

Lali Growther1*, Sukirtha K.2
1*Professor and Head, Department of Microbiology, Hindusthan College of Arts and Science, Coimbatore, India
2Assistant Professor, Department of Microbiology, Hindusthan College of Arts and Science, Coimbatore, India
*Corresponding author
Dr. Lali Growther,
Professor and Head,
Department of Microbiology
Hindusthan College of Arts and Science, Coimbatore-641028 India.
Abstract
Objective: Psidium guajava is a plant used in traditional medicine for different ailments. The parts of the plant like leaves, fruit and the bark are used as medicine. They are used as antibacterial, antiparasitic, febrifuge, antispasmodic, to treat rheumatism, convulsions, and as astringent. The bark of this plant is used to treat dysentery, astringent and used as a bath to treat skin ailments. The objective of this study is to study the antimicrobial activity of Psidium guajava leaves and bark extracts and the bioactive compounds of leaves extracts by HPLC and NMR. Materials and methods Psidium guajava leaves and bark were soxhlet extracted using different solvents. The antimicrobial activities were studied by agar well diffusion method. Phytochemical analysis was done and the methanol extract was subjected to HPLC. The purified ethyl acetate fraction was subjected to NMR. Results and conclusion: The leaves and bark extracts showed significant antibacterial and antifungal activity against Staphylococcus aureus, Streptococcus pyogenes, E.coli, Pseudomonas aeruginosa and Candida albicans. HPLC analysis of the leaf extracts revealed the presence of bioactive compound Quercetin.
Keywords: Psidium guajava, antimicrobial activity, Quercetin
Introduction
Pisdium guajava is the plant that bears the guava fruit and comes under the family of Myrtaceae. Extensive literature survey revealed that Pisdium guajava, acclaimed as ‘poor man’s apple of the tropics’, has a long history of traditional use for a wide range of diseases. The plant is readily available in the tropics and within the reach of the local populace. The leaves and bark of guava tree have a long history of medicinal uses. Various part of the guava tree have been traditionally used as a cough sedative (Joseph and Priya, 2011), an antidiarrheic, in the management of hypertension, obesity and in the control of diabetes mellitus (Begum et al., 2004). The leaf extract was found to possess anticestodal (Tangpu and Yadav, 2006), hepatoprotective (Chen et al., 2006), antioxidant activities (Nair and Chanda, 2007), anti-inflammatory (Ojewole, 2006) and antimicrobial (Roy et al., 2006). Four antibacterial compounds were isolated from leaves of Pisdium guajava L., two new flavonoid glycosides, morin-3-o-alpha-L-lyxopyranoside and morin-3-o-alpha-L-arabopyranoside and two known flavonoids, guaijavarin and quercetin. In India, decoction of the leaves and bark of guava is used to cure diarrhea, dysentery, vomiting and sore throats, and to regulate menstrual cycles. Guavas are free from fat and cholesterol. They are also an excellent source of fiber, potassium and vitamin A (Mishra et al., 2017).
Materials and methods
Preparation of extracts
Leaves and Barks of Psidium guajava were collected from Coimbatore. The barks were air dried in shade under natural conditions. Dried bark were powdered and extracted by soxhlet apparatus by increasing order of polarity with petroleum ether, benzene, chloroform, ethanol and methanol for 48 hours (Shirley and Growther, 2015). The various extracts obtained were concentrated and dissolved in Dimethyl sulphoxide (DMSO).
Antibacterial and antifungal activity of extracts
The prepared extracts were tested for antibacterial activity against different bacterial and fungal pathogens by agar well diffusion method. 1g of the prepared extract was dissolved in 1ml of DMSO. 6mm wells were made on Mueller Hinton agar plates seeded with the test organisms and different concentrations of the extracts (10-90mg) were loaded onto the wells. A gentamicin (G-30mcg) disc was used as a standard antibiotic and DMSO was added in a separate well. The plates were incubated for 24 hours and after incubation the zone of inhibition was noted against each extract (Growther et al., 2012).
