

Anitha Huliyurdurga Channigarayappa1, Sreenivasa Swamy1*, Mohan R Nadigar2, Vivek Chandramohan3, Shivaraja Govindaiaha1
1Department of Studies and Research in Chemistry, Tumkur University, Tumkur-572103, Karnataka, INDIA
2Department of PG Studies in Chemistry, Shridevi PG Centre. Sira Road, Tumkur-572106, Karnataka, INDIA.
3Department of Biotechnology, Siddaganga Institute of Technology, Tumkur-572103, Karnataka, INDIA
*Corresponding Author
Dr. S. Sreenivasa
Associate Professor and Coordinator
Department of Studies and Research in Chemistry,
Tumkur University, Tumkur -572103, Karnataka (state), India
Cell: +91 96202 30672 (P), +91 94482 33725 (R)
Abstract
Background: Piperazine derivatives are the most privileged structural motifs in the field of nitrogen heterocyclic chemistry. They occur in several natural and synthetic bioactive compounds and are widely used in medicine. Objective: Piperazine with sulfonyl moiety place an important role in showing diverse biological activities. The objective of present study was consisting of synthesis, characterization and biological evaluation of some sulfonyl piperazine derivatives for antimicrobial, anthelmintic and inflammatory activity. Methods: In present studies we describe synthesis of a new substituted sulfonyl piperazine derivatives (8a-j), by coupling 1-[(4-methylphenyl)sulfonyl]piperazine a key intermediate with different substituted benzyl bromide. Synthesized compounds were characterized by using 1H NMR, 13C NMR, IR spectroscopic and CHN elemental analysis and were screened for in vitro antimicrobial action against different bacterial strains and fungal cultures using micro dilution assay and disc diffusion method, anthelmintic activity using test model Pheretima posthuma. Some of the promising molecules were tested for their anti-inflammatory activity by carrageenan induced paw edema against albino wistar rats and hypothetical binding mode are also studied by molecular modeling study. Results: Compounds 8a, 8b, 8c, 8e and 8f showed good antimicrobial activity, compounds 8b, 8c and 8e displayed anthelmintic activity comparable to standard and compounds 8a, 8b and 8f showed demonstrated significant anti-inflammatory activity. Conclusion: Synthesized piperazine sulfonamides in good yield, biologically activity and future pharmacological studies can be done on these structural specific synthesised molecules and QSAR studies.
Keywords: N-Boc Piperazine, Sulfonamides, Antimicrobial, Anthelmintic, Anti-inflammatory
Introduction
Microorganisms are the foremost contributing agents for various types of infectious diseases (Jacquelyn et al., 1993). Major health problem faced by many scientifically developing countries are the infectious diseases, which are systematically treated with various drugs of synthetic and natural origin. Approval of new target drugs is very important in the context of the upswing in resistance, cellular level enzymatic activity is one of the impelling factors in current drugs resistance (Paul et al., 2015). Enzyme golden carotenoid dehydrosqualene synthase (CrtM) pigment of Staphylococcus aureus having a structural resemblance to human squalene synthase enzyme involved in the cholesterol synthesis pathway in humans is also known for its antioxidant property in bacterium (Kahlon et al., 2010). N-myristoyltransferase (NMT) a monomeric enzyme abundantly found in eukaryotic organisms catalyzes the irreversible covalent addition of fatty acid myristate to the glycine N-terminal of the eukaryotic protein (Fang et al., 2015, Sogabe et al., 2002), are responsible for microbial infections. Helminthic infections are also documented as chronic ill health and listlessness caused among children and affect domestic animals and livestock causing substantial economic loss (Raghavamma and Rama Rao, 2010).
A bifunctional enzyme Prostaglandin-endoperoxide synthase (Cyclooxygenase) catalyzes the reaction of arachidonic acid to prostaglandin (PG)H2, the close forerunner to prostaglandins are thromboxane and prostacyclin. The COX-1 enzyme is vital in many tissues for production of sufficient amounts of prostaglandins required for the devaluation of normal functions of organs (Harman et al., 2007) and during inflammation COX-2 is responsible for the elevated production of prostaglandins (Kurumbail, et al., 2007). In spite of the accessibility to the large volume of antibiotics for treatment of infusions diseases and a considerable increase in drug resistance. There is a considerable curiosity in finding and developing new classes of drugs.
