

Nitish Joshi1, Shivakumar Swamy1, Suresh Janadri*2, Vishakha Patel1
1 Department of Pharmacology, Mallige College of Pharmacy, Bangalore (Karnataka), India.
2 Department of Pharmacology, Acharya B.M, Reddy College of Pharmacy, Bangalore (Karnataka), India
*Address for Correspondence:
Dr. Suresh Janadri, Ph.D
Department of Pharmacology
Acharya & B.M Reddy College of Pharmacy
Achith nagar, Bengaluru - 560 107
Karnataka, India
Tel: +91-9591138301
| Abstract |
Objective: Liver diseases are a large public health problem in the world. Hence, herbal drugs have become increasingly popular and their use is widespread. The present study was carried out to hepatoprotective activity of 60% methanolic leaves and roots extract of Spilanthes radicans on carbontetrachloride induced hepatotoxicity in albino rats. Materials and methods: The 60% methanolic extract of S. radicans leaves and roots at a dose of 500 mg/kg were administered to albino rats per orally for five days. Hepatotoxicity was induced in albino rats by carbontetrachloride administration by subcutaneous on 2nd and 3rd day of treatment. The results of the rats treated with leaves and roots extract of S. radicans were compared with silymarin. Biochemical parameters such as serum SGPT, SGOT, ALP, ACP, direct bilirubin and total bilirubin were estimated to assess the liver functions by semi-autoanlyser using standard biochemical kits. Results: It was found that 60% methanolic leaves and roots extract at a dose of 500 mg/kg exhibited protective effect against hepatotoxicity in rats. The extract shows significant (p>0.01) reduction of SGPT, SGOT, ALP, ACP, Total and direct bilirubin levels compared to CCl4 treated rats. The hepatoprotective activity was furthermore supported by histopathological studies of liver tissue. Conclusion: The study indicates that 60% methanolic extract of S. radicans root exhibits potent hepatoprotective activity than S. radicans leaf.
Keywords: S. radicans, leaves, roots, CCl4, hepatoprotective
| Introduction |
Liver is the largest organ and key organ in regulating homeostasis within the body. It regulates several important functions including protein synthesis, storage and metabolism of fats and carbohydrates, detoxification of drugs and other toxins, metabolism of hormones and excretion of bilirubin. Liver diseases are associated with distortion of these metabolic functions (Ward and Daly, 2005; Wolf, 1999). According to report every year about 20,000 deaths are found due to liver disorders, infections and associated diseases (Lewis and Elvin-Lewis, 1977). Thus to maintain a healthy liver is essential factor for overall health and well beings (Sharma et al., 1991).
Hepatotoxicity is defined as injury to the liver that is associated with impaired liver functions caused by exposure to a drug or another non-infectious agent (Navarro and Senior, 2006). Hepatotoxic agents can react with the basic cellular components and consequently induce almost all types of liver lesions (Navarro and Senior, 2006; Grattagliano et al., 2009). Hepatotoxicity is one of very common aliment resulting into serious debilities ranging from severe metabolic disorders to even mortality (Patel et al., 2008). The available synthetic drugs to treat liver disorders many times cause further damage to the liver and treatment option still is limited. The plant derived natural products such as flavonoids, terpenoids and steroids have received considerable attention in recent years due to their diverse pharmacological properties including antioxidant and hepatoprotective activity (DeFeuids, 2003; Banskota, 2000; Takeoka and Dao, 2003).
Literature surveys we found that plant S. radicans belongs to family Asteraceae and it was claimed to contain a good quantity of alkalamides. Reports showed that alkalamides from other plants possesses hepatoprotective properties (Ramirez-Chavez et al., 2011). The species of S. radicans was reported for organ-protective in experimental rats. However there is no scientific basis or reports in the modern literature regarding S. radicans usefulness as hepatoprotective agent. Therefore the present study was conducted to evaluate the hepatoprotective activity of the leaf and root extract of S. radicans by administration of CCl4 induced hepatic injury in rats.
| Materials and methods |
Collection and authentication of plant material
The leaves and roots of S. radicans were collected from the surrounding gardens of FRLHT (Foundation for Revitalisation of Local Health Traditions) Jarakabande Kaval, Post Attur, via Yehalanka, Bengaluru. It is believed as a weed in agricultural practice. The plant was identified and authenticated by Dr. K. Ravikumar, senior botanist at FRLHT.