Minimum Bactericidal Concentration
The MBC was performed to test the antimicrobial activity of active methanol extract using tube dilution method. The MBC was defined as lowest concentration able to kill any microbe. Dilutions of the extract were prepared in sterile nutrient broth to get a final concentration of 2mg, 4mg, 8mg, 16mg, 32mg and 64mg/ml respectively (Sukirtha and Growther, 2012). To each of these dilutions, a loop full of the cultures adjusted to 0.5 Mc Farland standard, was inoculated and all the tubes were incubated at 37oC for 24 hrs. After incubation, loopful from each tube was inoculated onto nutrient agar plates. The plate without growth was recorded as MBC.
Phytochemical Screening
The extract with the potent antibacterial activity was used for further study. Phytochemical screening for flavonoids, alkaloids, tannins, saponins and terpenoids were done following standard methods (Harborne 1998; Trease and Evans, 1989; Sofowora, 1993; Vinothini and Growther, 2016).
High Performance Liquid Chromatography
The crude methanol extracts were subjected to HPLC. The HPLC was performed on YMC C18 column, 5µm, 250 × 4.6mm, using solvent mixtures – 80% hexane and 20% ethyl acetate. The flow rate was 1ml/min. The dried leaf samples were separately soxhlet extracted in 80% methanol (100 ml/ gm dry weight) on a water bath for 24 hrs (Subramanian and Nagarajan, 1969). The extract was concentrated and re-concentrated in petroleum ether (40°- 60°C) (fraction-I), ethyl ether (fraction II) and ethyl acetate (fraction-III) in succession. Each of the steps was repeated three times to ensure complete extraction in each case. Fraction I was rejected since it was rich in fatty substances whereas fraction III was hydrolyzed by refluxing with 7% H2SO4 (10 ml/gm residue) for 5 hours. The mixture was filtered and the filtrate extracted with ethyl acetate in a separating funnel. The ethyl acetate layer was washed with distilled water till neutrality and dried in vacuo (Zhu et al., 2013). The residues were taken up in small volumes of ethanol separately and then subjected to NMR analysis.
Results and discussion
Antimicrobial activity
The antibacterial activities of all the extracts were compared with that of the standard antibiotic, gentamicin and DMSO. Antifungal activity was compared with Amphotericin B. The antibacterial activities of different extracts (leaf and bark) at six different concentrations were analyzed and the results were tabulated (Table 1 and 2). The methanol extract of leaves of Psidium guajava showed high antimicrobial activity. Bark extracts showed comparatively lower activity and ethanol extracts were found to be more effective than methanol.
Table 1.Antimicrobial properties of Leaf extracts of Psidium guajava
|
Organisms |
Zone of Inhibition in mm |
|||||||||||||||||||||||||||||||||
|
Petroleum Ether |
Benzene |
Chloroform |
Ethanol |
Methanol |
Control |
|||||||||||||||||||||||||||||
|
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
Gentamicin |
DMSO |