A promising approach for designing novel selective inhibitors is, with the help of molecular modeling studies. Synthesizing new pharmacophores which differ from those of existing agents by incorporating different biologically potent scaffolds which are not only new but also capable of showing enhanced activity of known biological properties. Literature revealed cis and trans 2,5-disubstituted N, N-dialkylpiperazines are synthesized directly from non-activated N-alkyl aryl aziridines (Piera Trinchera et al., 2012, Francesco Affortunato et al., 2008). Sulfonamides and piperazine derivatives represent a class of biologically exciting molecules showing distinguishable pharmacological properties upon a slight change in substitution pattern in their structures (Al-ghorbani et al., 2015). Substituted piperazines are observed to be possessing significant pharmacological action like antihistaminic (Abou-Gharbia, et al., 1995), antimicrobial (Chaudhary et al., 2006), acetylcholine esterase inhibitors (Sugimoto H et al., 2002), antimalarial (Deprez Poulain et al., 2005) and HIV-protease inhibitor (Rossen et al., 1995; I et al., 1994).
In past decade many aryl piperazines such as pyrimidine piperazine buspirone, chlorophenyl piperazine derivatives, nefazodone and much more had touched the stage of clinical application for the treatment of depression, psychosis or anxiety (Fu et al., 2002). Mono piperazine-2,6-diones are found to be very active agents in various areas including pharmaceutical, agrochemical and organic industries (Winter et al., 1982;Of, M et al., 1978), N-substituted succinic acid and piperazine-2, 6-dione exhibit analgesic effect. Our previous studies have shown the biological activity of newly synthesized derivatives of 4-acetylated piperazine-2,6-diones (Svedaite et al., 1999). Several nitrogen and sulphur containing heterocyclic compounds along with sulfonyl functionality have shown a varied range of pharmaceutical properties: as antimalarial (Maloy et al., 2008), anti-tumor (Mohan et al., 2013), antibacterial (Mohan N R et al., 2013), anti-fungal (Mohan et al., 2013), anti-helminthic (Mohan et al., 2013), anti-oxidant (Saingar et al., 2011), anti-HIV (Mallesha et al., 2011) activities. Prompted by the above findings and need for better lead molecules, we plan to synthesise a novel series of substituted piperazine sulfonamides by incorporating pharmacologically compatible piperazine attaching to sulfonamide moiety as shown in the scheme 1, a simple and efficient method to evaluate their biological potency.
Materials and Methods
Melting points reported determined were using open capillary tubes. Structures of the synthesized compounds were characterized using Infrared Spectroscopy, Proton Nuclear Magnetic Resonance Spectroscopy, 13-carbon Nuclear Magnetic Resonance Spectroscopy. IR Spectra was recorded on Jasco FT-IR Spectrometer, 1H and 13C NMR were recorded in DMSO-d6 at 399.65 MHz and 100.40 MHz respectively. All the chemical shifts were reported in parts per million (ppm). Elemental analysis was executed on Elementar Vario MICRO cube. All chemicals were purchased from Merk India, Spectrochem, and Sigma-Aldrich. Solvents used were of lab grade. The purity of the compounds was confirmed using pre-coated silica gel TLC plates, further purification was done by means of column chromatography. All the bacterial strains were procured from CSIR – National Chemical Laboratory (NCL), Pune, India.
