

Anis Ahmed Shaikh, Mohamad Asif, Prashant Netankar, Sayyad Sultan Kasim*
Post Graduate and Research Centre , Maulana Azad College, Aurangabad 431001 Maharashtra, India
*Address for Corresponding Author
Sayyad Sultan Kasim
Post Graduate and Research Centre, Maulana Azad College, Aurangabad 431001 Maharashtra, India
Abstract
Objective: The main objective of this research work was to synthesis the triazoles compounds by Click reaction and evaluated their antibacterial activities against gram positive and gram negative bacteria. Material and Methods: In this work 1,4-disubstituted 1,2,3-triazole compounds (3a-3i) were synthesized by Azide Alkyne Cycloaddition reaction and characterized by IR,1H NMR and Mass spectroscopy. Further these compounds were evaluated for antibacterial activity against Bacillus substilus, Staphylococcus aureus and Escherichia coli by using Ciprofloxacin as standard compound with various concentrations. Results: Among all the compounds (3a-3i) which were screened for antibacterial activity, the compounds 3c, 3d, 3e and 3i were found to be excellent antibacterial agents. Conclusion: The presence of electron donating, electron withdrawing and halogen groups plays an important role of biological activity of compounds.
Keywords: Alkyne, antibacterial activity, azide, click reaction, triazole
Introduction
Triazoles are important five membered nitrogen heterocycles having three nitrogen atoms and two carbon atoms in ring. 1,2,4 triazole moiety containing drugs are now a days available in market due to their excellent biological activity, it includes Fluconazole, hexaconazole, rizatriptan as shown in figure.
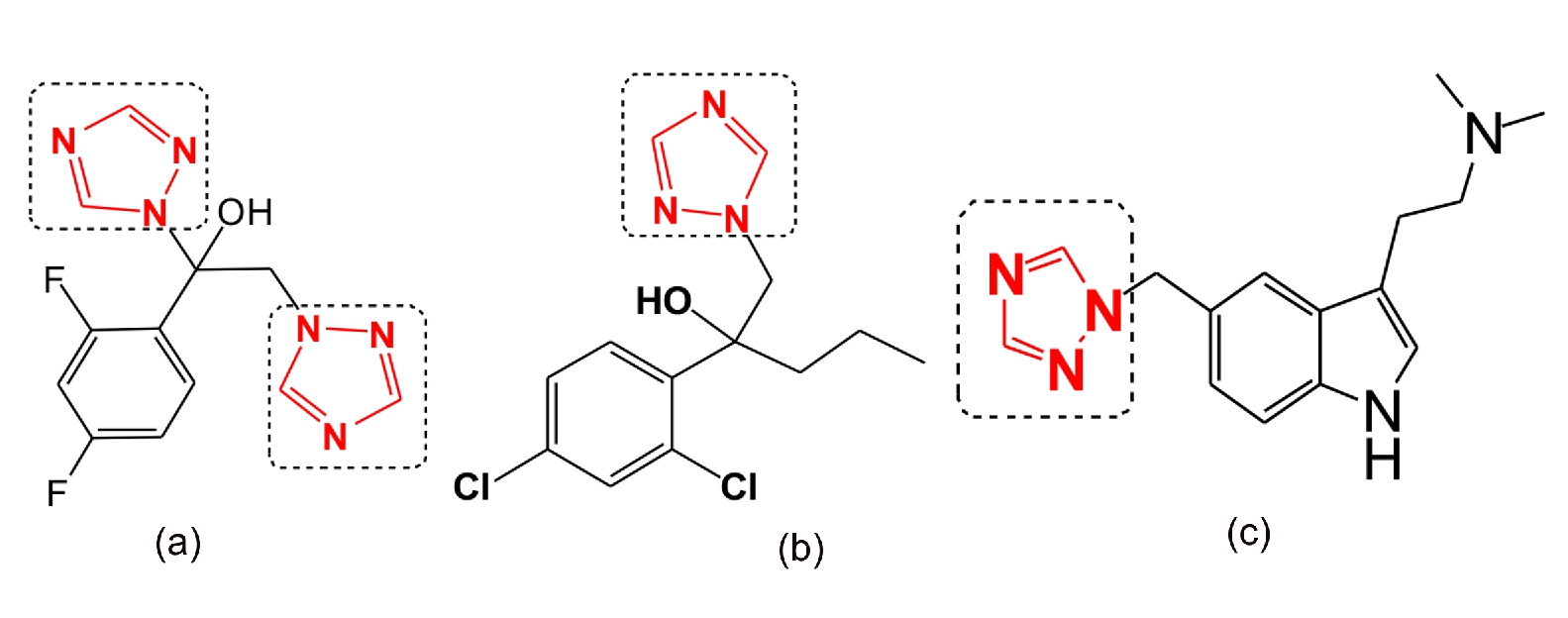
Figure 1. Bioactive 1,2,4 Triazole Compounds: (a) Fluconazole as Antimycotics, (b) Hexaconazole as Antifungal, (c) Rizatriptan as Antimigrane
