

Veerpal Kaur,1 Supandeep Singh Hallan,1 Nidhi,1 A.N. Kalia,2 Neeraj Mishra1*
1Department of Pharmaceutics, ISF College of Pharmacy, Moga, Punjab, India
2Department of Pharmacognosy, ISF College of Pharmacy, Moga, Punjab, India
*Corresponding Author
Dr. Neeraj Mishra
Department of Pharmaceutics, ISF College of Pharmacy,
Ferozepur Road, Ghal Kalan
Moga, and Punjab, India Postal Code: 142001,
Tel Nos. 01636-324200, Fax No. 01636-239515
Email: neerajdops@rediffmail.com
Abstract
Objective: Aim of present study to evaluate anticancer activity of dried fruit of Embelia ribes containing embelin in various types of cancer. Material and Method: Embelin was extracted and isolated from Embelia ribes berries of Indian origin. Isolated embelin was characterized by melting point, TLC, partition coefficient, solubility, FT-IR, NMR and Mass spectroscopy. Assessment of anticancer activity was done by Cytotoxicity assay and Apoptosis assay by using MCF‑7 breast cancer cells. Isolated Embelin was investigated on MCF-7 breast cancer cell line by MTT assay and apoptosis assay. Different concentration of embelin were used and observed that 80μg/ml was the IC50 value of embelin in MCF‑7 breast cancer cells and it was found that embelin was able to affect the viability of cells and also induce apoptosis of MCF‑7 breast cancer cells in a dose dependent manner. Results: We have found that the isolated embelin have significant (٭p < 0.0001) potency against the MCF-7 breast cancer cells in dose dependent manner and showed significant increase in apoptotic cells at IC50 value. Conclusion: Isolated embelin have significant potency against the MCF-7 breast cancer cells in dose dependent manner.
Keywords: Embelin, Apoptosis, Cancer, Cytotoxicity, Breast Cancer, TLC.
Introduction
The term “breast cancer” is a type of malignant tumor that originates from cells in the breast and is defined as the rapid and unlimited growth of abnormal cells in the mammalian glands which produces milk from breast or in the ducts that deliver milk to the nipples. The main site of breast cancer is stromal cell which include fibrous connective tissues and fatty. It is a leading disease affecting females in particular, and its occurrence is growing annually (Benson and Jatoi, 2012; Anderson et al., 2011). Widely, estimated number of cases of breast cancer reached 1.2 million. Worldwide, in every 3 minutes, a woman is identified with breast cancer and nearly about 1 million annually (Parkin, 2001). Hence, the crucial therapeutic methodology is required for the treatment of breast cancer.
However, market survey or literature review reveals that numerous pharmacological agents are used in the treatment of breast cancer include anastrozole, arimidex, carboplatin, capecitabine, docetaxel, cisplatin, cyclophosphamide, doxorubicin, epirubicin hydrochloride, flurouracil, fulvestrant, gemcitabine, herceptin, letrizole, methotrexate, paclitaxel, pertuzumab, tamoxifen, zoladex, vincristine and vinblastine but these drugs shows many side-effects like bleeding, decrease in blood cell counts, diarrhoea, allergic reaction, loss of appetite, hair loss, joint pain (short term), change in taste, weight gain, numbness or tingling in the fingers or toes, nausea and vomiting. Hence, alternative therapy having fewer side effects with maximal therapeutic efficacy is required to combat with this dreadful disease.
Embelin, (2, 5-dihydroxy-3-undecyl-p-benzoquinone), a naturally occurring quinonoid compound, is found to be the major constituent of Embelia ribes (Myrsinaceae). Embelia ribes commonly known as Vidanga or Via-vidang is a large woody climbing shrub and is widely distributed throughout Pakistan and India. The fruit contains embelic acid, tannins, christembine and embelin as major constituents. Vilangin, 2, 5-isobutylamine salts, quercetol and volatile oil are also present. The dried fruit has been used in India from time immemorial as an anthelmintic. Embelia ribes has been shown to possess astringent, carminative, stimulant, antioxidant, anti-spermetogenic, anti-bacterial, anticancer, anti-inflammatory, antidiabetic, antidyslipidemic, analgesic and cardio-protective activity. The fruit cures tumors, ascites, bronchitis, jaundice and mental disorders. The plant is used as an anti-inflammatory drug to relieve rheumatism and fever. Embelia ribes is extensively used in Indian traditional medicine for treating various disease conditions including chronic inflammatory disorders, heart and urinary conditions, snake and insect bites, and tumor (Kapoor et al., 1983). Embelin is available commercially as tablets and powders in spite of its not absorbed at all in entire upper portions of gastrointestinal tract and remain as such in colon. The log P value of embelin about 4.8 is indicating its high lipophilicity. Chemical structure of embelin is having quite resemblance with the structure of natural Coenzyme Q10 (ubiquinones) and the role of this is well defined in various biochemical protective mechanism (Mozaina et al., 2008).