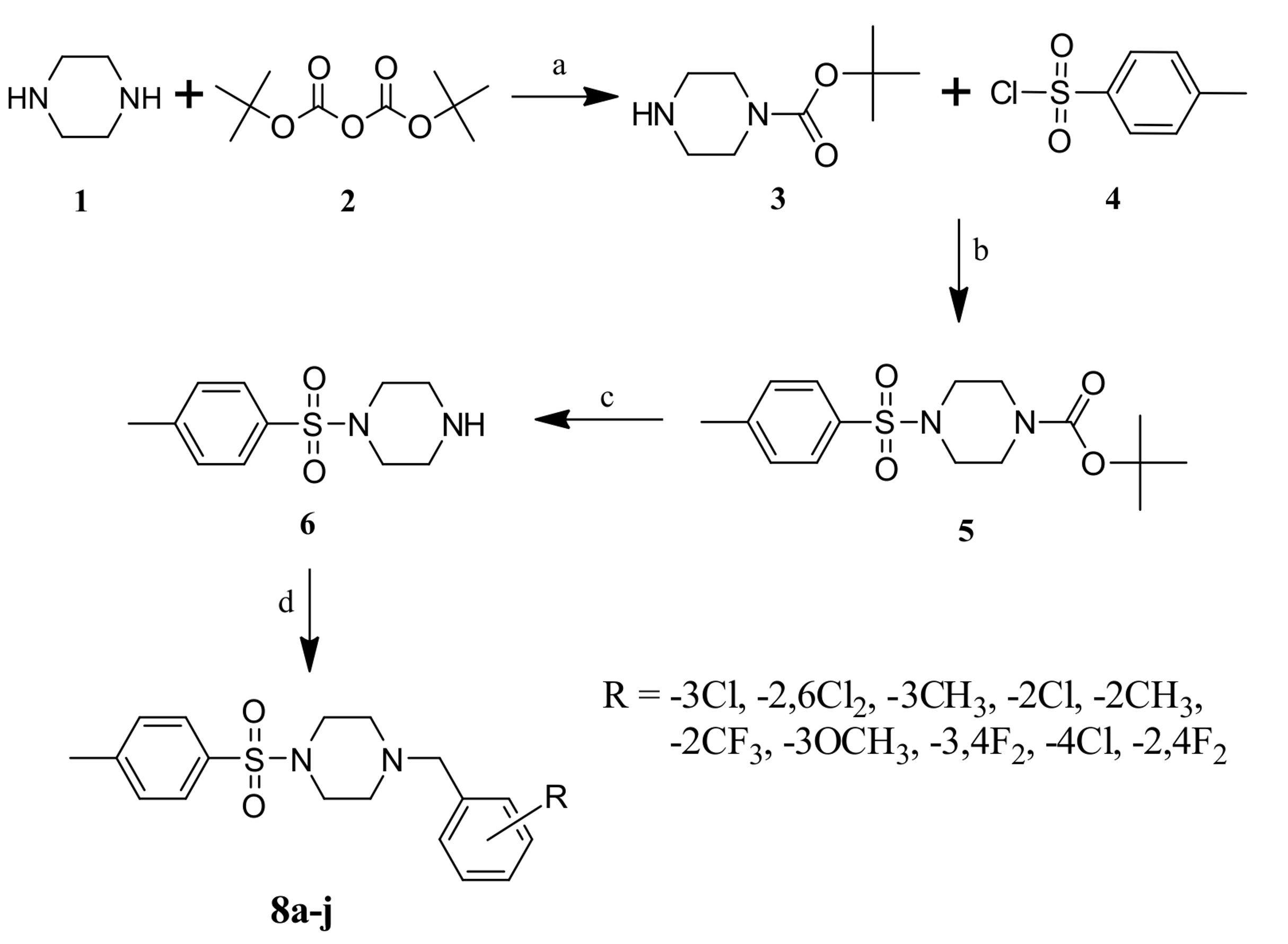
Figure 1. Scheme (1) Reagents and conditions- a) K2CO3/MDC, 6h; b) TEA/MDC, 4h; c) TFA, 2h; d) 7a-j substituted benzyl bromides, K2CO3/MDC, 8h
Synthesis of tert-butyl piperazine-1-carboxylate (3)
A mixture of piperazine (1) (0.13 mol) and K2CO3 (0.39 mol) in 250 mL of methylene dichloride were stirred for 30 min at room temperature, slow addition of Boc anhydride (2) (0.13 mol) to the reaction mixture over a period of 1h and stirring was continued for 6h. Completion of the reaction was monitored by TLC, contents were filtered, the filtrate was washed with water and with brine, dried over anhydrous MgSO4 to get compound (3) as white solid. IR (KBr, cm-1): 1698 (CO); 1H-NMR (CDCl3) δ ppm: 3.80 (s, 1H, NH), 3.51-3.48 (t, 4H, H2C-N-CH2), 2.97-2.94 (t, 4H, H2C-N-CH2), 1.40 (s, 9H, 3CH3); MP: 107-108 oC; Yield: 85%.
