

Pushpendra K. Jain1, Alok Pal Jain2
1Deapartment of Pharmaceutical Sciences, Dr. H. S. Gour University (M.P.), India
2RKDF College of Pharmecy, SRK University, Hoshangabad Road, Jat Khedi, Misrod, Bhopal (M.P.) India
Corresponding Address
Dr. Alok Pal Jain
RKDF College of Pharmecy
SRK University, Hoshangabad Road, Jat Khedi, Misrod, Bhopal
Abstract
Objective: Lichens are symbiotic organisms consisting of a fungus and a photosynthetic partner which can be either an alga or cyanobacterium. The objective of present study was to test dermal wound healing effect of Parmotrema reticulatum lichens with possible antioxidant and antibacterial mechanism. Materials and methods: The acetone extract was separate from powdered P. reticulatum lichens for phytochemical and wound healing screening. In-vivo wound healing activity was investigated using incision and dead space wound methods. Tensile strength and biochemical parameters (collagen and protein content) were determined from healed tissues by standard methods. Results: The results of phytochemical studies showed that acetone extract of P. reticulatum confirmed the presence of flavonoids, glycosides and terpenoids. The tensile strength of skint tissues treated with acetone extract was increased significantly in comparison to control group. The hydroxyproline content and protein content found increased on 9th day after treatment with 5% w/w ointment containing acetone extract. The antioxidant status of healed skin tissues was improved upon treatment with acetone extract ointment (5%w/w). Conclusions: The results of present studies can be concluded that the significant dermal wound healing activity may be due to presence of flavanoids and usnic acid in acetone extract by possible antioxidant and antibacterial activity.
Keywords: Antibacterial, Parmotrema reticulatum, dermal wound healing, usnic acid
Introduction
Wound repair is a complex dynamic process integrated sequence of biochemical, cellular and physiological process. The sequential phases of healing process are inflammation, proliferation and migration of connective tissue cells, production of extracellular matrix including collagen synthesis, epithelial cells migration and proliferation leading to neovascularization of wounded tissue. In the inflammatory phase, polymorphonuclear leukocytes and lymphocytes are attracted by soluble mediators and play a key role by secreting cytokines and growth factors. The proliferative phase is characterized by angiogenesis, collagen deposition, epithelialization and wound contraction. The last phase re-modeling phase is characterized by re-model of collagen and other extracellular matrix proteins convert into mature scar tissue (Wilgus, 2008).
Lichens, are symbiotic organisms of fungi and algae, synthesize characteristic secondary compounds. Some species of Parmotrema have also been reported for antimicrobial (Sati and Joshi, 2011; Balaji and Hariharan, 2007) and antioxidant (Sharma and Kalikotay, 2012; Stanly et al., 2011) properties. Parmotrema reticulatum, was first described by Taylor (1836; as Parmelia reticulate Taylor) based on the reticulated maculae of the upper surface, and characterized by marginal soralia and simple to squarrose rhizines (Divakar et al., 2005). P. reticulatum occur in Dima Hasao Hills district of Assam, India. Besides, lichen metabolites exert a wide variety of biological activity including antimycobacterial, antiviral, anti-inflammatory, antiproliferative and cytotoxic effects (Muller, 2001). Usnic acid is a naturally occurring dibenzofuran derivative found in P. reticulatum. The objective of present study was to investigate dermal wound healing activity of P. reticulatum acetone extracts in albino rats.
Material and methods
Collection and identification of lichen materials
The P. reticulatum lichens were collected from the stem barks of trees growing in the location of Amarkantak town, India. The lichens material was identified and a voucher specimen was submitted into the Department of Botany, Dr. H. S. Gour University (M.P.), India. The powdered materials were sieved through 40-mesh size and stored in well-closed container for extraction purpose.
Preparation of extract ointment and phytochemical screening
Powdered dried lichens were extracted in soxhlet apparatus with petroleum ether for defating. The defatted lichen material was extracted with acetone repeatedly until complete the extraction. Acetone extract was concentrated under reduced pressure to yield dried extract and were subjected to ointment preparation by using simple ointment base B.P (Anonymous, 1953). Before formulation preparation, the extract was tested for presence of different phytoconstituents.
Dermal wound healing activity
Animals selection
Wistar albino rats (150-160 g) were selected for wound healing study. The rats were acclimatized to the laboratory environment for about 7 day’s period prior experiment. Six animals were taken in each group for study. Group I was referred as control group, while Group II and III referred as treated groups received ethanol extract 2% w/w and 5% w/w ointment, respectively. Group IV referred as reference group that received Povidone-Iodine ointment. The animal experimentation was preformed according to permission of Institutional animal ethical Committee.