Preparation of extract
The leaves and roots of S. radicans were dried under shade and then powdered with a mechanical grinder. The powder was stored in an air tight container for further use. The coarse powder was extracted directly with 60% methanol (hydro-alcoholic extract) after defating with petroleum ether by soxhelet extraction. The extracts were concentrated under reduced pressure and stored in a container until further use.
Experimental animals
Wistar albino rats (weighing 150-200 g) and albino mice (weighing 20-25 g) of either sex were used in this study. They were procured from Bioneeds Private Limited, Tumakur. The animals were acclimatized for one week under laboratory conditions. They were housed in polypropylene cages and maintained at 27°C ± 2°C under 12 hours dark / light cycle. They were fed with standard rat feed and water ad libitum was provided. Ethical clearance for handling the animals was obtained from the Institutional Animal Ethical Committee (RRCOP/IAEC.CLEAR/2011-12/APRIL) prior to the beginning of the research work.
Acute toxicity
The maximum lethal dose of S. radicans (5000 mg/kg, p.o.) hence 1/10th of lethal dose was taken as effective dose (500 mg/kg body weight) for the 60% methanolic extract of leaves and roots of S. radicans for hepatoprotective activity.
Carbontetrachloride induced hepatotoxicity
Adult male albino rats (150-200 g) were used for the study. Animals were divided into five groups of six animals each. Group I served as vehicle control was received vehicle per orally for five days. Group II served as hepatotoxic control. Group III and IV were administered with S. radicans leaves and roots extract at a dose of 500 mg/kg p.o. for five days. Group V was treated with the standard drug Silymarin at a dose of 25 mg/kg, p.o. for five days. On 2nd and 3rd day of the treatment group I received liquid paraffin (1 ml/kg s.c.), group II was received CCl4: liquid paraffin (1:1) at a dose of 2 ml/kg s.c., group III and IV received 500 mg/kg leaves and roots extract of S. radicans respectively and group V received Silymarin (25 mg/kg, p.o.) 30 minutes after the vehicle administration. At the end of the experimental period, all the animals were sacrificed by cervical dislocation and blood samples were collected by cardiac puncture for biochemical estimation (Gini and Muraleedhara, 2008).
Biochemical estimation
The blood samples were collected without any anticoagulant and were allowed to clot for 45 minutes at room temperature. The blood was centrifuged at 3000 rpm for 15 minutes at 30°C. The obtained serum was stored at 4°C for the estimation of SGOT, SGPT, ALP, ACP, direct and total billirubin. These estimations were performed according to the standard biochemical kits.
Histopathology
Small piece of liver tissue was collected in 10% formalin for proper fixation. These tissues were processed and embedded in paraffin wax. Section of 5-6 microns in thickness were cut and stained with hematoxylin and eosin. All the sections of the tissues were examined under microscope for the analyzing the altered architecture of the liver tissue.
Statistical analysis
Data were expressed as mean ± standard error of mean. Statistical comparisons were made by using one-way ANOVA followed by Dunnet test. The results were considered statistically significant if P < 0.05, P < 0.01.
| Results |
Phytochemical screening
The leaves and roots 60% methanolic extract of S. radicans were subjected to preliminary phytochemical investigations, it was found that, the leaves extract contains carbohydrates, resins, alkaloids, fixed oils, terpenoids, flavonoids, tannins, proteins and amino acids and the roots extract contain carbohydrates, glycosides, resins, tannins, fixed oils, terpenoids, flavonoids, proteins and amino acids.
Carbontetrachloride induced hepatotoxicity
The result of this study were represented in Table 1, it is clearly evident that the CCl4 caused significant (p<0.01) elevation of liver serum markers. The 60% methanolic leaf extract of S. radicans shows significant reduction of SGPT (p<0.01), SGOT, direct bilirubin (p<0.05) but statistically non-significant reduction of ALP, ACP and total bilirubin compared to silymarin. The 60% methanolic root extract of S. radicans shows significant reduction of SGPT, SGOT, direct bilirubin, total bilirubin (p<0.01) and ALP, ACP (p<0.05) compared to silymarin. The standard drug silymarin (25 mg/kg, p.o.) exhibited significant reduction of liver serum markers compared to heptotoxic control. In contrast the groups treated with 60% methanolic leaves and roots extract of S. radicans at a dose of 500 mg/kg prevented the hepatotoxicity in rats.