Similarly 1,2,3 triazoles moiety containing drugs are excellent antimicrobial (Phillips et al., 2009), antitubercular (Patpi et al., 2012), antiallergic (Buckle et al., 1984), antrifungal (Ferreira et al., 2015), antiHIV (Mohammed et al., 2016), anti-inflammatory (Shafi et al., 2012), anticancer (Grana et al., 2006; Zhang et al., 2008 ) antiplatelet (Palhegan et al., 2001), anti-Alzheimer (Monceaux et al., 2011) and antiviral (Joan et al., 1998) agents. The biologically important drugs containing 1,2,3 triazole moieties are shown in figure 2.

Figure 2. Examples of drugs containing 1,2,3 triazole moiety
In spite of biologically significant, the 1,2,3 triazoles are also found to have wide range of agricultural (Gisi et al., 2002) and industrial (Kim et al., 2010) applications. Due to simple structure and versatile applications of triazoles, several methods have been introduced for synthesis of 1,4 and 1,5 substituted triazoles compounds. The Husgen 1,3 dipolar cycloaddition reaction (Rostovtsev et al., 2002) is well known which produced 1,4 substituted triazole in presence of copper. The literature survey revealed (Holla et al., 2005) that 1,4 disubstituted 1,2,3 triazoles compounds with electron donating, withdrawing and halogens substituents plays an important role in bioactivity. Therefore the biological importance of 1,4 disubstituted 1,2,3 triazole with various substituents on ring inspired us to synthesis the substituted triazole and evaluate their biological activity. By taking the factors like presence of haloges, electron donating and withdrawing groups, position of substituent we have performed the reaction between alkyne (1a-i) with azides (2a-i) catalysed by copper to give the 1,4 disubstituted 1,2,3 triazoles (3a-i) as shown in scheme 1. The resulting triazoles (3a-i) were then screened for antibacterial activity against Bacillus substilius, Staphylococcus aureus and Escherichia coli. The results obtained are presented in table 1.
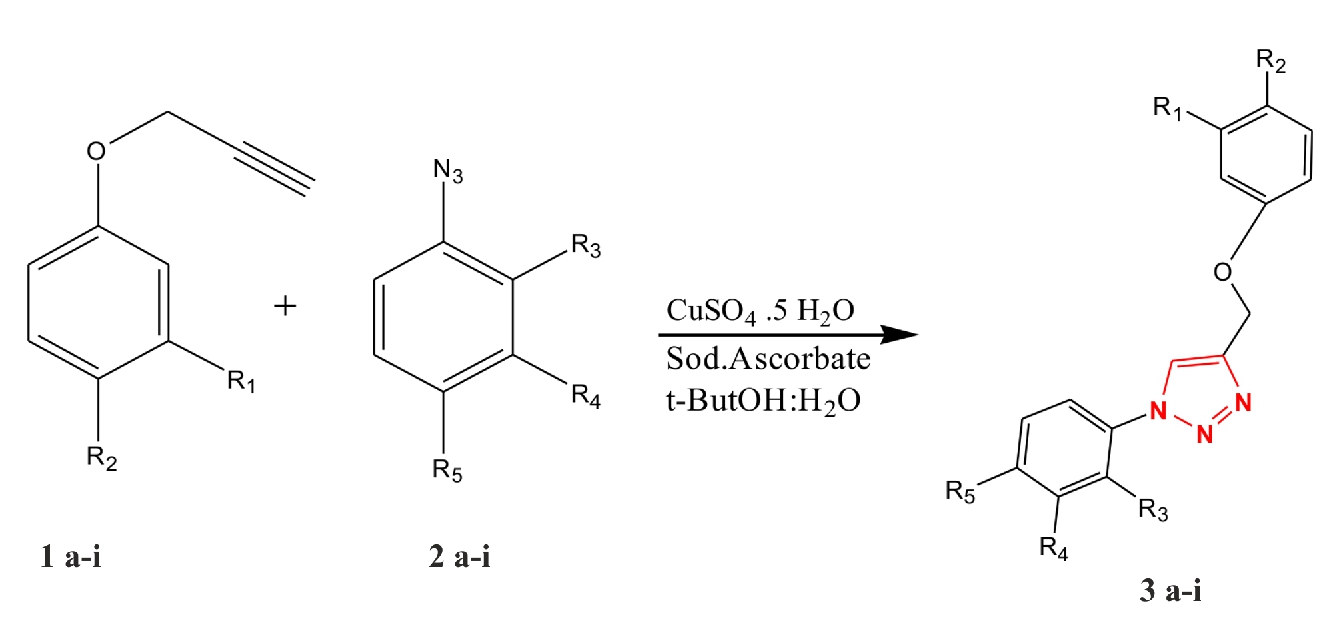
Scheme 1. Synthesis of 1,4 disubstituted 1,2,3 Triazoles
Where