Longitudinal incision wound method
Incision wound method was selected to study of tensile strength of healed skin. The albino rats were anaesthetized before wound creation and a 1.5 cm long incision was made through the skin at dorsal portion of rat skin. No local or systemic antimicrobials were used throughout the experiment. The both edges of wound kept together and stitched with black silk surgical thread and a curved needle used for stitching (Hemalata et al., 2001). Both wound edges were tightened for good closure of the wound and after stitching, wound was left undressed. All extracts and reference drug ointment were applied daily up to 9 days; upto wounds were healed completely. The sutures were removed on the 9th day and tensile strength of healed wound skin was measured by Tensiometer.
Dead space wound method
This model is used for the study of granuloma tissue. Animals were anaesthetized by light ether and an implantation of polypropylene tube (2.0 x 0.5), by making longitudinal incision in the lumber region on the dorsal surface. On the 9th post-wounding day, granuloma tissue formed on an implanted tube was dissected out carefully. Granuloma tissue was dried (60oC) and stored in 10% formalin for the biochemical parameters study (Shirwaiker et. al, 2003). After 9th day of healing skin tissues were investigated for hydroxyproline and protein content determination.
Measurement of tensile strength
The tensile strength of skin tissue indicates the degree of wound healing. For the measurement of tensile strength, an instrument is known as Tensiometer, was used which is designed by Kuwano method (Kuwano et al., 1994). The different extracts ointment and reference were topically applied daily up to 9 days. The tissue samples on day 9th were taken from each group. The tensile strength of treated wounds was compared with control and reference ointment treated groups. Increased tensile strength indicates the better wound healing stimulated by applied topical extracts.
Biochemical estimations
Wound tissues were analyzed for collagen content by determination of hydroxyproline content, which is a basic constituent of collagen. Tissues were dried in a hot air oven at 60-70 oC to constant weight and hydrolysed in 6 N HCl at 130 0C for 4 hour in sealed tubes. The hydrolysate was neutralized to pH 7 then subjected to Chloramine-T oxidation for 20 minutes (Woessner, 1961). The reaction was terminated by addition of 0.4M perchloric acid and developed color with Ehlrich reagent at 60 oC was read at 557 nm in UV (Agilant Technology) Spectrophotometer.
Tissue protein level was determined by treatment of tissue lysate with a mixture of sodium tartrate, copper sulphate and sodium carbonate. This was left to stand for 10 minutes and then treated with Folin-Ciocalteau reagent that resulted in a bluish color in 20-30 minutes. The absorbance was measured using standard Spectrophotometry techniques 660 nm (Lowry et al., 1951).
SampleS of granuloma tissue collected from wound sites were tested for antioxidants assay. Superoxide dismutase (SOD) was assayed (Misra and Fridovich, 1972) based on the inhibition of epinephrine autoxidation by the enzyme. Reduced glutathione (GSH) level was determined by the method of Moron (Moron et al., 1979). Catalase was estimated following the breakdown of hydrogen peroxide (Beers and Sizer, 1952). Skin homogenates were immediately precipitated with 0.1 ml of 25% TCA and removed after centrifugation. Free-SH groups were assayed in a total 3 ml volume by the addition of 2 ml of 0.6 mM DTNB and 0.9 ml 0.2 mM sodium phosphate buffer (pH 8.0) to 0.1 ml of the supernatant and the absorbance was read at 412 nm using UV spectrophotometer.
Statistical analysis
The data were expressed as mean values ±SEM using InStat software 3 and tested with analysis of variance followed by the multiple comparison test of Tukey–Kramer with P< 0.01 were considered significant.
Results
Phytochemical screening
The qualitative chemical screening of acetone extract was resulted that it confirmed the presence of glycosides and flavonoids. The reported literature are also supported the presence of usnic acid in acetone extract of P. reticulatum.
Pharmacological screening for dermal wound healing
Results of tensile strength measurement are shown in figure 1. Tensile strength of wound skin was determined at 9th day of treatment in longitudinal incision wound. The tensile strength of skin tissues collected from the test group treated with acetone extract ointment (5% w/w) ointment (641.73±57.21) was observed significant (P< 0.05) improvement in comparison to control group (358.2±28.17). It is also approx similar to the reference animal group.