|||
|
S.aureus |
7 |
8 |
8 |
10 |
11 |
12 |
8 |
8 |
9 |
10 |
11 |
11 |
- |
- |
7 |
8 |
9 |
10 |
10 |
11 |
12 |
14 |
16 |
18 |
16 |
17 |
17 |
18 |
19 |
20 |
14 |
- |
||
|
K.pneumoniae |
8 |
10 |
12 |
14 |
16 |
17 |
9 |
9 |
10 |
10 |
11 |
11 |
- |
8 |
9 |
10 |
11 |
12 |
10 |
11 |
12 |
13 |
14 |
14 |
12 |
12 |
14 |
15 |
16 |
18 |
11 |
- |
||
|
S. typhi |
10 |
11 |
11 |
12 |
13 |
15 |
7 |
7 |
8 |
8 |
9 |
10 |
- |
9 |
10 |
11 |
12 |
14 |
8 |
9 |
10 |
12 |
13 |
15 |
12 |
14 |
15 |
16 |
18 |
19 |
10 |
- |
||
|
S.dysentriae |
7 |
8 |
9 |
10 |
10 |
10 |
- |
- |
- |
8 |
9 |
9 |
8 |
9 |
9 |
10 |
10 |
11 |
9 |
10 |
11 |
12 |
14 |
16 |
12 |
12 |
12 |
13 |
14 |
16 |
10 |
- |
||
|
E.coli |
8 |
8 |
9 |
9 |
10 |
10 |
7 |
8 |
9 |
10 |
11 |
11 |
7 |
8 |
10 |
11 |
12 |
14 |
10 |
11 |
12 |
14 |
16 |
18 |
14 |
16 |
18 |
20 |
21 |
21 |
11 |
- |
||
|
P.aeruginosa |
- |
- |
8 |
8 |
10 |
10 |
- |
8 |
8 |
9 |
9 |
11 |
- |
9 |
10 |
12 |
13 |
14 |
- |
8 |
9 |
11 |
12 |
13 |
12 |
13 |
14 |
16 |
16 |
20 |
14 |
- |
||
|
C. albicans |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
8 |
8 |
9 |
11 |
12 |
15 |
16 |
12 |
13 |
14 |
15 |
16 |
18 |
20 |
20 |
22 |
22 |
24 |
25 |
18 Amphotericin B |
- |
||
Table 2.Antimicrobial properties of Bark extracts of Psidium guajava
|
Organisms |
Zone of Inhibition in mm |
|||||||||||||||||||||||||||||||||
|
Petroleum Ether |
Benzene |
Chloroform |
Ethanol |
Methanol |
Control |
|||||||||||||||||||||||||||||
|
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
10 |
20 |
30 |
50 |
70 |
90 |
Gentamicin |
DMSO |
|||
|
S.aureus |
- |
- |
- |
8 |
9 |
10 |
- |
- |
- |
8 |
9 |
9 |
10 |
11 |
12 |
13 |
14 |
15 |
12 |
13 |
14 |
15 |
16 |
18 |
10 |
11 |
13 |
14 |
15 |
16 |
14 |
- |
||
|
K.pneumoniae |
- |
- |
- |
- |
8 |
8 |
- |
- |
- |
7 |
8 |
9 |
- |
8 |
9 |
11 |
12 |
13 |
12 |
14 |
15 |
16 |
17 |
17 |
8 |
9 |
11 |
12 |
13 |
14 |
11 |
- |
||
|
S. typhi |
- |
- |
- |
- |
8 |
9 |
- |
- |
- |
8 |
10 |
12 |
10 |
11 |
12 |
13 |
14 |
15 |
13 |
14 |
15 |
16 |
17 |
18 |
7 |
8 |
10 |
11 |
12 |
12 |
10 |
- |
||
|
S.dysentriae |
- |
- |
- |
- |
- |
- |
- |
- |
- |
7 |
8 |
9 |
9 |
9 |
10 |
11 |
13 |
15 |
12 |
12 |
12 |
14 |
15 |
16 |
- |
- |
8 |
10 |
11 |
12 |
10 |
- |
||
|
E.coli |
- |
- |
- |
7 |
8 |
9 |
8 |
9 |
10 |
11 |
12 |
14 |
9 |
9 |
9 |
10 |
12 |
14 |
14 |
15 |
16 |
17 |
18 |
20 |
10 |
11 |
12 |
13 |
14 |
15 |
11 |
- |
||
|
P.aeruginosa |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
7 |
8 |
9 |
9 |
10 |
10 |
- |
8 |
9 |
10 |
11 |
12 |
- |
- |
- |
8 |
9 |
10 |
14 |
- |
||
|
C. albicans |
- |
- |
- |
- |
- |
- |
9 |
9 |
10 |
12 |
13 |
14 |
- |
8 |
9 |
10 |
11 |
12 |
12 |
13 |
14 |
16 |
18 |
20 |
10 |
11 |
12 |
13 |
14 |
15 |
18 Amphotericin B |
- |
||
Minimum Bactericidal Concentration
The active methanol extract of Psidium guajava leaves and ethanol extracts of bark were subjected to the determination of Minimum bactericidal concentration. Minimum bactericidal concentration was found to be 8.0mg/ml for Psidium guajava leaf extract and 16mg/ml for bark extracts.