1a : R1=H, R2=NO2 2a : R3=H,R4=H,R5=COCH3 3a : R1=H, R2=NO2,
R3=H,R4=H,R5=COCH3
1b : R1=H, R2=OCH3 2b : R3=H,R4=H,R5=COCH3 3b : R1=H, R2= OCH3
R3=H,R4=H,R5=COCH3
1c : R1= NO2, R2=H 2c : R3=H,R4=H,R5=COCH3 3c : R1=NO2, R2= H
R3=H,R4=H,R5=COCH3
1d : R1=H, R2=NO2 2d : R3=H,R4=H,R5= NO2, 3d : R1=H, R2=NO2,
R3=H,R4=H,R5= NO2,
1e : R1= NO2 , R2=H 2e : R3=H,R4=H,R5= NO2, 3e : R1= NO2 , R2=H
R3=H,R4=H,R5= NO2,
1f : R1=H, R2=NO2 2f : R3= COCH3,R4=H,R5= H, 3f : R1=H, R2=NO2,
R3= COCH3,R4=H,R5= H
1g : R1= NO2 , R2=H 2g : R3= COCH3,R4=H,R5= H, 3g : R1= NO2 , R2=H
R3= COCH3,R4=H,R5= H
1h : R1=H, R2=NO2 2h : R3= NO2,R4= NO2,R5= H 3h : R1=H, R2=NO2
R3= NO2,R4= NO2,R5= H
1i : R1= NO2 , R2=H 2i : R3= NO2,R4= NO2,R5= H 3i : R1= NO2 , R2=H
R3= NO2,R4= NO2,R5= H
Material and methods
The SD-Fine brand chemicals were used for this work. Melting points were determined in an open capillary by micro controller based melting point apparatus of Chemline company and are uncorrected. 1H NMR spectra were recorded on Bruker 400 MHz spectrometer by TMS as standard. Mass Spectra were recorded using Thermo Fischer mass spectrometer. IR Spectra were determined.
Synthesis of 1, 4 disubstituted 1,2,3-triazoles (3a-3i)
In a round bottom flask alkyne 1a-i (3 mmol) and azide 2a-i (3.5 mmol) were added in 1:1 mixture of ter-butyl alcohol and water (40 ml).Sodium Ascorbate (60 mg, 0.3 mmol, 1 ml of freshly prepared 3M solution in water) was added, followed by copper sulphate pentahydrate (7.5 mg, 0.03 mmol, 1 ml of 0.3 M solution in water). The resulting mixture was stirred at room temperature for 24 hrs the completion of reaction was confirmed by TLC. Then the reaction mixture was diluted with water, extracted with CH2Cl2, dried over Na2SO4, filtered and concentrated in vacuum to give a crude product, which was purified by column chromatography on silica gel to obtain pure product. The structures of desired products were confirmed by 1HNMR, Mass and IR Spectroscopy.
