

Sri Padmavathi School of Pharmacy, Tiruchanoor, Tirupathi-517503, Andhra Pradesh, India
Abstract
Objectives: It is to demonstrate that a heterocyclic derivatives acts as magic moiety in antagonizing numerous undesirable diseases. Intact the 1,5-Benzothiazepine is a six member benzene ring condensed to a hetero seven member ring shows a various pharmacological properties in the field of medicine. Material and methods: The molecular design for the depicted compounds is identified for its ADME properties and drug likeliness is studied using “Mcule” a software tool available online. Further the selected compounds are prepared in wet lab, a series of substituted Benzothiazepine are prepared by cyclo condensation of α, β-unsaturated ketones (chalcones) with O-amino thiophenol under the influence of glacial acetic acid. Structural characterization and in-vitro anti-inflammatory (by Protein denaturation inhibition) and anti-oxidant (by H2O2 scavenging activity) activity are performed with slight revising the procedure. From the obtain result the docking process made on target Leukotriene-C4 synthase (LTC4S) enzyme need for synthesis of leukotriene C4 from arachidonic acid to produce inflammatory response on bronchial asthma and another target Peroxisome proliferator-activated receptor alpha (PPARA) need for metabolism of TG and fatty acid to generate energy, cholesterol and LDL synthesis. Results: Some of the compound code BTP-3 and BTP-5 shows appreciable ligand-target interaction with glide score of -11.2 and -10.8 ΔG, kcal/mol on PDB-2uuh (LTC4S) compound BTP-5 and BTP-2 shows glide score of -11.8 & -10.8 ΔG, kcal/mol on PDB- 1kkq (PPARA) target. All compounds shows good probability of physico-chemical property as per “Lipinski rule of five”. Compound BTP-5, 6 and 7 shows significant in-vitro activity studies on anti-inflammatory and anti-oxidant activity as compared with standard drug as show in respective histogram. Conclusion: 1, 5-Benzothiazepine derivatives shows a prominent activity by in-silico and in-vitro model, from this it is considered as potent pharmacophore moiety for drug development studies and used for in-vivo evaluation studies by standard methods.
Introduction
Thiazepines a heterocyclic compound is a lead molecule it acquires a prime position in field of drug designing in medicinal chemistry for its enormous biological activities (Fernandes, 2017). The Thiazepines nucleus is found as a building block in synthesis of bioactive pharmaceuticals. It alone have various activity like anti-arrhythmic, antispasmodic, anti-anginal, antimicrobial, analgesic, anticancer, anti-inflammatory, antidepressant, anticonvulsant, anti-hyperglycemic, antipyretic and antioxidant so on (Kaur et al., 2016). 2,3-dihydro1,5- Benzothiazepine is an one of the versatile six member benzene was condensed with seven memberd thiazepine as heterocyclic pharmacophore moiety. The plausible benzo-condensed derivatives are 1,4-, 4,1- and 1,5-benzothiazepines derivatives (Figure 1a)(Khairy and El-Bayouki, 2013). The most common method of synthesizing 1,5-benzothiazepine molecule is thia-Michael addition of α-amino thiophenol with different chalcones in weak acidic condition undergoes cyclo-condensation (Kumar et al., 2015). The keen interest to synthesis of new Benzothiazepine derivatives due to its improve in biological potential (Figure 1b).