Phytochemical analysis
Phytochemical analysis of these extracts showed the presence of alkaloids, steroids, flavanoids, tannins and Saponins in Psidium guajava (Table.3). This proves the scientific basis of the use of these plant extracts in folk medicine. Bark extracts were rich in tannins.
Table 3. Phytochemical analysis of Psidium guajava extracts
|
S. No |
Source |
Phytochemicals |
||||
|
Alkaloids |
Steroids |
Flavonoids |
Saponins |
Tannins |
||
|
1. |
Leaf extract |
+ |
- |
+ |
+ |
- |
|
2. |
Bark extract |
+ |
+ |
+ |
+ |
+ |
HPLC analysis
The methanol extract showed 4 peaks (Figure 1) with retention time (RT) of 2.5, 2.59, 2.9 and 5.1.min. The peak with RT of 2.5min coincided with that of standard quercetin (Figure 2). Thus the presence of the compound Quercetin was confirmed by HPLC. The NMR analysis of the leaf extracts were shown in figure 3 and 4.
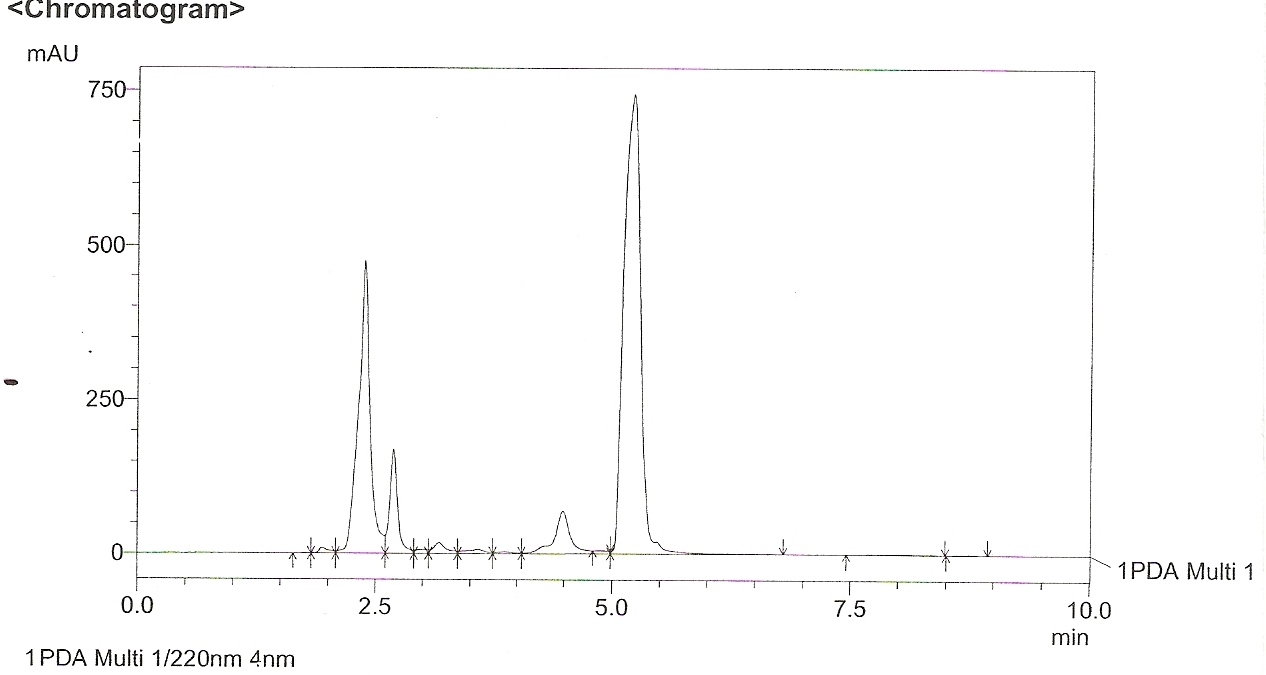 Figure 1. HPLC Chromatogram of crude methanol extract of Psidium guajava
Figure 1. HPLC Chromatogram of crude methanol extract of Psidium guajava
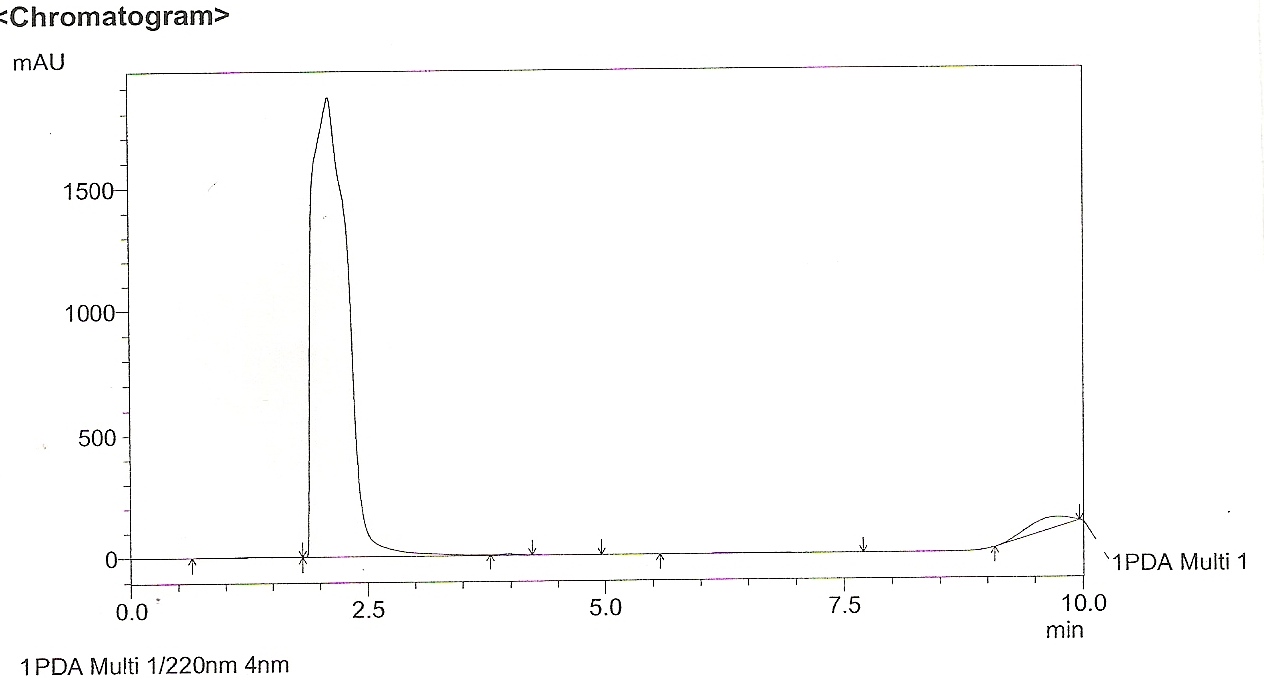 Figure 2. HPLC Chromatogram of standard quercetin
Figure 2. HPLC Chromatogram of standard quercetin