Embelin is a polyphenolic compound that binds to the Smac binding site in the BIR3 domain of XIAP protein molecules and inhibit XIAP (X-linked apoptosis protein) (Reuter et al., 2010; Hu et al., 2011; Nikolovska-Coleska et al., 2004). Various studies have confirmed that embelin has anti‑oxidative biological and also has anti‑inflammatory effects (Joshi et al., 2007; Sreepriya and Bali, 2006; Mahendran et al., 2011; Thippeswamy et al., 2011). It has been established that embelin has a wide-ranging antitumor nature and is competent to control the development of different tumor cells, like breast, prostate, hepatic pancreatic and colon cancer (Danquah et al., 2009; Dai and Wang, 2010; Aird et al., 2008; Mori et al., 2007). However, the exhaustive mechanism of embelin for its antitumor activity remains unknown.
Embelin releases cytochrome C and activates the caspase family to result in the induction of breast cancer apoptosis through regulation of the action of the Bcl‑2/Bax family in the mitochondrial pathway (Li et al. 2013). It clearly inhibits NF-αB activation, which makes it a potentially effective suppressor of tumor cell survival, proliferation, invasion, angiogenesis, and inflammation (Ahn et al., 2007). PEG3.5K-EB2 conjugate and preliminary in-vitro study suggested that it functioned as a dual delivery system and showed synergistic activity with co-delivered PTX against several cancer cell lines (Huang et al., 2012). Similarly a conjugate of PEG5K with two embelin molecules (PEG5K-EB2) forms small-sized micelles (20-30 nm) that effectively solubilize hydrophobic drugs such as PTX. PTX-loaded PEG5K-EB2 micelles show kinetics of sustained release and are effectively targeted to tumors in vivo with minimal nonspecific uptake by RES and exhibited potent cytotoxicity to several cultured cancer cell lines (Lu et al., 2013). The aim of present study was to extract embelin from Embelia ribes and evalate its anti-cancer activity on MCF-7 breast cancer cell line using MTT assay and apoptosis assay.
Materials and methods
Plant material
Embelia ribes fruits were purchased from Kharibabli, Delhi and authenticated by Dr. H.B Singh, Chief Scientist & Head, Raw Material Herbarium & Museum, NISCAIR, New Delhi, India (NISCAIR/RHMD/Consult/-2012-2013/2090/97).
Chemicals
Standard embelin was purchased from Indofine chemical company, Inc,USA. Taxol was obtained as gift sample from Mac-Chem Products (India) Pvt. Ltd., Mumbai. TLC aluminium sheets silica gel 60 was supplied by E.Merck, Mumbai, India. MCF-7 cancer cellline was purchased from NCCS, Pune. MTT (3-(4, 5-dimethylthazol-2-yl)-2, 5-diphenyl tetrazolium bromide) and Annexin V-FITC Apoptosis Detection Kit were purchased from Sigma Aldrich, USA. All solvents used in the experiment procedure were analytical grade.
Isolation of Embelin
The pure embelin was isolated from the Embelia ribes by using extraction procedure (Suthar et al., 2012). Berries of Embelia ribes were air dried, coarsely powdered and stored in closed container. Dried powder (250 gm) was extracted with n-Hexane by using Soxhlet apparatus for 6 hours. Residue was obtained by removal of solvent by distillation. Then the residue was washed with cold pet ether. After washing, the residue was dissolved in mixture of dichloromethane (DCM) and methanol. Then the solution was kept for crystallization for 24 hours. After crystallization the crystals were filtered out from the mother solution. Finally washed the crystals with n-hexane and followed by DCM. The golden coloured embelin crystals were obtained and characterized the embelin using organoleptic properties like appearance, colour and consistency, melting point, chemical test, TLC and solubility.