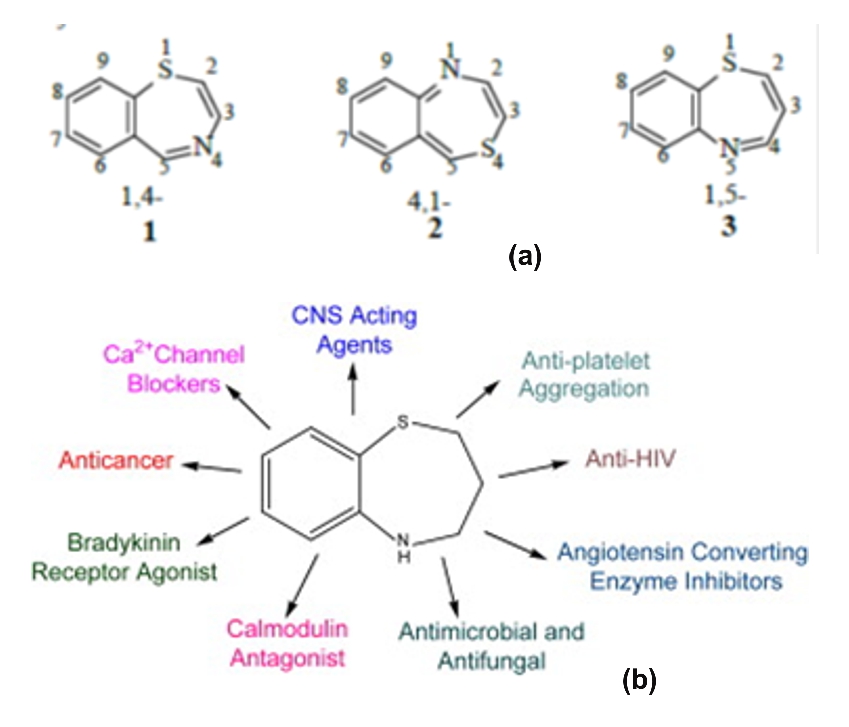
Figure 1. (a) Benzothiazepine derivatives and, (b) biological potential of benzothiazepine derivatives
Moreover, if thiazepine nucleus was substituted with 2,4-disubstituted with aryl, hetero aryl or aryl substituent’s may be hydrated derivatives shows a extent in their pharmacological activity. The 1,5-benzothiazepine and its derivatives are pertain for lead molecule because of its active function against various targets (Rao et al., 2016). The foremost use of 1,5 Benzothiazepine clinically approved was Diltiazem, followed by Clentiazem for Cardio protective agent (Zhang et al, 2010). Few of 1,5-benzothiazepine derivatives were also clinically ruled for CNS disorders like Thiazesim and Quetiapine fumarate (Takada et al, 2012). 1,5-benzothiazepine moiety is a favored group of pharmacophore, as compounds bearing this structural unit possess a broad spectrum of biological activities such as anticancer (Prasad et al, 2018), antimicrobial, V2 arginine vasopressin receptor antagonist, anti arrhythmic, Hypolipidemic, Bradykinin agonist, vasodilator, Anticholinesterase inhibitor, anticonvulsant, HIV-1 reverse transcriptase inhibitor, Glycogen synthase kinase-3β inhibitor, Ca+2 Channel blocking, anti-angina, anti HIV and Squalene synthetase inhibitor so on (Raghavendra et al., 2015). From the varied list of reviewed gives us a information that 1,5-benzothiazepine derivatives with different pharmacological effects which fancier to prepare and evaluate in this present work.
Materials and methods
The software tool Mcule is an online available of version 3 Docking vina, the physic chemical parameters and 3D docking score are determined from it. The chemicals employed in the titled work were purchased from Otto chemicals, Hi-media, Merck and SD fine chemicals of high grade. The melting point for the synthesized compounds were determined by open capillary method which are incorrect, all the synthesized compounds are characterized and identified by FT-IR by KBr method using SHIMADZU IR-Spirit FTIR spectrophotometer. Few compounds are characterized by 1H-NMR by VARIAN MERCURY YH-400 using TMS as internal standard in DMSO-d6 solvent and Mass by EI-MS for confirmation studies.
Experiment procedure
Step I: Synthesis of substituted α, β-unsaturated ketones (chalcones)
The detailed method of preparation of chalcones by claisen-schmidt condensation with slight revising in using PEG-400 as reversible catalyst (Balaji et al., 2014).
Step II: Synthesis of 1,5-Benzothiazepine from substituted chalcones
The 1,5-benzothiazepine was prepared by reacting 1: 2 ration of above prepared chalcones of 0.01M with O-amino thiophenol of 0.02M in a 250ml round bottomed flask with 30ml of absolute alcohol. Connect this RB flask to a set of condenser places on water bath and heated at 850c, after 15min of reaction add 0.5ml of glacial acetic acid as catalyst and allow the condensation for >5hrs with occasional shaking on water bath. The end of the reaction for individual compound was confirmed by performing TLC to obtain single spot. Later cool the mixture and transfer the content to beaker contain crushed ice, stir vigorously to separate desired compounds, dried and re-crystallized it with methanol (Shah et al., 2011).