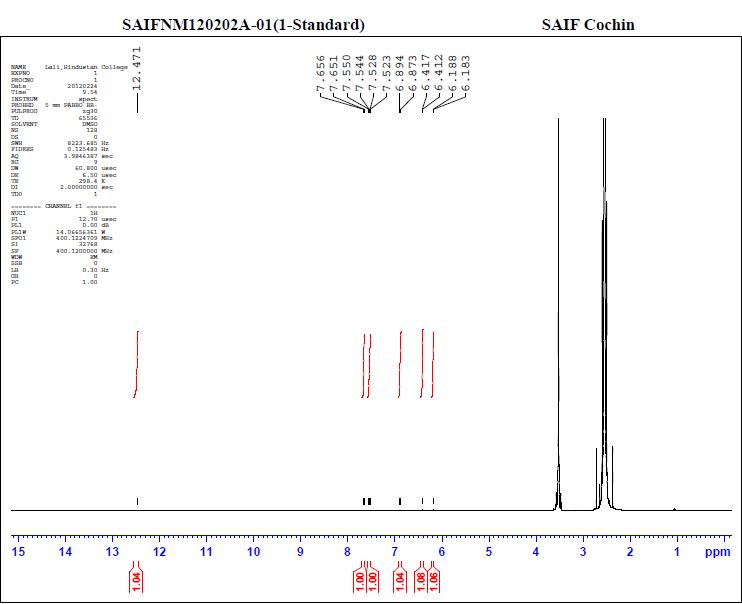 Figure 3. NMR analysis of the standard quercetin
Figure 3. NMR analysis of the standard quercetin
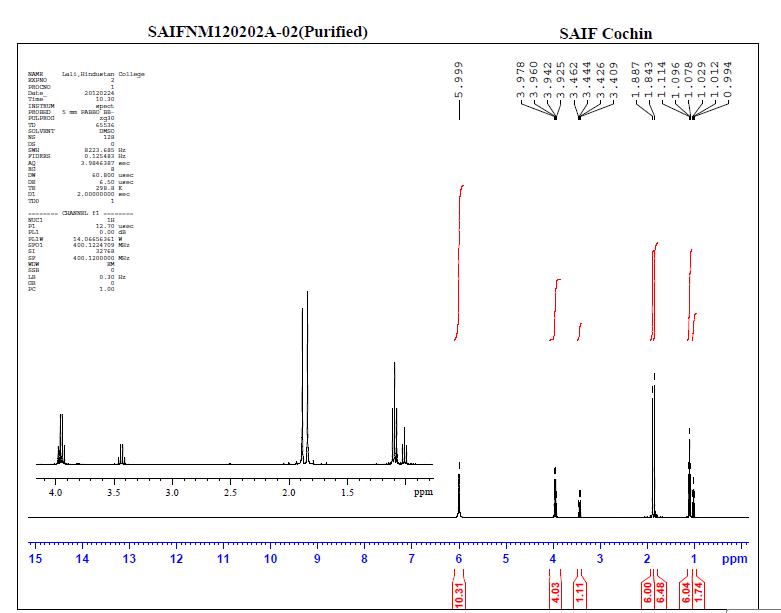 Figure 4. NMR analysis of the leaf extracts
Figure 4. NMR analysis of the leaf extracts
Pisdium guajava leaf and bark extracts showed good antibacterial activity. Methanol extracts of leaf were more effective. These zone diameters were higher than the zone of the standard antibiotic gentamicin. The methanol extract with good antibacterial activity was further analysed for their bio-active phytochemicals. According to the Henie et al. (2009), the methanol extracts of Psidium guajava produced the highest antimicrobial activity against microorganisms like Aeromonas caviae, S. typhimurium, S. enteritidis, and Vibrio parahemolyticus but E.coli was not inhibited at 10 µg concentrations. MIC of methanol extract of Psidium guajava against the different microorganisms ranged between 1-5 µg/ml. But other studies by Caceres et al.