
All the release data was fitted into various kinetic models like, zero order, first order, Higuchi, and Korsmeyer-Peppas, in order to find out the mechanism of drug release from polymeric spheres.
The value of ‘n’ gives an indication of the release mechanism; when n = 1, the release rate is independent of time (zero-order) (case II transport), n = 0.5 for Fickian diffusion and when 0.5 < n <1.0, diffusion and non-Fickian transport are implicated. Lastly, when n > 1.0 super case II transport is apparent. Regression coefficient and ‘n’ were calculated and is given in Table: 3. From the table, the value showed that, prepared microbeads exhibited zero order kinetics followed by super case -II transport.
Ex-vivo mucoadhesion study of microspheres
The results of the ex-vivo mucoadhesion study indicated that optimized microbeads had good mucoadhesive properties. The percentage of microbeads attached to the mucosa up to 6 hours. It was found that sodium alginate microbeads coated with HPMC show more mucoadhesion property as compared to other formulation. Because combination of both the polymers increases the viscosity; produce more viscous gel which helps to increase adhesion with intestinal mucosa. Therefore, it is suggested that prepared microbeads adhere to the intestinal mucosa for a prolonged period where they release drug in a sustained manner before being eroded off (Table 4).
Conclusions
In conclusion, ionotropic gelation and cross linking technique can be used for preparation of Rioprostil microbeads using sodium alginate and with other coating polymers like chitosan and HPMC as drug release retardants. Prepared microbeads shown higher drug entrapment and prolonged release characteristics. Rioprostil release from microbeads was influenced by alginate and coating polymer concentration. The beads formed have a spherical shape with rough surface as evidenced by SEM. Didn’t reveal any significant drug interaction. From the in vitro wash off test for mucoadhesive strength determination of various formulations, it was observed that microbeads formulation (F3) exhibits greater mucoadhesive strength than other formulation (F7).The result of in-vitro release and release kinetic indicated sustained release and exhibited zero order kinetic followed by super case II- transport. Therefore, one can assume that the Rioprostil microbeads are promising pharmaceutical dosage forms by providing controlled release drug delivery systems and improving bioavailability. The entire process is feasible in an industrial scale and demands pilot study.
Acknowledgements
The authors are grateful to the Hetero drugs limited (Hyderabad, India) for providing gift drug sample.
References
Bansal P, Vasireddy S, Parikh D. 1993. Effect of compression on the release properties of polymer coated niacin granules. Journal of Controlled Release, 27: 157–163.
Barratt G. 2000. Therapeutic applications of colloidal drug carriers. Pharmaceutical Science and Technology Today, 3: 163–71.
Bechgaard H, Nielson G. 1978. Controlled release multiple units and single-unit doses. Drug Development and Industrial Pharmacy, 4: 53–67.
Dashevsky A, Kolter K, Bodmeier R. 2004. Compression of pellets coated with various aqueous polymer dispersions. International Journal of Pharmaceutics, 279: 19–26.
Dressman J, Reppas C. 2000. In vitro-in vivo correlations for lipophilic, poorly water-soluble drugs. European Journal of Pharmaceutical Science, 11: 73-80.
Eldem T, Speiser P, Hincal A. 1991. Optimization of spray-dried and -congealed lipid micropellets and characterization of their surface morphology by scanning electron microscopy. Pharmacological Research, 8: 47–54.
Florence A. 1997. The oral absorption of micro- and nanoparticulates: neither exceptional nor unusual. Pharmaceutical Research, 14: 259–266.
Korsmeyer R, Peppas N. 1981. Effect of the morphology of hydrophilic polymeric matrices on the diffusion and release of water-soluble drugs. Journal of Membrane Science, 9: 211-227.
Lambert O, Oscar N, Veronique L, Daniel B. 2000. Poly (ethylene carbonate) microspheres: manufacturing process and internal structure characterization. Journal of Controlled Release, 67(1): 89-99.
Lim L, Wan S. 1997. Propranolol hydrochloride binding in calcium alginate bead. Drug Development and Industrial Pharmacy, 23 (10): 973-980.
Millili G, Schwartz J. 1990. The strength of microcrystalline cellulose pellets – the effect of granulating with water/ethanol mixtures. Drug Development and Industrial Pharmacy, 16: 1411-1426.
Nagata K, Takagi E, Tsuda M. 1995. Inhibitory action of Lansoprazole and its analogs against helicobacter pylori: inhibition of growth is not related to inhibition of urease. Antimicrobial Agents and Chemotherapy, 39: 567-70.
Narkar M, Sher P, Pawar A. 2010. Stomach specific controlled release gellan beads of acid soluble drug prepared by ionotropic gelation method. AAPS Pharm SciTech, 11(1): 267-277.
Petersen K, Schmutzler W. 1999. Proton pump inhibitors. Active substance release from different preparations. Deutsche Apotheker Zeitung, 139: 64–65.
Pillay V, Fassihi R. 1999. In-vitro release modulation from cross linked pellets for site-specific drug delivery to the gastrointestinal tract: I. Comparison of pH-responsive drug release and associated kinetics. Journal of Controlled Release, 59(2): 229-242.
Ragnarsson G, Sandberg A, Johansson M. 1992. In-vitro release characteristics of a membrane-coated pellet formulation-Influence of drug solubility and particle-size International Journal of Pharmaceutics, 79: 223-232.
Roy S, Das S. 1989. Design and in-vitro evaluation of dapsone-loaded micropellets of ethyl cellulose. Pharmaceutical Research, 6(11): 945-948.
Tomofumi Y, Hiraku O, Yoshiharu M. 2001. Sustained release ketoprofen microparticles with ethyl cellulose and carboxyl methyl ethyl cellulose. Journal of Controlled Release, 75(3): 271-282.
Turkan E, Speiser P, Hincal A. 1991. Optimization of Spray-Dried and Lipid Congealed Micropellets and Characterization of Their Surface Morphology by Scanning Electron Microscopy. Pharmaceutical Research 8(1): 47-54.
Ulrich K, Matthias S. 2005. Topical delivery of therapeutic agents in the treatment of inflammatory bowel disease. Advanced Drug Delivery Review. 57(2): 267-279.