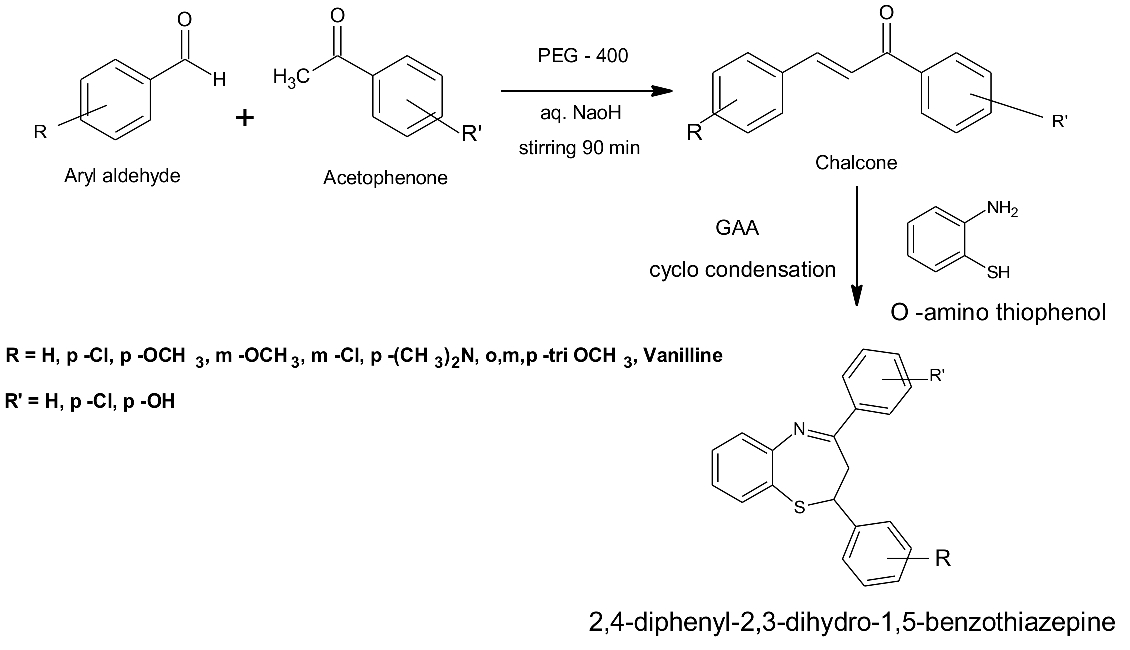 Figure 2. Contrived scheme for synthesis of 1,5-Benzothiazepine
Figure 2. Contrived scheme for synthesis of 1,5-Benzothiazepine
Table 1. Physicochemical properties of 1,5 Benzothiazepine
|
S. No. |
R |
R1 |
Mol Form |
M.P oC |
Rf Value |
Mol Wt |
clog P |
Hyd-B.A |
Hyd-B.D |
Docking score (ΔG, kcal/mol) |
|
|
2uuh |
1kkq |
||||||||||
|
BTP-1 |
p- OCH3 |
p-Cl |
C22H18ClNOS |
121 |
0.68 |
379.9 |
3.94 |
2 |
1 |
-9.8 |
-8.6 |
|
BTP-2 |
p-Cl |
H |
C21H16ClNS |
137 |
0.70 |
349.8 |
4.21 |
1 |
0 |
-6.3 |
-10.8 |
|
BTP-3 |
m-OCH3 |
p-OH |
C22H19NO2S |
144 |
0.58 |
361.4 |
3.19 |
3 |
1 |
-11.2 |
-9.3 |
|
BTP-4 |
p- OCH3 |
H |
C22H19NOS |
153 |
0.54 |
345.4 |
4.48 |
2 |
0 |
-8.5 |
-9.7 |
|
BTP-5 |
m-OH, p- OCH3 |
p-Cl |
C22H18 ClNO2S |
102 |
0.52 |
395.9 |
4.82 |
3 |
1 |
-10.8 |
-11.8 |
|
BTP-6 |
3,4,5-tri OCH3 |
p-OH |
C24H23NO4S |
98 |
0.63 |
421.5 |
3.21 |
5 |
1 |
-7.8 |
-10.2 |
|
BTP-7 |
m-Cl |
p-OH |
C21H16ClNOS |
108 |
0.67 |
365.8 |
3.83 |
2 |
1 |
-6.3 |
-7.2 |
|
BTP-8 |
p-(CH3)2N |
p-Cl |
C23H21ClN2S |
117 |
0.66 |
393.9 |
4.91 |
2 |
0 |
-9.3 |
-9.6 |
|
BTP-9 |
H |
H |
C21H17NS |
104 |
0.59 |
315.4 |
3.62 |
1 |
0 |
-6.8 |
-9.1 |
Mobile phase: Chloroform: Ethyl acetate (8.5: 1.5); PDB- (2uuh) - Leukotriene-C4 synthase (LTC4S) & PDB-(1kkq) - Peroxisome proliferator-activated receptor alpha (PPARA); HBA/HBD – Hydrogen bond acceptor/donor
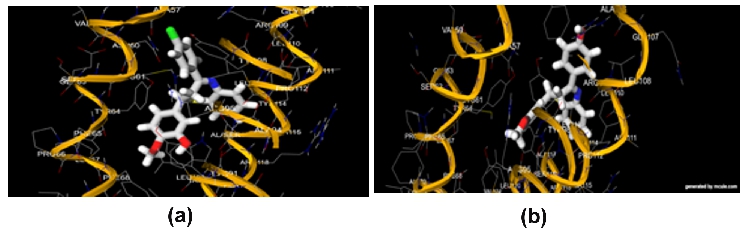