(1990), Lin et al. (2002) and Voravuthikunchai et al. (2004), had showed that E.coli O157: 47 and E. coli were sensitive to Psidium guajava leaf extracts. According to Voravuthikunchai et al. (2004) MIC of Psidium guajava leaf extracts ranged from 0.19-0.78µg/ml for aqueous extract and 6.25-12.5 mg/ml for the ethanolic extracts. Lutterodt et al. (1999) suggested that the high activity of Psidium guajava against the bacteria tested may allow it to be used in the treatment of food borne diseases or prevent death due to the dehydration caused by continuous diarrhea especially in children. Thus pharmacological studies of purified compounds are warranted from this potential plant extracts to identify new antibacterial compounds to combat antimicrobial resistance.
Conflicts of interest: None
References
Begum S, Hassan SI, Ali SN, Siddiqui BS. 2004.Chemical constituents from the leaves of Pisdium guajava. Natural Product Research, 18(2):135-140.
Caceres A, Cano O, Samayoa B, Aguilar L. 1990. Plants used in Guatemala for the treatment of gastrointestinal disorders. Screening of 84 plants against enterobacteria. Journal of Ethnopharmacology, 30(1):55-73.
Chen HC, Sheu MJ, Wu CM. 2006. Characterization of volatiles in guava (Pisdium guajava L. cv. Chung-Shan-Yueh-Pa) fruit from Taiwan. Journal of Food and Drug analysis, 14(4):398-402.