Physical Parameters of isolated embelin
Isolated embelin from Embelia ribes were identified according to standard procedures.
Colour and consistency
Physical appearance of embelia like colour and consistency was analyzed by visual observation.
Melting point
A capillary melting point apparatus was used to determine melting point of the drug. A small amount of drug was filled in the previously one sided sealed capillary and melting point analyzed in melting point apparatus to observe melting point range.
TLC profiling
1 mg isolated embelin was dissolved in 10 ml DCM. 10µl of test sample was applied on pre-coated silica gel G aluminum plate. The chromatogram was run in the mobile phase Ethyl acetate: Toluene: Methanol: Formic Acid (0.5: 0.4: 0.1: 0.05) in twin trough chamber. After the saturation of chamber, TLC plate was developed and observed under ultra violet light, Rf value was compared with the standard marker (Chauhan et al., 1999).
Chemical Test
Identification of embelin was confirmed by chemical test. Embelin was dissolved in pet ether and then diluted ammonia solution was added to it. Formation of bluish-violet precipitate indicates the presence of embelin.
FT-IR spectroscopy
FT-IR spectra were obtained using a FT-IR spectrophotometer (Thermo Nicolet, USA). The potassium bromide (KBr) disk method was employed, mixing small amount of drug powder with spectroscopic KBr and compressed in a vacuum press to obtain a disk. Infrared Spectrum was recorded by scanning over a wave number region of 400-4000 cm-1 using Nicolet Omnic software. The characteristic I.R. spectrum and peaks were observed and were compared with the spectrum and peaks of the reference spectrum of the embelin (Radhakrishnan et al., 2011).
1H NMR Spectra
1H-NMR was done by sophisticated multinuclear FT NMR Spectrometer model Avance-II (Bruker) in the Centre. The instrument is equipped with a cryomagnet of field strength 9.4 T. Its 1H frequency is 400 Mhz, while for 13C the frequency is 100MHz.
Partition coefficient
Partition coefficient is a measure of drug lipophilicity and an indication of its ability to cross bio-membrane factor. It is also useful in screening of biologic properties. The partition coefficient is defined as ratio of unionized drug distributed between organic and aqueous phase. The partition coefficient of embelin was determined by Shake Flask Method (De Bruijn et al., 1989). The partition coefficient of embelin was determined in octanol: water. The octanol: water mixture was kept to equilibrate for 24 hours in separating funnel. Then 1 mg of the embelin in 25 ml of water and 25 ml of n-octanol was added to it and after 5 hours both the phase was separated and organic phase was analyzed using UV spectrophotometer. The concentration in aqueous phase was determined by difference between initial drugs added to mixture with drug concentration in organic phase.
PO/W = Concentration in organic phase/concentration in aqueous phase
Saturation solubility
The solubility of embelin in various solvents was determined by using shake flask method (Ibrahim et al., 2011). Briefly, an excess amount of embelin was added to each vial containing 1 ml of the selected vehicle. After sealing, the mixture was vortex for 10 min in order to facilitate proper mixing of embelin with the vehicles. Mixtures were then stirred for 24h on magnetic stirrer maintained at room temperature. After stirring mixtures were centrifuged at 5000 rpm for 5 min and drug in the supernatant was analyzed for embelin by UV-visible spectrophotometric method at λ max of 292.75 nm.
In-vitro Cytotoxicity Assay
Cell culture
MCF‑7 breast cancer cells were purchased from the Culture Collection (NCCS, Pune, India). Following cell passage, cells were inoculated in MEM culture medium containing 10% fetal calf serum. The cells were then cultured in an incubator containing 5% CO2 and 95% O2, at 37˚C (Chaudhari et al., 2012).
Cell viability
MCF-7 cells in the logarithmic growth phase were collected and cultured in 96‑well culture plates inoculated for 24 h. When the cells had grown to adherence, different concentration of embelin (40, 60, 80, 100, 120 μg/ml) were administered to various groups with triplicate wells for each concentration. A negative control group without embelin was also created. All cells were incubated in 5% CO2 for further culture for 24 prior to the colour reaction. Subsequently, MTT was added to each well, and cells were cultured in CO2 at 37˚C in an incubator for 2 h. The optical density (OD) values (A570nm) of each well were measured with a microplate reader (Bio‑Rad; Hercules, CA, USA) (Chaudhari et al., 2012).