Figure 3. Docking snapshot of the best compound on LTC4S enzyme: (a) BTP-3 (Docking score -11.2 kcal/mol); (b) BTP-5 (Docking score -10.8 kcal/mol)
Figure 4. Docking snapshot of the best compound on PPARA enzyme: (a) BTP-5 (Docking score -11.8 kcal/mol); (b) BTP-2 (Docking score -10.8 kcal/mol)
Figure 3 and 4 showing docking view of compound BTP-3,5 shows good Glide score of -11.2 and -10.8 kcal/mol inhibition of the enzyme Leukotriene-C4 synthase (LTC4S) is an MAPEG metabolism involved in synthesis of Leukotriene which act as mediator for anaphylaxis and inflammatory component in bronchial asthma. And BTP-5,2 shows Glide score of -11.8 & -10.8 kcal/mol in inhibition of Peroxisome proliferator-activated receptor alpha (PPARA) enzyme needed for metabolism of triglycerides in liver for energy production, ketones bodies synthesis, LDL synthesis and cholesterol biosynthesis. So from this the Benzothiazepine compounds show plausible inhibition activity over human enzymes with good binding to its protein surface.
Spectroscopic characterization data of 1,5-Benzothiazepine its derivativestabl
BTP- 2,4-diphenyl-2,3-dihydro-1,5-benzothiazepineand its derivatives
FT-IR spectrophotometer (by KBr pellet method) υmax (cm-1): 810 (C-Cl), 2908 (Ar-OCH3), 1540 (C=C), 735 (C-S-C), 3080 (C-H), 1715 (C=N), 3360 (Ar-OH), 1280 (C-N).
Proton Nuclear magnetic Spectroscopy by using TMS as internal standard in DMSO-d6 solvent δ ppm - 4.6 (s, 1H, CH-S), 3.5 (s, 3H, OCH3), 7.3-7.6 (m, 6H, Benzothiazepine), 7.8-7.9 (m, 8H, aromatic ring), 9.8 (s, 1H, OH), 2.9-3.2 (m, 6H, N(CH3)2).