In case of dead space wound model, the hydroxyproline content and protein content found significantly in the group treated with acetone 2 and 5% w/w ointment in dose dependent manner. The hydroxyproline content of acetone extract treated group of animals were found 41.62±3.60and 52.43±4.72 respectively for 2% and 5% w/w ointment which were significantly greater than the control group.
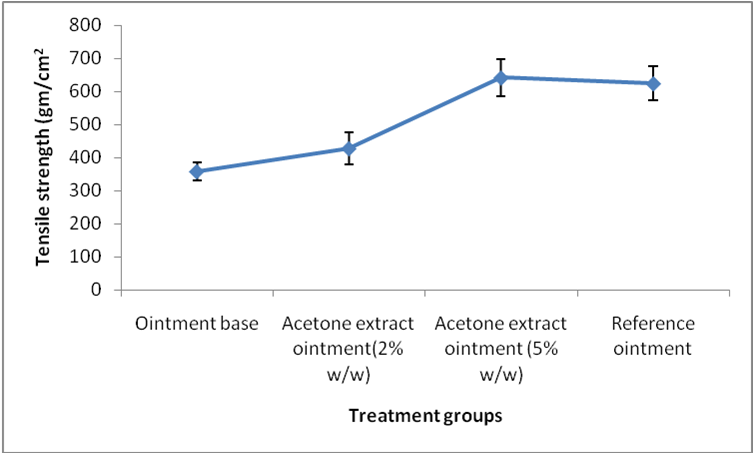
Figure 1. Effect of acetone extracts of P. reticulatum on tensile strength of longitudinal incision wound
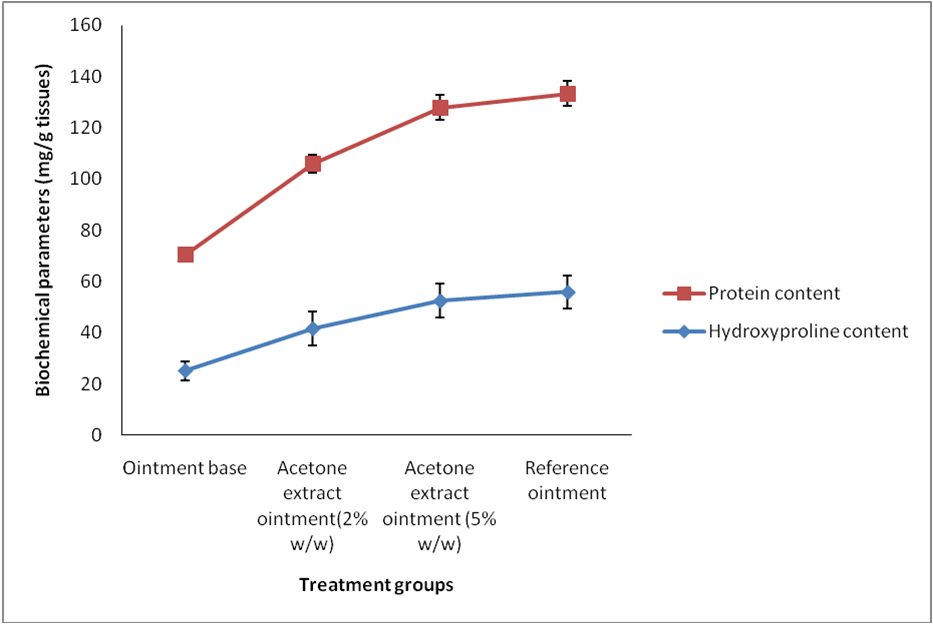
Figure 2. Effect of acetone extract of P. reticulatum on hydroxyproline and protein content of skin tissues collected from dead space wound
The protein content of skin tissues from 5% w/w acetone ointment treated groups was showed higher (75.46±6.51) than the control group (45.20±3.68) and comparable to the reference ointment group (Table 1). The higher protein content of treated animals suggests that acetone extract stimulate cellular proliferation through an unknown mechanism.
Table 1. Effect of acetone extract of P. reticulatum on antioxidants levels of skin tissues collected from dead space wound
|
Animal groups |
Enzymatic study |
||
|
SOD(μg/50mg tissue) |
CAT(μmol/50 mg tissue) |
GSH(μmol/50 mg tissue) |
|
|
Ointment base |
16.14±1.02 |
13.24±1.07 |
19.52±1.67 |
|
Acetone extract ointment(2% w/w) |
25.18±1.64* |
23.48±1.98* |
26.42±2.07 |
|
Acetone extract ointment (5% w/w) |
35.27±2.46* |
28.17±2.19* |
34.27±2.50 |
|
Reference ointment |
39.51±2.61* |
35.24±2.46* |
36.24±2.46* |
n = 6 albino rats per group, value represents Mean ± S.D; *P< 0.01, when compared each treated group with control group in respective parameters
Levels of antioxidant enzymes in granuloma tissue collected after 9th day of wound creation were found increased during by treatment with acetone extract ointment in dose dependent manner. SOD and CAT activity in granulation tissue were found to be significantly increased in 2 & 5% w/w acetone extract ointment when compared with control group (Table 3). GSH concentration (34.27±2.50) in granulation tissue was significantly increased by treatment with 5% w/w acetone ointment treated when compared to control (19.52±1.67).