Apoptosis Assay
Human breast cancer cell line (MCF-7) was treated with Annexin V-FITC Apoptosis detection kit. The cells were plated in 6 well plates and treated with different concentration of drugs and incubated. Media was removed and washed with PBS and re-suspended in annexin binding buffer. 1 μl annexin V dye and PI was added and kept in dark for 30 min. Finally cells were washed and observed under fluorescent microscope using appropriate filters.
The cells should separate into three groups: live, apoptotic and dead. Live cells show only weak annexin V staining of the cellular membrane, while apoptotic cells show asignificantly higher degree of surface labelling. Dead cells show both membrane staining by Annexin V and strong nuclear staining from the propidium iodide (Chaudhari et al., 2012).
Statistical analysis
All the results are expressed as mean ± standard deviation. The treated groups were compared with control by applying analysis of variance (ANOVA). The statistical analysis was carried out Graph Pad Software Corp., San Diego, CA. The p value <0.0001 was considered to indicate a statistically significant difference between groups.
Results
The pure embelin was isolated from the Embelia ribes by using extraction procedure and conformer its purity by physical appearance like colour, consistency, solvent system, Rf value and chemical test. The results of identification tests are in Table 1.
Table 1. Identification test, % Yield and Solvent System of Isolated Embelin
|
Test |
Results |
|
|
Colour |
Golden Yellow |
|
|
Consistency |
Crystalline |
|
|
% Yield |
8.4% |
|
|
Solvent System |
Ethyl acetate : Toluene : Methanol : Formic Acid 0.5 : 0.4 : 0.1 : 0.05 |
|
|
Rf Value |
0.752 |
|
|
Chemical Test |
Pet ether solution of embelin gives bluish-violet precipitate with dil. ammonia solution, which indicates the presence of embelin |
|
|
Melting Point |
Standard Embelin |
143-146°C |
|
Isolated Embelin |
142-145°C |
|
Table 2. Interpretation of FTIR of Embelin
|
Standard embelin peaks (cm-1) |
Isolated embelin peaks (cm-1) |
Interpretation |
|
1195 |
1194 |
C-O bond |
|
1615 |
1615 |
Stretching vibration of C =O |
|
2849 |
2848 |
Stretching vibration of methyl C-H |
|
3309 |
3308 |
Stretching vibration of OH group). |
|
2920 |
2920 |
Aromatic C-H stretching |
Table 3 Solubility profile of Isolated Embelin
|
Solvents |
Saturated solubility of Isolated Embelin (mg/ml) |
Description |
|
DMSO |
120-126 mg/ml |
Freely Soluble |
|
Methanol |
5-7 mg/ml |
Slightly Soluble |
|
Ethanol |
4-6 mg/ml |
Slightly Soluble |
|
Water |
0.2-0.3 mg/ml |
Insoluble |
FT-IR spectroscopy
Observed peaks in IR spectrum of standard embelin (Figure 1) and isolated embelin (Figure 2) were found to be concordant with functional groups present in structure of standard embelin and isolated embelin.
Table 2 shows frequency of observed bands and its interpretation. Purity of procured sample was confirmed from its IR spectrum.
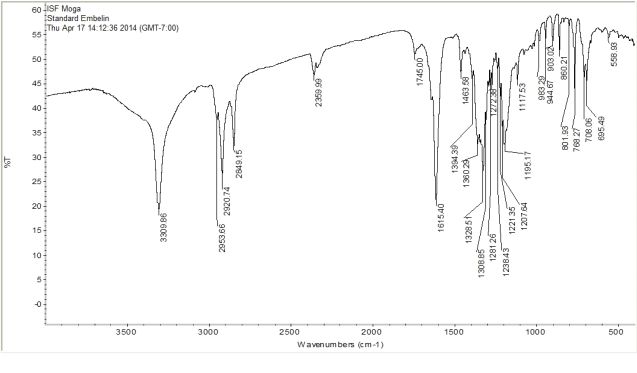
Figure 1. FT-IR spectrum of Standard Embelin.