Electron Induced-MS: 393.9(cal) 392.88(obt), 421.5(cal) 422.2(obt), 365.8(cal) 366.2(obt), 349.8(cal) 350.8(obt).
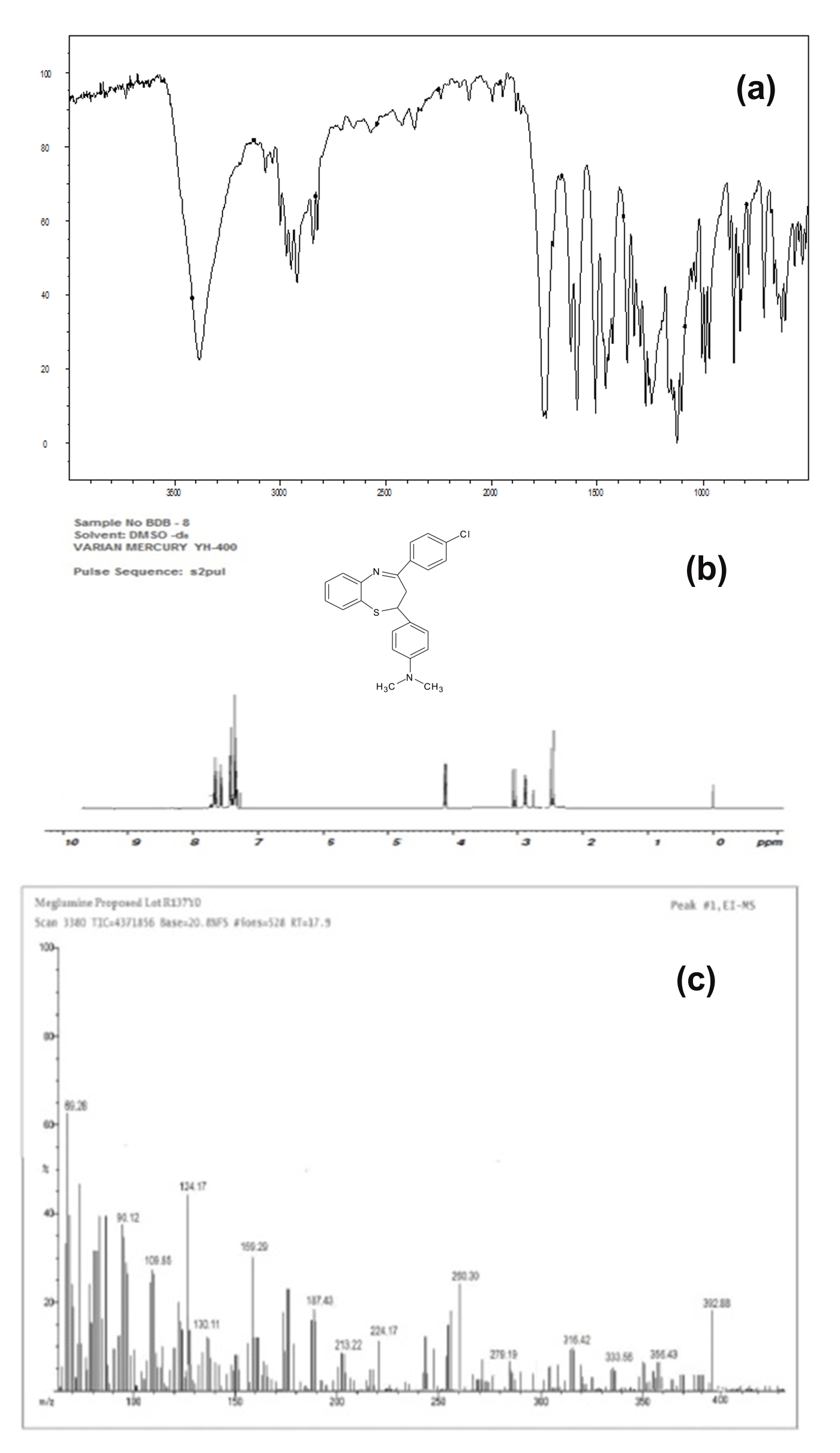
Figure 5. Sampling of BTP-8: 4-[4-(4-chlorophenyl)-2,3-dihydro-1,5-benzothiazepin-2-yl]-N,N-dimethylaniline (a) FTIR spectra; (b) NMR Spectra; (c) MS Spectra
Biological activity
In-vitro anti-inflammatory activity by inhibition of protein denaturation method (Balaji et al, 2012)
To evaluate the anti-inflammatory activity for synthesized 1,5-benzothiazepine its derivatives, by the standard protocol was used with applicable modifications. A volume of 5 ml of different concentration like 100, 200 & 400 μg/ml of test & Diclofenac sodium of 400 μg/ml taken separately was homogenized with 0.2 ml of 1% mM of bovine serum albumin in sufficient quantity of phosphate buffered saline (PBS, pH 6.4) upto the mark was taken, and incubated at 27°C for 15 minutes. The mixture of distilled water, BSA and PBS of 5ml as in the control tube. Denaturation of the proteins was caused by placing the mixture in a water bath for 10 minutes at 70°C. The mixture was kept for cooling in ambient room temperature, and the activity of each mixture was measured at 660 nm. Each test was done three times. The following equation (1) was used to calculated inhibition of protein denaturation percentage.
 ----------- (1)
----------- (1)
(a) In-vitro anti-inflammatory activity by Protein denaturation inhibition method
In-vitro antioxidant by hydrogen peroxide (H2O2) scavenging model (Seelolla et al., 2014)
The hydrogen peroxide scavenging ability of test compound was determined according to the method of Ruch et al. (2014) A solution of 30% hydrogen peroxide 40mM was prepared in phosphate buffer (pH-7.4) 100, 200 & 400 μg/ml conc. of the all test compounds. Standard L-ascorbic acid is prepared by dissolving in suitable solvent. In a test tube add 1ml of different concentrations of test and standards are taken. To this 3.4 ml of phosphate buffer were added, 0.6ml of prepared hydrogen peroxide solution 40mM is added. Keep the mixture for 10 min at room temperature or incubate at 37 0C for 5 min. The absorbance value of the reaction mixture was recorded at 230nm. The percentage of scavenging of hydrogen peroxide was calculated by using equation (2).