Table 1 Effect of 60% methanolic leaves and roots extract of S. radicans in carbontetrachloride induced hepatotoxicity in rats
|
Groups |
Treatment |
Serum profiles |
|||||
|
SGPT (IU/L) |
SGOT (IU/L) |
ALP (IU/L) |
ACP (IU/L) |
D. Bilirubin (mg/dl) |
T. Bilirubin (mg/dl) |
||
|
I |
Vehicle control (1 ml/kg, s.c) |
46.48 ± 2.34** |
69.58 ± 3.62** |
153.03 ± 2.80** |
14.95 ± 0.698** |
0.23 ± 0.02** |
0.52 ± 0.07* |
|
II |
CCl4+liquid paraffin (2 ml/kg, s.c) |
178.38 ± 5.77** |
282.6 ± 19.0** |
243.65 ± 7.69** |
43.85 ± 2.43** |
1.82 ± 0.05** |
2.08 ± 0.08** |
|
III |
60% Methanolic leaf extract of S. radicans (500 mg/kg, p.o.) |
126.55 ± 2.11** |
168.55 ± 5.19* |
216.6 ± 4.27ns |
34.5 ± 1.58ns |
0.93 ± 0.05* |
1.90 ± 0.08ns |
|
IV |
60% Methanolic root extract of S. radicans (500 mg/kg, p.o.) |
114.95 ± 3.31** |
123.3 ± 3.70** |
199.71 ± 2.58* |
26.61 ± 2.03* |
0.68 ± 0.02** |
1.66 ± 0.07** |
|
V |
Silymarin (25 mg/kg, p.o.) |
104.38 ± 2.66** |
108 ± 2.65** |
186.75 ± 2.52** |
21.41 ± 1.01* |
0.73 ± 0.04** |
1.21 ± 0.04** |
All the values were expressed in Mean ± SEM (n=6). The statistical analysis was carried out using one way ANOVA. Significant after analysis of variance (ANOVA) followed by Dunnett multiple comparison test. *P<0.5, **P<0.01, ns-Non-Significant when compared to CCl4 control group
Histopathology
Histopathological studies were performed on both healthy rats, diseases rats and the observations in Group-I vehicle control, photomicrograph showed normal cellular architecture in the hepatocyte (Figure 1A). Group-II hepatotoxic control, histopathology of liver suggests severe damage to the hepatocyte (Figure 1B). Group-III, 60% methanolic leaf extract of S. radicans (500 mg/kg, p.o.) shows focal area of necrotic changes and mitotic division in hepatocyte (Figure 1C). Group-IV, 60% methanolic root extract of S. radicans (500 mg/kg, p.o.) shows less focal area of necrotic changes and mitotic division in hepatocyte (Figure 1D). Group-V, Silymarin (25 mg/kg, p.o.) shown the restoration and normal arrangement of hepatocytes was observed (Figure 1E).
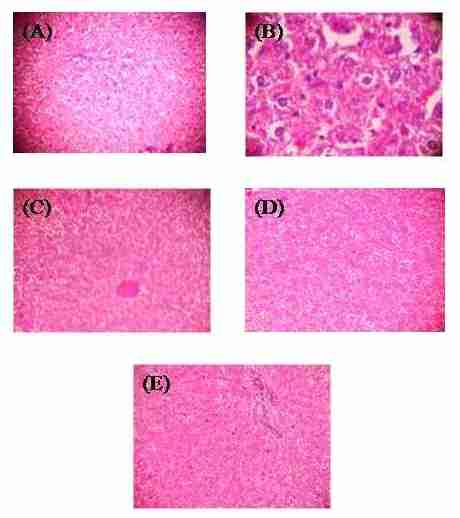
Figure1. Histopathological evaluation of rats liver sections. Normal cellular architecture in the hepatocyte (A), severe damage to the hepatocyte (B), focal area of necrotic changes and mitotic division in hepatocyte (C), less focal area of necrotic changes and mitotic division in hepatocyte (D), normal arrangement of hepatocyte (E). Hematoxylin and eosin staining, 10x magnification
| Discussion and conclusion |
CCl4 has been demonstrated to cause acute hepatotoxicity with necrotic and apoptotic hepatocellular injury and impairment of liver function (Kovalovich et al., 2001). The mechanism of CCl4 injury involves oxidative damage by metabolism of CCl4 to CCl3• in hepatocytes which ultimately results in cell death with accumulation of lipid peroxidation and intracellular calcium ions and triggers secondary damage from the inflammatory process (Medina and Moreno-Otero, 2005). The ethanol extract of the whole plant of S. ciliate was screened for its hepatoprotective of paracetamol induced hepatotoxicity in rats by reduced the serum liver enzymes (Ramirez-Chavez et al., 2011).