Discussion and conclusion
Wound healing is a complex process that results after wound contraction, wound closure and restoration of the functional barrier. Dermal wound repair is accompanied by a definite sequence of biological events starting with wound closure and progressing to the repair and remodeling of damaged tissue (Rajan et al., 2004). The present study was aimed to investigate dermal wound healing activity of acetone extract of P. reticulatum lichens by longitudinal incision and dead space wound methods.
It is already reported that P. reticulatum lichens contains usnic acid and flavonoids which are responsible for strong antibacterial activity (Cansaran et al., 2006).Therefore, it could be suggested that the antibacterial and antioxidant effects of acetone extract of P. reticulatum may be due to their contents of flavenoids. Phytochemical studies resulted that acetone extract of P. reticulatum contains flavonoids which have been reported to antibacterial activity and antioxidant effect (Mahajan et al, 1999). Breakdown of collagen liberates free hydroxyproline and its peptides. Measurement of this hydroxyproline used as an index of collagen turnover. The increased hydroxyproline content of skin tissue collected from wound has suggested improved collagen formation leading to rapid healing with concurrent increase in the tensile strength of the treated wounds. The flavonoids may significantly improve the antioxidant enzyme activities (CAT, SOD and GSH) to maintain the balance of the antioxidant inside the body (Chen et al., 2009).
The improved antioxidants level also suggested that acetone extract of P. reticulatum has antioxidant property may be due to presence of flavonoids. Many studies suggested that increased production of reactive oxygen species and lipid peroxidation play a major role in modulation of fibroblast proliferation (Murrell et al, 1990). Dermal wounding creates a depression in the overall antioxidant status that facilitates more vulnerable to oxygen radical attack (Shukla et al, 1997). All these reported findings indicate that antioxidants may play an important role in wound healing.
Reduced glutathione is a potent-free radical scavenger. Depletion of GSH results in enhanced lipid peroxidation. This can cause increased GSH consumption and can be correlated to the increase in the level of oxidised glutathione. Treatment with acetone extract ointment resulted elevation of the GSH levels, which protect the cell membrane from oxidative damage by controlling the redox status of membrane protein. SOD, and CAT are enzymes that inhibits the peroxides and work as antioxidant defenses to an organism (Khan and Akhtar, 2015; Lodhi et al., 2010). The functions of all these enzymes are correlated to each other and decreasing their activities results in the accumulation of lipid peroxides and increased oxidative stress in wounded rats. Treatment of acetone extract also increased the activity of these enzymes and thus may help to overcome free radicals production during severe wounds.
Conclusion
In conclusion, our findings suggest that flavonoid containing acetone extract of P. reticulatum has a potential benefit in enhancing the dermal wound healing. This activity may be due to the free radical scavenging property and antibacterial activity of acetone extract. All finding indicate that in wound healing, antioxidants may play an important role. Flavonoids may help to prevent injury caused by free radicals. Our findings suggest that P. reticulatum lichens have a potential effect in enhancing the dermal wound healing process with antioxidant mechanism.
Acknowledgments
The authors would like to acknowledge to all research colleagues for supporting role in this research work.
Conflict of interest
Author does not have any conflict of interest.
References
Anonymous. 1953. British Pharmacopoeia, General Medical Council, The Pharmaceutical Press: 17, Bloomsbury Square, London, WCI.P.
Balaji P, Hariharan GN. 2007. In vitro antimicrobial activity of Parmotrema praesorediosum. Research Journal of Botany, 2: 54–59.
Beers RF, Sizer IW. 1952. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. The Journal of Biological Chemistry, 195: 133–140.
Cansaran D, Kahya D, Yurdakulola E, Atakol O. 2006. Identification and quantitation of usnic acid from the lichen Usnea species of Anatolia and antimicrobial activity. Journal of Biosciences, 61(11-12): 773-6.