 ………….. (2)
………….. (2)
Figure 6. (a) In-vitro anti-inflammatory activity by Protein denaturation inhibition method; (b) in-vitro anti-oxidant activity by hydrogen peroxide (H2O2) scavenging model
Results and discussion
In-Silico Prediction
In-silico is an external method which has a significant impact on a number of sub disciplines in supporting the toxicological study of the lead molecule. It was performed before the wet practical process has to be done. It is a computational method by applying the software tool we can predict the drug likeliness of the chemical compound, its pharmacokinetics and toxicity criteria on identified targets done by molecular docking tools. An open access Molecular docking software Mcule of version 3 has used to detect the physico chemical property of the sketched 1,5-benzothiazepine compounds and study done on Lipinski rule of five for drug likeliness nature and docking model on selected target i.e Leukotriene-C4 synthase (LTC4S) of human whose PDB is 2uuh an enzyme involved in producing inflammatory component in lungs and other colonel region of human body and another target Peroxisome proliferator-activated receptor alpha (PPARA) human based enzyme whose of PDB 1kkg, this enzyme mainly found in liver, works to metabolize the lipids at low energy condition to increase energy production, ketones bodies biosynthesis, cholesterol biosynthesis and LDL synthesis in body. The main reason to target these enzyme to predominate the role of 1,5-benzothiazepine in treating inflammation and hyperlipidemic treatment. Compound BTP- 3 & 5 shows good docking pose with Glide score of -11.2 & -10.8 kcal/mol on LTC4S, compound BTP- 5 & 2 shows Glide score of -11.8 & -10.8 kcal/mol. This shows the synthesis of Benzo1,5-thiazepine plays good pharmacophore nature in field of therapeutic studies.