Antibacterial evaluation
The stock solution of all the synthesised compound (3a-i) were prepared by dissolving them in DMSO. The antibacterial activity of compounds 3a-i and Ciprofloxacin was performed by close disc method (Bhalodia et.al, 2011). All the cultures were placed for maintenance on nutrient agar and incubated at 40 0 C overnight. After centrifugation and sterilisation 0.1 ml of bacterial culture solution was spread on nutrient agar plate. During the process Ciprofloxacin antibiotic disc was used as controller. The disc was then kept on nutrient agar plate and then incubated for 24 hours at 400C and zone of inhibition were measured in millimetre.
Results and discussion
1,4 disubstituted 1,2,3-triazoles (3a-i) were successfully synthesised by click reaction and their characterisation results showed the acceptable results. The synthesized compounds were evaluated for their antibacterial activity and found that all the compounds showed good to moderate antibacterial activity. The compounds 3c, 3d, 3e and 3i were found to be excellent antibacterial agents (Table 1).
Table 1. Antibacterial activity of 1, 4 disubstitiuted 1,2,3 triazoles (3a-3i) (Zone of inhibition in millimetre at 300 μg/ml concentration)
|
Compounds |
Concentration (μg/ml) |
Bacillus subtillus |
Staphylococcus aureus |
Escherichia coli |
|
3a |
300 |
17 |
19 |
18 |
|
3b |
300 |
16 |
15 |
- |
|
3c |
300 |
11 |
12 |
14 |
|
3d |
300 |
14 |
15 |
14 |
|
3e |
300 |
10 |
11 |
13 |
|
3f |
300 |
19 |
16 |
19 |
|
3g |
300 |
14 |
16 |
- |
|
3h |
300 |
- |
17 |
15 |
|
3i |
300 |
11 |
10 |
11 |
|
Ciprofloxacin |
30 (μg/disc) |
23 |
21 |
22 |
Compound 3a (1-{4-[1-(4-Nitro-phenyl)-1H- 1,2,3-triazol-4-ylmethoxy]-phenyl}-ethanone):
Yield 73 %, m.f. C17H14N4O4 , M.Pt.178-183 0 C. 1H NMR (CDCl3, 400 MHz): δ 2.5 (s, 3H), 5.3 (s, 2H), 7.9 (s, 1H), 7.1 (d, 2H), 7.9 (d, 2H), 8.4 (d, 2H) 9.0 (d, 2H), LC–MS: m/z: 339.31 (M+ 1)+ , IR KBr cm-1 2915(CH3),1713(C=O),1597(NO2)1257(OCH2)
Compound 3b (1-{4-[1-(4-Methoxyphenyl)-1H- 1,2,3-triazol-4-ylmethoxy]-phenyl}-ethanone):
Yield 91 %, m.f. C18H17N3O3 , M.Pt. 121 -127 0 C. 1H NMR (CDCl3, 400 MHz): δ 2.5 (s, 3H), 3.8 (s, 2H),5.3 (s,2H), 7.0 (d, 2H), 7.1 (d, 2H), 7.6(s, 1H), 7.7 (d, 2H) 7.9 (d, 2H), LC–MS: m/z: 324.34 (M+ 1)+ , ,IR KBr cm-1 2920(CH3),1719(C=O),1250(OCH2)
Compound 3c (1-{4-[1-(Nitro-phenyl)-1H- 1,2,3-triazol-4-ylmethoxy]-phenyl}-ethanone):