Harborne JB. 1998. Phytochemical methods guide to modern Technique of Plant analysis (3rd Edn). Chapman and Hall London. pp:135.
Henie EFP, Zaiton H, Suhaila M. 2009. Bacterial membrane disruption in food pathogens by Psidium guajava leaf extracts. International Food Research Journal, 16:297 -311.
Joseph B, Priya RM. 2011. Phytochemical and Biopharmaceutical Aspects of Pisdium guajava (L.) Essential Oil: A Review. Reseach Journal of Medicinal Plant, 5(40):432-442.
Lali Growther, Sukirtha K, Savitha N, Niren Andrew S. 2012. Antibacterial activity of Punica granatum peel extracts against shiga toxin producing E. coli, In,ternational Journal of Life Sciences Biotechnology and Pharma Research, 163-172.
Lin J, Puckree T, Mvelase TP. 2002. Antidiarrhoeal evaluation of some medicinal plants used by Zulu traditional healers. Journal of Ethnopharmacology, 79(1):53-56.
Lutterodt GD, Ismail A, Basheer RH and Baharudin HM.1999. Antimicrobial effects of Psidium guajava extract as one mechanism of its antidiarrhoeal action. Malaysian Journal of Medical Sciences, 6(2):17-20.
Mishra R, Tiwari P, Srivastava M, Singh CS, Ghoshal S. 2017. A comprehensive review on Psidium guajava Linn (Amaratafalam). International Journal of Ethnobiology & Ethnomedicine, 4(1):1-6.
Nair R, Chanda S. 2007. In vitro antimicrobial activity of Pisdium guajava L. leaf extracts against clinically important pathogenic microbial strains. Brazilian Journal of Microbiology, 38:452-458.
Ojewole JAO. 2006. Anti – Inflammatory and analgesic effects of Pisdium guajava L.(Myrtaceae) leaf aqueous extracts in rats and mice. Methods and Findings in Experimental and Clinical Pharmacology, 28(7):441-446.
Renita Shirley, Lali Growther. 2015. Antibacterial activity of ethnomedicinal plants of Irulars of western ghats, International Journal of Pharmaceutical Science and Research, 6(7):2837-2840
Roy CK, Kamath JV, Asand M. 2006. Hepatoprotective activity of Pisdium guajava Linn.leaf extract. Indian Journal of Experimental Biology, 44:305-311.
Sofowora A. 1993. Medicinal plants and Traditional medicine in Africa. 2nd Edn. John Wily and Sons. New York, 6-56.
Subramanian SS, Nagarajan S. 1969. Flavonoids of the seeds of Crotlaria retusa and C.striata. Current Science, 38:65- 68.
Sukirtha K, Lali Growther. 2012. Antibacterial, antifungal and phytochemical analysis of selected medicinal plants. Journal of Natural Product and Plant Resources, 2(6):644-648.
Tangpu TV, Yadav AK. 2006. Anticestodal efficacy of Pisdium guajava against experimental Hymenolepis diminuta infection in rats. Indian Journal of Pharmacology, 38:29-32.
Trease GE, Evans WC. 1989. “Pharmacognosy” 14th Edn. W.B. Scandars Company, Ltd. London. 269-300.
Vinothini R, Lali Growther. 2016. Antimicrobial and Phytochemical Analysis of Methanolic and Aqueous Extract of Annona muricata (Leaf and Fruit). International Journal of Current Microbiology and Applied Sciences, 5(10):617-625.
Voravuthikunchai S, Lortheeranuwat A, Jeeju W, Sririrak T, Phongpaichit S, Supawita T. 2004. Effective medicinal plants against enterohaemorrhagic Escherichia coli O157:H7. Journal of Ethnopharmacology, 94(1):49-54.
Y Zhu, Y Liu, Y Zhan, L Liu, Y Xu, T Xu, T Liu. 2013. Preparative Isolation and Purification of Five Flavonoid Glycosides and One Benzophenone Galloyl Glycoside from Psidium guajava by High-Speed Counter-Current Chromatography (HSCCC), Molecules, 18(12):15648-15661.