Synthesis and characterization
The synthesis of 1,5-benzothiazepine from substituted chalcones were done by preparing the individual chalcones by reacting substituted aryl aldehyde with substituted acetophenone in aqueous ethanol in presence of alkali catalyst, to this reaction PEG-400 which acts as biodegradable, increasing the solubility property of reacting mixture and speed up the reaction rate, which can easily separated as it is water soluble. From the previous experimentation the time taken for chalcones formation is two folds high as compared with presence of PEG-400 which finish the product formation with in 90min, the yield of the product is also appreciable. The obtain chalcones are further treated with O-amino thiophenol of 1:2 ration in presence of weak acid called glacial acetic acid as catalyst in alcohol media undergoes thia-Michael addition of the thiol group to the enone functionality of the chalcones to form the intermediate followed by cyclo condensation to produce Benzo1,5-thiazepine. Which are purified and evaluated for structural characterization by FT-IR using KBr pellet method for determining regions like C=C at 1540 bending, CH at 3080 stretching, functional group like –OH at 3360, -OCH3 at 2900, C=N at 1715 and C-N at 1200 cm−1. 1H-NMR done to determine the δ value in ppm - 4.6 (s, 1H, CH-S), 3.5 (s, 3H, OCH3), 7.3-7.6 (m, 6H, Benzothiazepine), 7.8-7.9 (m, 8H, aromatic ring), 9.8 (s, 1H, OH), 2.9-3.2 (m, 6H, N(CH3)2). The exact mass of the compounds are obtain by EI-MS whose compounds shows appropriate mass value in m/z. From the above spectral data the plausible structure of the 1,5-benzothiazepine and its derivatives are formed and confirmed.
Biological evaluation
All the above synthesized compounds are preliminary evaluated for anti-inflammatory activity by protein denaturation inhibition and antioxidant for hydrogen peroxide scavenging activity. The above In-vitro studies are performed from a standard procedure with slight modification in it. The compound BTP- 5 >7>9 shows a prominent anti-inflammatory activity as compared with standard value shown by Diclofenac sodium at three different concentration of 100,200 &400 μg/ml. Were the rest of the compounds shows moderate to good activity of protein denaturation inhibition. Compound BTP- 6>3>7 shows potent activity against neutralizing the H2O2 to water. Here by the compound shows appreciable antioxidant activity against scavenging of hydrogen peroxide as compared with standard L-ascorbic acid.
Conclusion
In silico computational models were the fast & novel access process of drug discovery and development. A quickened development in the field of pharmaceutical sciences, a increasing demand has come up regarding the evolution of reliable techniques for predicting the pharmacokinetic properties of the new drugs to reduce preparation costs and time which involved in production of new drugs. The various physicochemical properties which predict by In-silico process such as solubility, hydrogen bond donate/accept, lipophilicity, as well as the permeability across the biological membranes. By this the wet lab procedure are applied to prepare some series of 1,5-Benzothiazepine its derivatives has succeed and preliminary investigation also performed to know its strength and considerable. 1,5-benzothiazepine contain side chain of OH, OCH3 and Cl at Para position of benzene shows plausible activity. Based on the above results selected compounds are used for In-Vivo evaluation in further studies.
Conflict of interest
The author has declared there is no conflict of interest as the complete research work was carried out with self finance.
References
Balaji PN, Lakshmi LK, Mohan K. 2012. In-vitro anti-inflammatory and antimicrobial activity of synthesized some novel pyrazole derivatives from coumarin chalcones. Der Pharmacia Sinica, 3(6):685-689.
Balaji PN, Lavanya KC. 2017. Evaluation of in vitro anti-inflammatory activity for novel imine derivatives from acetyl coumarin by catalyst-free green solvent. Chemical Science Transactions, 6(3):480-484.
Balaji PN, Ranganayakulu D, Subba Reddy GVS. 2017. Synthesis, in vitro evaluation for anthelmintic and antimicrobial activity for the novel thiazolidine-4-one incorporate substituted chloro-quinoline. Asian Journal of Pharmacy and Pharmacology, 3(1): 9-15.