NMR (Nuclear Magnetic Resonance)
(a) 1H NMR Spectra of Standard Embelin
Molecular Formula C17H26O4
Proton NMR (1H NMR- 300 MHz, DMSO) demonstrates chemical shifts at δ 7.8 (2H, board spectra, 2xOH), 5.8(1H, singlet, H-3), 1.5 (2H, triplet, H-7), 1.24 (18H, board spectra, H-8- H-16), 0.88 (3H, triplet, H-17). 1H NMR Spectra of standard embelin is shown in Figure 3.
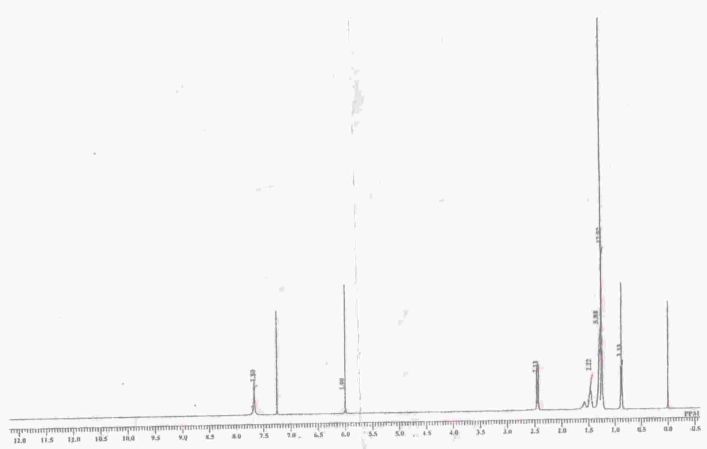 Figure 3. 1H NMR Spectra of Standard Embelin
Figure 3. 1H NMR Spectra of Standard Embelin
(b) 1H NMR Spectra of Isolated Embelin
Molecular Formula C17H26O4
Proton NMR (1H NMR- 400 MHz, DMSO) demonstrates chemical shifts at δ 10.97 (2H, board spectra, 2xOH), 5.75(1H, singlet, H-3), 2.30 (2H, triplet , H-7), 1.36 (2H,board spectra, H-8), 1.24 (16H, board spectra, H-16), 0.86 (3H, triplet, H-17. 1H NMR Spectra of Isolated Embelin is shown in Figure 4.
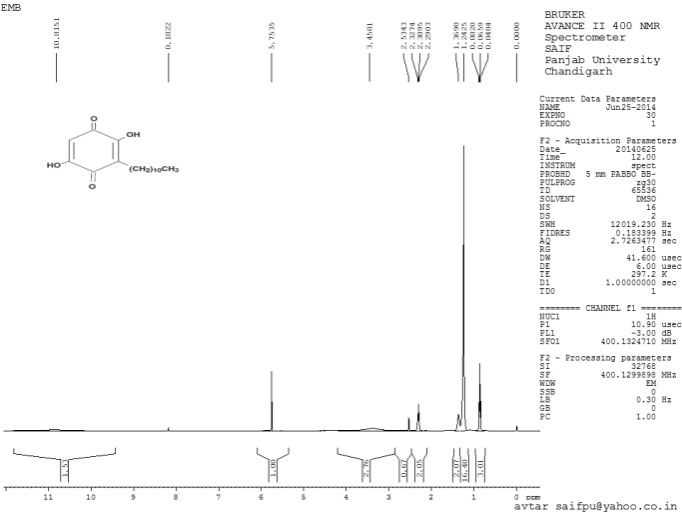
Figure 4. 1H NMR Spectra of Isolated Embelin
Partition coefficient
Partition coefficient of isolated embelin was found to be 4.595, which is comparable to the standard drug i.e. 4.609 by using formula given below;
Log Po/w = log (Solute Octanol/ Solute water)
Solubility of Embelin
Solubility studies were carried out by dissolving embelin in 1ml of different solvent and results were depicted Table 3.
In-vitro Cytotoxicity Assay
Embelin stimulates the inhibition of cell growth in MCFs-7 breast cancer cells. Various concentrations of isolated embelin i.e. 40, 60, 80, 100 and 120 μg/ml were added to MCF-7 breast cancer cells for 24 h. Then MTT method was utilized to verify the cell activity which is different at different concentrations (Figure 5). The results demonstrated that cytotoxicity potential of breast cancer cells significantly decreased, when the concentration of isolated embelin was increased and isolated embelin has shown significant cytotoxic potential in MCF-7 breast cancer cells and its IC50 value 80 μg/ml. This result indicates that isolated embelin has an inhibitory effect on the growth of MCF-7 breast cancer cells.