Chen XP, Chen Y, Li SB, Chen YG, Lan JY, Liu LP. 2009. Free radical scavenging of Ganoderma lucidum polysaccharides and its effect on antioxidant enzymes and immunity activities in cervical carcinoma rats. Carbohydrate Polymer, 77(2): 389-393.
Divakar PK, Blanco O, Hawksworth DL, Crespo A. 2005. Molecular phylogenetic studies on the Parmotrema reticulatum (syn. Rimelia reticulata) complex, including the confirmation of P. pseudoreticulatum as a distinct species. Lichenologist, 37(1): 55–65.
Hemalata S, Subramanian N, Ravichandran V, Chinnaswamy K. 2001. Wound healing activity of Indigofera ennaphylla Linn. Indian Journal of Pharmaceutical Sciences, 63(4): 331- 333.
Kuwano H, Yano K, Ohano S, Ikebe M, Kitampura K, Toh Y, Mori M, Sugimachi K. 1994. Dipyridamole Inhibits early wound healing in rat skin incisions. Journal of Surgical Research, 56: 267-270.
Lodhi S, Jain AP, Sharma VK, Singhai AK. 2013. Wound-Healing Effect of Flavonoid-Rich Fraction from Tephrosia purpurea Linn. on Streptozotocin-Induced Diabetic Rats. Journal of Herbs, Spices and Medicinal Plants, 19: 191-205.
Lodhi S, Pawar RS, Jain AP, Jain A, Singhai AK. 2010. Effect of Tephrosia purpurea (L) Pers. on partial thickness and full thickness burn wounds in rats. Journal of Complementary and Integrative Medicine, 7(1): 13.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. 1951. Protein measurement with the Folin Phenol Reagent. Journal of Biological Chemistry, 193: 265-275.
Khan M Sajjad, Akhtar N. 2015. Regulation of Stress: Need of an hour to prevent human health. Asian Journal of Pharmacy and Pharmacology, 1(1): 1-9
Mahajan RT, Chaudhary GS, Chopra MZ. 1999. Screening of some indigenous plants for their possible antibacterial activity. Environmental Bulletin, 15: 61-62.
Misra HP, Fridovich I. 1972. The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. Journal of Biological Chemistry, 247: 3170–3175.
Moron MA, Depierre JW, Mannervick B. 1979. Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochimica et Biophysica Acta, 82: 67–78.
Muller K. 2001. Pharmaceutically relevant metabolites from lichens. Appllied Microbiology and Biotechnology, 56: 9–16.
Murrell GAC, Francis MJO, Bromley L. 1990. Modulation of fibroblast proliferation by oxygen free radicals. Biochemical Journal, 265: 659-665.
Rajan RG, Kumar MV, Rao CV, Shirwaikar A, Mehrotra S, Pushpangadan P. 2004. Healing potential of Anogeissus latifolia for dermal wounds in rats. Acta Pharmaceutica, 54: 331-338.
Shukla R, Rai G, Jain AP. 2015. Preclinical evaluation of Alpinia galanga rhizomes for wound healing activity with reducing oxidative status. Asian Journal of Pharmacy and Pharmacology, 1(2): 65-69.
Sati SC, Joshi S. 2011. Antibacterial activity of the Himalayan Lichen Parmotrema nilgherrense extracts. British Microbiological Research Journal, 1: 26–32.
Sharma BC, Kalikotay S. 2012. Screening of antioxidant activity of lichens Parmotrema reticulatum and Usnea sp. from Darjeeling hills, India. IOSR Jounal of Pharmacy, 2: 54–60.
Shirwaikar A, Jahagirdar S, Udupa AL. 2003. Wound healing activity of Desmodium triquetrum leaves. Indian Journal Pharmaceutical Sciences, 65(5): 461-464.
Shukla A, Rasik AM, Patnaik GK. 1997. Depletion of reduced glutathione, ascorbic acid, vitamin E and antioxidant defense enzymes in a healing cutaneous wound. Free Radical Research, 26: 93-101.
Stanly C, Hag Ali DM, Keng CL, Boey PL, Bhatt A. 2011. Comparative evaluation of antioxidant activity and total phenolic content of selected lichen species from Malaysia. Journal of Pharmacy Research, 4: 2824.
Wilgus TA. 2008. Immune cells in the healing skin wound: influential players at each stage of repair. Pharmacological Research 58, 112–116.
Woessner JF, 1961. The determination of hydroxyproline in tissue and protein samples containing small portion of this imino acid. Archieves of Biochemistry and Biophysics, 193: 440-447.