The present finding can be supported by a study which correlated with presence of several terpenoids, flavonoids and proteins. To sum up the above discussion, altogether it is proved that 60% methanolic extract of S. radicans extract, inhibited CCl4 induced hepatotoxicity by regulating various biochemical parameters such SGPT, SGOT, ALP, bilirubin and liver metabolites. Histopathology also supported for the study to restore the normal architecture of hepatocytes. The roots and leaves of S. radicans possess significant hepatoprotective property. The roots extract of S. radicans has shown more hepatoprotection than the leaves extract because they had shown a significant restoration of biochemical markers in CCl4 induced hepatotoxicity model. Hence, the degree of protection is more in roots extract of S. radicans as compared to leaves extract of S. radicans.
Acknowledgement
The authors are thankful to Dr. A.C. Raghuram and Dr. A.C. Sreeram, management of Mallige education foundation for provided necessary facility to carry out the research in laboratory.
Conflict of interest
There is no conflict of interest in the present study.
| References |
Banskota AH, Tezuka Y, Adnyana IK, Xiong Q, Hase K, Tran KQ. 2000. Hepatoprotective effect of Commbretum quadrangulare and its constituents. Biological and Pharmaceutical Bulletin, 23:456-560.
Chopra RN, Nayara SL, Chopra IC. 1956. Glossary of Indian Medicinal Plants, Council of Scientific and Industrial Research, New Delhi, pp 168-169.
DeFeuids FV, Papadopoulos V, Drieu K. 2003. Ginko biloba extracts and cancer a research area in its infancy. Fundamental Clinical Pharmacology, 17:405‐417.
Gini CK, Muraleedhara GK. 2008. Anti-oxidant activity of Aulosira fertilisima on CCl4 induced hepatotoxicity in rats. Indian Journal of Experimental Biology, 46;52-59.
Grattagliano I, Bonfrate L, Catia VD, Wang HH, Wang DQH, Portincasa P. 2009. Biochemical mechanisms in drug-induced liver injury. World Journal Gastroenterology, 15:4865-4876.
Kovalovich K, Li W, DeAngelis R, Greenbaum LE, Ciliberto G, Taub R. 2001. Interleukin-6 protects against Fas-mediated death by establishing a critical level of anti-apoptotic hepatic proteins FLIP, Bcl-2, and Bcl-xL. Journal of Biological Chemistry, 276: 26605–26613.
Lewis HW, Elvin-Lewis MPH. 1997. Pants affecting man’s Health, John Wiley and Sons, New York, pp 217-218.
Medina J, Moreno-Otero R. 2005. Pathophysiological basis for antioxidant therapy in chronic liver disease. Drugs, 65(17):2445-2461.
Navarro VJ, Senior JR. 2006. Drug-related hepatotoxicity. New England Journal Medicine, 354:731-739.
Patel RK, Patel MM, Patel MP, Kanzaria NR, Vaghela KR, Patel NJ. 2008. Hepatoprotective activity of Moringa oleifera Lam. Fruit on isolated rat hepatocytes. Pharmacognosy magazine, 4:118‐123.
Ramirez-Chavez E, Molina-Torres J, Rios-Chavez P. 2011. Natural distribution and alkamides production in Acmella radicans. Emirates Journal of Food and Agriculture, 23(3): 275-282.
Sharma A, Chakraborti KK, Handa SS. 1991. Anti-hepatotoxic activity of some Indian herbal formulations as compared to Silymarin. Fitoterapia, 62:229-235.
Takeoka GR, Dao LT. 2003. Antioxidant constituent of almond (Prunus dulcis (Mill.) D.A.Webb) hulls. Journal Agricultural Food Chemistry, 51:496‐501.
Ward FM, Daly MJ. 2005. Liver disease, In: Roger walker, Clive Edwards, Clinical Pharmacy and Therapeutics, 3 rd edition, Churchil Livingstone, New York, pp 209.
Wolf PL. 1999. Biochemical diagnosis of liver disease. Indian Journal of Clinical Biochemistry, 14:59-90.