Balaji PN, Ranganayakulu D. 2014. Anthelmintic and Anti-microbial activities of synthesized heterocyclic pyrazole and its derivatives from fluoro substituted hydrazino benzothiazole. International Journal of Pharm Tech Research, 6(7): 1970-1975.
Balaji PN, Reddy GVS. 2017. Design, synthesis, characterization of substituted 3- (benzothiazole)-2-(phenyl)-thiazolidin-4-one heterocyclic compounds and its biological evaluation. Journal of Global Trends in Pharmaceutical Sciences, 8(4):4557 – 4566.
Fernandes JPS. 2017. The Importance of the Medicinal Chemistry. American Journal of Pharmaceutical Education, 82(2): 6083.
Gangadhara S, Prasad C, Venkateswarlu P. 2014. Synthesis, Antimicrobial and Antioxidant Activities of Novel series of Cinnamamide Derivatives having morpholine moiety. Medicinal Chemistry, 4(12): 778-783.
Inada Y, Tanabe M, Itoh K, Sugihara H, Nishikawa K. 1988. Inhibition of angiotensin converting enzyme by (R)-3-(S)-1-carboxy-5-(4-piperidyl) pentyl amino-4-oxo-2,3,4,5-tetra- hydro-1,5-benzothiazepine-6-acetic acid (CV-5975), a non-sulfhydryl compound. Japanese Journal of Pharmacology, 48:323-330.
Kaur R, Singh K, Singh R. 2016. 1,5-Benzothiazepine: Bioactivity and targets. Chem. Biol. Lett. 3(1), 18-31.
Khairy AM, El-Bayouki. 2013. Benzo[1,5]thiazepine: Synthesis, Reactions, Spectroscopy, and Applications. Organic Chemistry International. Article ID 210474: 71.
Khan AJ, Baseer MA, Dhole JM, Shah S. 2011. Synthesis, experimental studies of the antimicrobial potential of some novel 1, 5- benzothiazepine derivative. International Journal of Pharmaceutical Science and Research, 2:2619-2622.
Kumar A, Renuka N, Raghavendra KR. 2015. New insights to the chemistry of benzothiazepines-An overview. International Journal of Basic and Applied Chemical Sciences, 5(1): 79-88.
Lauraperez-Benito, Henrik Keranengary, Tresadern. 2018. Structure Based Drug Design in Drug Discovery: An outline. Pharmacy World and Science, 1(2): 666-680.
Paul DL, Brain S. 2007. The Influence of drug-like concepts on decision-making in medicinal chemistry. Nature Reviews Drug Discovery, 6(11):881-90.
Petrova K, Petrov O, Antonova A, Kalcheva V. 2003. Synthesis of benzo[b]imidazo[1,5-d][1,5]-thiazepines. Derivatives of a novel ring system. Synthetic Communications, 33(24):4355–4366.
Rao CHMMP, Yejella RP, Rehman SA. 2018. Novel series of 1, 5 Benzothiazepine skeleton based compounds as anti-cancer agents – In silico and MTT assay based study. Journal of Peer Scientist, 1(2): 1-10.
Rao CHMMP. 2016. Synthesis, characterization and anti bacterial and cytotoxic Studies of novel 1, 5 benzothiazepines from chalcones of 2, 4 di fluoro acetophenone, Asian Journal of Research in Chemistry and Pharmaceutical Sciences, 4(4): 128 – 139.
Rao CM, Yejella RP, Rehman RS, Basha SH. 2015. Molecular docking based screening of novel designed chalcone series of compounds for their anti-cancer activity targeting EGFR kinase domain. Bioinformation 7: 322.
Shi QW, Li LG, Huo CH, Zhang ML, Wang YF. 2010. Study on natural medicinal chemistry and new drug development. Chinese Traditional and Herbal Drugs, 41:1583–1589.
Tabata H, Wada N, Takada Y, Nakagomi J, Miike T, Shirahase H, Oshitari T, Takahashi H, Natsugari H. 2012. Active Conformation of Seven-Membered-Ring Benzolactams as New ACAT Inhibitors: Latent Chirality at N5 in the 1,5-Benzodiazepin-2-one Nucleus. Chemistry -A European Journal, 18:1572-1576.