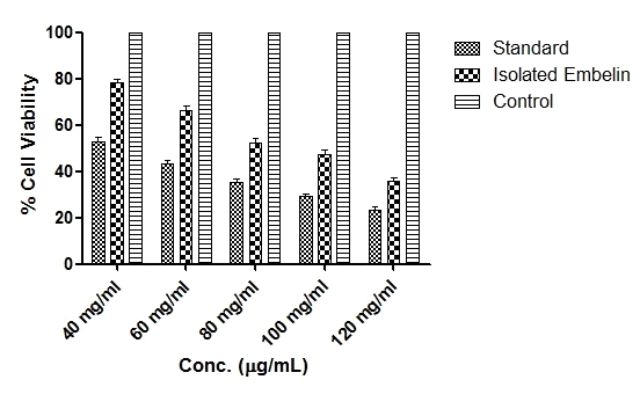 Figure 5. Cytotoxicity study of standard (Taxol) and Isolated Embelin in MCF-7 cell line by using MTT assay vs. control
Figure 5. Cytotoxicity study of standard (Taxol) and Isolated Embelin in MCF-7 cell line by using MTT assay vs. control
Apoptosis Assay
Embelin also induces apoptosis of MCF-7 breast cancer cells. The cancerous cells were treated with different concentration of isolated embelin for 24 h and observed under fluorescent microscope to determine the effect on the rate of apoptosis (Figure 6). The observed results established that the rate of MCF-7 breast cancer cell apoptosis significantly increased with increase in concentration of embelin in dose-dependent manner. At IC50 value of isolated embelin 80 μg/ml the rate of apoptotic cells was significant.
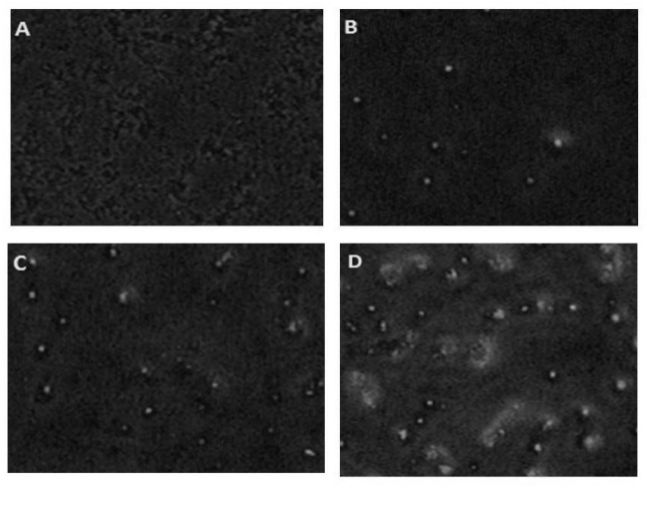 Figure 6. Apoptosis-inducing activity of isolated embelin in MCF-7 cell-line
Figure 6. Apoptosis-inducing activity of isolated embelin in MCF-7 cell-line
Discussion
Cancer is a very fast developing, serious and life‑threatening malignant tumor. It is important to study the disease in detail and find effective antitumor drugs, which are safe and efficient. Plants derived anticancer drugs have advantages of including a wide variety, low toxicity and few side-effects and adverse reactions. So, very effective anti-cancer drugs from plants are being explored. Embelin affects the explosion and apoptosis of a wide range of cancer cells because it acts as a small molecular inhibitor with specific inhibition of XIAP. Researchers have studied that embelin inhibits the cell multiplication of various abnormal cancerous cells, significant results have been seen in breast cancer and other solid cancerous cells (Allensworth et al., 2012).
In the present study, isolation of embelin was carried out by using n-hexane as a solvent reported by Suthar et al. with little modification. Ganesan and Madhavan demonstrated that isolation of embelin with other solvents like acetone, chloroform, carbon tetrachloride, diethyl ether, ethyl acetate, methanol and propanol (Ganesan et al., 2010; Madhavan et al., 2011). After isolation and purification, pure embelin was obtained in crystal form. The characterization studies of isolated embelin i.e. Colour, melting point, FTIR-Spectra, NMR-Spectra were compared with results of standard embelin, which were almost identical.