Yield 89 %, m.f. C17H14N4O4, M.Pt.155-161 0 C 1H NMR (CDCl3, 400 MHz): δ 2.5 (s, 3H), 5.4 (s, 2H), 7.1 (d, 2H), 7.6 (d, 2H),7.7 (s,1H),8.1(d 1H), 8.2(d,2H),8.3 (m, 1H), 8.6 (s, 1H), LC–MS: m/z: 339.31 (M+ 1)+ , IR KBr cm-1 2931(CH3),1775(C=O),1696(CH=CH)
Compound 3d (1-(4-Nitro-phenoxymethyl)-1-(4-nitro-phenyl)-1H-1,2,3 triazole):
Yield 69 %, m.f. C15H11N5O5, M.Pt.210-216 0 C 1H NMR (CDCl3, 400 MHz): δ 5.4 (s, 2H), 7.1 (d, 2H),7.5 (d,2H), 7.8 (s, 1H), 7.9 (d, 2H), 8.4(d, 2H), LC–MS: m/z: 342.18 (M+ 1)+ , IR KBr cm-1 1720,(CH=CH),1590(NO2),1255(OCH2)
Compound 3e (1-(3-Nitro-phenoxymethyl)-1-(4-nitro-phenyl)-1H-1,2,3 triazole):
Yield 81 %, m.f. C15H11N5O5, M.Pt.163-167 0 C 1H NMR (CDCl3, 400 MHz): δ 5.4 (s, 2H), 7.1-7.6 (m, 4H), 7.8 (d, 2H),7.7(s,1H), 8.3(d, 2H), LC–MS: m/z: 342.18 (M+ 1)+ , IR KBr cm-1 1713(CH=CH),1574(NO2)1260(OCH2)
Compound 3f (1-{2-[1-(4-Nitro-phenyl)-1H- 1,2,3-triazol-4-ylmethoxy]-phenyl}-ethanone):
Yield 73 %, m.f. C17H14N4O4, M.Pt.127-131 0 C 1H NMR (CDCl3, 400 MHz): δ 2.6 (s, 3H), 5.4 (s,2H),7.0-7.5 (m,4H), 7.4(d,2H),7.7(s,1H), 8.4(d, 2H), LC–MS: m/z: 339.31 (M+ 1)+ , IR KBr cm-1 2913(CH3),1725(C=O),1305(N-O)
Compound 3g (1-{2-[1-(3-Nitro-phenyl)-1H- 1,2,3-triazol-4-ylmethoxy]-phenyl}-ethanone):
Yield 82 %, m.f. C17H14N4O4, M.Pt.137-143 0 C 1H NMR (CDCl3, 400 MHz): δ 2.6 (s, 3H), 5.4 (s,2H),7.0-7.7 (m,4H), 8.1-8.6(m,3H),7.7(s,1H), 8.6(s, 1H), LC–MS: m/z: 339.31 (M+ 1)+ , IR KBr cm-1 2940(CH3),1712(C=O),1561(NO2)
Compound 3h (4-(2,3-Dinitro-phenoxymethyl)-1-(4-nitro-phenyl)-1H-1,2,3-trizole):
Yield 63 %, m.f. C15H10N6O7, M.Pt.171-175 0 C 1H NMR (CDCl3, 400 MHz): δ 5.6 (s,2H),7.8 (d,2H), 8.1-8.4(m,3H),8.9(d,2H), LC–MS: m/z: 387.16 (M+ 1)+, IR KBr cm-11696(CH=CH),1539(NO2 ),1261(OCH2)
Compound 3i (4-(2,3-Dinitro-phenoxymethyl)-1-(3-nitro-phenyl)-1H-1,2,3-triazole):
Yield 63 %, m.f. C15H10N6O7 ,M.Pt.153-157 0 C 1H NMR (CDCl3, 400 MHz): δ 5.6 (s,2H),7.2 (s,1H), 7.5-8.4(m,3H),8.7(s,1H),7.7-8.7(m,3H) LC–MS: m/z: 387.16 (M+ 1)+ , IR KBr cm-1 1708(CH=CH),1570(NO2 ),1211(OCH2)
Conclusion
In conclusion we have synthesised the 1,4 disubstituted 1,2,3 triazoles by well known Click reaction and evaluate their antibacterial activities against Bacillus subtilus, Staphylococcus aureus and Escherichia coli and found that these are good antibacterial agents. Out of which, the compounds 3c, 3d, 3e and 3i showed excellent antibacterial activity, it is due to presence of nitro and carbonyl groups at different positions of ring which enhance the activity of these compounds as compared to other compounds.