Based on our results cytotoxicity assay, we conclude that embelin is a potent inhibitor of XIAP, which make it potentially effective suppressor of tumor cell survival. We demonstrated that when breast cancer cells (MCF-7 cell line) had been treated with different concentration of embelin i.e. 40, 60, 80, 100, 120μg/ml for 24 hr, the rate of cell viability decrease with increase in concentration of embelin. This result indicates that the embelin showed inhibitory effect on the growth of breast cancer cells. The results were shown significant (٭p < 0.0001) activity of the isolated embelin was compared with the Standard (Taxol). These results were found to be similar as the studies done by Yang et al., 2013 by using embelin (Li et al., 2013).
Embelin is capable to induce breast cancer cell apoptosis instead of directly causing cell death. In figure 5, control treatment group (0μg/ml) shows absence of apoptotic cells due to absence of embelin. The percentage of apoptotic cells was higher at 40μg/ml as compared to control group but less than the percentage of apoptotic cells at 60μg/ml. At IC50 value (80μg/ml), high percentage of apoptotic cells was present. Our IC50 were found to be similar with the studies done by Yang et al., 2013. However, embelin induce higher apoptosis at higher concentration in dose dependent manner.
Conclusion
In this study, embelin was isolated from Embelia ribes and confirmed its chemical structure by FTIR and 1H NMR spectroscopy. We have found that the isolated embelin have significant (٭p < 0.0001) potency against the MCF-7 breast cancer cells in dose dependent manner and showed significant increase in apoptotic cells at IC50 value. So from the above results we can conclude that isolated embelin can effectively used in the treatment of breast cancer in dose dependent manner.
Acknowledgement
We are grateful to Shri Parveen Garg (Chairman), I.S.F. College of Pharmacy, Ferozepur Road, Moga 142001, INDIA for their necessary support and motivation.
Declaration of Interest
The authors have declared no conflict of interest.
References
Ahn KS, Sethi G, Aggarwal BB. 2007. Embelin, an inhibitor of X chromosome-linked inhibitor-of-apoptosis protein, blocks nuclear factor-kappa B (NF-kappa B) signaling pathway leading to suppression of NF-kappa B-regulated antiapoptotic and metastatic gene products. Molecular Pharmacology 71: 209-219.
Aird KM, Ding X, Baras A, Wei J, Morse MA, Clay T, Lyerly HK and Devi GR. 2008. Trastuzumab signaling in ErbB2-overexpressing inflammatory breast cancer correlates with X-linked inhibitor of apoptosis protein expression. Molecular Cancer Therapeutics 7:38-47.
Allensworth JI, Aird KM, Aldrich AJ, Batinic-Haberle I & Devi GR. 2012. XIAP inhibition and generation of reactive oxygen species enhances TRAIL sensitivity in inflammatory breast cancer cells. Molecular Cancer Therapeutics 11 :1518-1527.
Anderson WF, Katki HA and Rosenberg PS. 2011. Incidence of breast cancer in the United States: current and future trends. Journal of the National Cancer Institute 103:1397-1402.
Benson JR and Jatoi I. The global breast cancer burden. 2012. Future Oncology 8: 697-702.
Chaudhari KR, Ukawala M, Manjappa AS, Kumar A, Mundada PK 2012. Opsonization, biodistribution, cellular uptake and apoptosis study of PEGylated PBCA nanoparticle as potential drug delivery carrier. Pharmaceutical Research 29: 53-68.
Chauhan S, Singh B & Agarwal S. 1999. A TLC Identification and Spectrophotometric Estimation of Embelin in Embelia ribes. Ancient Science of Life 19:46-48.
Dai Y, Wang WH. 2010. Peroxisome proliferator-activated receptor γ and colorectal cancer, World Journal of Gastrointestinal Oncology 2:159-164.
Danquah M, Li F, Duke Iii CB, Miller DD, Mahato RI. 2009. Micellar delivery of bicalutamide and embelin for treating prostate cancer. Pharmaceutical Research 26:2081-2092.
De Bruijn J, Busser F, Seinen W, Hermens J. 1989. Determination of octanol/water partition coefficients for hydrophobic organic chemicals with the “slow‐stirring” method. Environmental Toxicology and Chemistry 8:499-512.
Ganesan B, Perumal P, Manickam V, Gotteti SD, Srikakolapu SR 2010. Optimization of extraction conditions for embelin in Embelia ribes by UV spectrophotometry. Archives of Applied Science Research 2:49-53.