Conflicts of interest
The author declares that there is no conflict of interest.
References
Bhalodia NR, Shukla VJ. 2011. Antibacterial and antifungal activities from leaf extracts of Cassia fistula l: An ethnomedicinal plant. Journal of Advance Pharmaceutical and Technological Research, 2:104–109.
Buckle DR, Rockell CJ, Smith MH, Spicer BA. 1984. Studies on 1,2,3,-triazoles. Synthesis and antiallergic properties of 9-oxo-1H, 9H-benzothiopyrano [2,3-d]-1,2,3-triazoles and their S-oxides. Journal of Medicinal Chemistry, 27:223.
Ferreira SZ, Carneiro HC, Lara HA. 2015. Synthesis of a New Peptide–Coumarin Conjugate: A Potential Agent against Cryptococcosis. American Chemical Society, Medicinal Chemistry Letters, 6:271.
Gisi U, Sierotzki H, Cook A, McCaffery A. 2002. Novel Optical/Electrochemical Selective 1,2,3-Triazole Ring-Appended Chemosensor for the Al3+ Ion. Pest Management Science, 58:859.
Grana GJ. 2006. Adjuvant aromatase inhibitor therapy for early breast cancer: A review of the most recent data. Surgical Oncology, 93:585.
Holla BS, Mahalinga M, Karthikeyan MS, Poojary B, Akberali PM, Kumari NS. 2005. Synthesis characterization and antimicrobial activity of some substituted 1,2,3-triazoles. European Journal of Medicinal Chemistry, 40:1173.
Joan CF, Elizabeth HT, Beatrice M, Daniel PB. 1998. Mechanisms influencing the evolution of resistance to Qo inhibitor fungicides. Antimicrobial Agents and Chemotherapy Journal 42:313.
Kim H, Lee S, Lee J, Tae J. 2010. Rhodamine triazole based fluorescent probe for the detection of Pt2+. Organic Letters, 12:5342.
Mohammed I, Kummetha IR, Singh G. 2016. 1,2,3-Triazoles as Amide Bioisosteres: Discovery of a New Class of Potent HIV-I. Antagonists. Journal of Medicinal Chemistry, 59:7677.
Monceaux CJ, Hirata C, Fukae PC, Lam. 2011. Triazole-linked reduced amide isosteres: An approach for the fragment-based drug discovery of anti-Alzheimer’s BACE1 inhibitors Bio-organic and Medicinal Chemistry Letters, 21:3992
Palhagen S, Canger R, Henriksen O. 2001. Rufinamide: a double-blind, placebo-controlled proof of principle trial in patients with epilepsy. Epilepsy Research, 43:115.
Patpi SR, Pulipati L, Yogeeswari P, Sriram D, Jain N, Sridhar B, Murthy R, Anjana DT, Kalivendi SV, Kanteveri S. 2012. Design, Synthesis and Structure Activity Correlations of Novel Dibenzo [b,d] furan, Dibenzo [b,d] thiophene, and N-Methylcarbazole Clubbed 1,2,3-Triazoles as Potent Inhibitors of Mycobacterium tuberculosis. Journal of Medicinal Chemistry, 55:3911.
Phillips OA, Udo EE, Abdel-Hamid ME. 2009. Synthesis and antibacterial activity of novel 5-(4-methyl-1H-1,2,3-triazole) methyl oxazolidinones. European Journal of Medicinal Chemistry, 44:3217.
Rostovtsev VV, Green LG, Fokin V, Sharpless KB. 2002. A Stepwise Huisgen Cycloaddition Process: Copper (I)‐Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes. Angewandte Chemie International Edition, 41:2596.
Shafi S, Alam MM, Mulakayala N. 2012. Synthesis of novel 2-mercapto benzothiazole and 1,2,3-triazole based bis-heterocycles: Their anti-inflammatory and anti-nociceptive activitie. European Jornal of Medicinal Chemistry, 49:324.
Zhang J, Garrossian M. 2008. Synthesis and anticancer activity studies of cyclopamine derivatives. Bioorganic and Medicinal Chemistry Letters, 18:1359.