Hu R, Zhu K, Li Y, Yao K, Zhang R. 2011. Embelin induces apoptosis through down-regulation of XIAP in human leukemia cells. Medical Oncology 28:1584-1588.
Huang Y, Lu J, Gao X, Li J, Zhao W. 2012. PEG-derivatized embelin as a dual functional carrier for the delivery of paclitaxel. Bioconjugate chemistry 23:1443-1451.
Ibrahim MA, Mahrous GM, El-Badry M, Al-Anazi FK. 2011. Indomethacin-loaded pellets prepared by extrusion/spheronization: Effect of Cosolvents. Farmacia 59: 483-499.
Joshi R, Kamat J, Mukherjee T. 2007. Free radical scavenging reactions and antioxidant activity of embelin: biochemical and pulse radiolytic studies. Chemico-Biological Interactions 167:125-134.
Kapoor V, Chawla A, Kumar M, Kumar P. 1983. Anti-inflammatory agent in Indian Laboratories. Indian Drugs 30 :481-488.
Li Y, Li D, Yuan S, Wang Z, Tang F. 2013. Embelin-induced MCF-7 breast cancer cell apoptosis and blockade of MCF-7 cells in the G2/M phasevia the mitochondrial pathway. Oncology Letters 5 :1005-1009.
Lu J, Huang Y, Zhao W, Marquez RT, Meng X. 2013. PEG-derivatized embelin as a nanomicellar carrier for delivery of paclitaxel to breast and prostate cancers. Biomaterials 34:1591-1600.
Madhavan SN, Arimboor R, Arumughan C. 2011. RP‐HPLC‐DAD method for the estimation of embelin as marker in Embelia ribes and its polyherbal formulations. Biomedical Chromatography 25: 600-605.
Mahendran S, Badami S, Ravi S, Thippeswamy BS & Veerapur VP. 2011. Synthesis and evaluation of analgesic and anti-inflammatory activities of most active free radical scavenging derivatives of embelin-A structure-activity relationship. Chemical and Pharmaceutical Bulletin 59 : 913-919.
Mori T, Doi R, Kida A, Nagai K, Kami K. 2007. Effect of the XIAP inhibitor Embelin on TRAIL-induced apoptosis of pancreatic cancer cells. Journal of Surgical Research 142 : 281-286.
Mozaina K, Cantrell CL, Mims AB, Lax AR, Tellez MR. 2008. Activity of 1, 4-benzoquinones against formosan subterranean termites (Coptotermes formosanus). Journal of Agricultural and Food Chemistry 56 : 4021-4026.
Nikolovska-Coleska Z, Xu L, Hu Z, Tomita Y, Li P. 2004. Discovery of embelin as a cell-permeable, small-molecular weight inhibitor of XIAP through structure-based computational screening of a traditional herbal medicine three-dimensional structure database. Journal of Medicinal Chemistry 47 : 2430-2440.
Parkin DM. 2001. Global cancer statistics in the year 2000, The Lancet Oncology 2 :533-543.
Radhakrishnan N, Gnanamani A & Mandal A. 2011. A potential antibacterial agent Embelin, a natural benzoquinone extracted from Embelia ribes. Biology and Medicine 3 :1-7.
Reuter S, Prasad S, Phromnoi K, Kannappan R, Yadav VR. 2010. Embelin suppresses osteoclastogenesis induced by receptor activator of NF-κB ligand and tumor cells in vitro through inhibition of the NF-κB cell signaling pathway. Molecular Cancer Research 8: 1425-1436.
Sreepriya M, Bali G. 2006. Effects of administration of embelin and curcumin on lipid peroxidation, hepatic glutathione antioxidant defense and hematopoietic system during N-nitrosodiethylamine/phenobarbital-induced hepatocarcinogenesis in Wistar rats. Molecular and Cellular Biochemistry 284: 49-55.
Suthar M, Patel R, Hapani K & Patel A. 2009. Screening of Embelia ribes for antifungal activity. Int J Pharma Sci Drug Res 1:203-206.
Thippeswamy BS, Mahendran S, Biradar MI, Raj P, Srivastava K. 2011. Protective effect of embelin against acetic acid induced ulcerative colitis in rats. European journal of Pharmacology. 654: 100-105.