Synthesis of tertbutyl 4-[(4-methylphenyl)sulfonyl] piperazine-1-carboxylate (5)
Compound (3) (0.11 mol) in dichloromethane, Triethylamine (TEA) (0.33 mol) and para-toluenesulfonyl chloride (4) (0.11 mol) and the reaction was kept for stirring at room temperature for 4h. The progress of the reaction was monitored by TLC, the reaction mixture was washed with water, brine, dried over Na2SO4 to get the crude compound, it was further purified using column chromatography to obtain tert-butyl-4-[(4-methylphenyl) sulfonyl]piperazine-1-carboxylate (5). 1H-NMR (CDCl3) δ ppm: 7.42-7.40 (d, 2H, J = 7.5 Hz, Ar-H), 7.38-7.36 (d, 2H, J = 7.3 Hz, Ar-H), 3.92-3.81 (t, 4H, H2C-N-CH2), 3.75-3.70 (t, 4H, H2C-N-CH2), 2.34 (s, 3H, Ar-CH3), 1.39 (s, 9H, 3CH3); MP: 190-191oC. Yield: 88%.
Synthesis of 1-[(4-methylphenyl)sulfonyl]piperazine (6)
Solution of tertbutyl-4-[(4-methylphenyl)sulfonyl]piperazine-1-carboxylate (5) in trifluoroacetic acid stirred at 50 oC for 2h. Reaction completion was monitored by TLC; excess solvent was removed under pressure and triturated with petroleum ether to obtained compound (6) as trifluoroacetate salt white solid. 1H NMR (CDCl3) δ ppm: 78-7.76 (d, 2H, J = 7.5 Hz, Ar-H), 7.29-7.28 (d, 2H, J = 7.3 Hz, Ar-H), 3.8 (s, 1H, NH), 3.57-3.25 (m, 8H, (H2C-N-CH2)2), 2.49 (s, 3H, Ar-CH3); MP: 211-213 oC; Yield 42 %.
General Procedure for the synthesis of 1-(substituted)-4-[(4-methylphenyl) sulfonyl]piperazine (8a-j)
Equimolar quantities of 1-[(4-methylphenyl)sulfonyl]piperazine (6) (0.001 mol), different substituted benzyl bromide (7a-j) (0.001 mol) and K2CO3 (0.003 mol) are taken in methylene dichloride (MDC) and was stirred for 8h, reaction completion was monitored by TLC, contents of the RB was filtered, concentrated and crude obtained was purified by column chromatography using (7:3) petroleum ether/ethyl acetate as an eluent to get alkylated piperazine containing sulfonyl nucleus (8a-j) in high yield. Physical data of all the final compounds are entered in Table 1.
Table 1. Physical data of sulfonyl piperazine derivative (8a-j)
|
Comp. Code |
Molecular Formula |
R |
Formula Weight |
Melting Point (oC) |
Yield (%) |
|
8a |
C18H21ClN2O2S |
-3Cl |
364.8 |
145 |
55 |
|
8b |
C18H20Cl2N2O2S |
-2,5Cl2 |
399.3 |
138 |
62 |
|
8c |
C19H24N2O2S |
-3CH3 |
344.4 |
187 |
61 |
|
8d |
C18H21ClN2O2S |
-2Cl |
364.8 |
197 |
76 |
|
8e |
C19H24N2O2S |
-2CH3 |
344.4 |
178 |
52 |
|
8f |
C19H21F3N2O2S |
-2CF3 |
398.4 |
158 |
68 |
|
8g |
C19H24N2O3S |
-3OCH3 |
360.4 |
132 |
71 |
|
8h |
C18H20F2N2O2S |
-3,4F2 |
366.4 |
156 |
58 |
|
8i |
C18H21ClN2O2S |
-4Cl |
364.8 |
143 |
57 |
|
8j |
C18H20F2N2O2S |
-2,4F2 |
366.4 |
167 |
64 |
tert-butyl piperazine-1-carboxylate (3): IR (KBr, cm-1): 1698 (CO); 1H-NMR (CDCl3) δ ppm: 3.80 (s, 1H, NH), 3.51-3.48 (t, 4H, H2C-N-CH2), 2.97-2.94 (t, 4H, H2C-N-CH2), 1.40 (s, 9H, 3CH3); MP: 107-108 oC; Yield: 85%.
tert-butyl 4-tosylpiperazine-1-carboxylate (5): 1H-NMR (CDCl3) δ ppm: 7.64-7.62 (d, 2H, J = 7.5 Hz, Ar-H), 7.34-7.32 (d, 2H, J = 7.3 Hz, Ar-H), 3.51-3.48 (t, 4H, (-CH2)2), 2.9-2.94 (t, 4H, (-CH2)2), 2.43 (s, 3H, -CH3), 1.40 (s, 9H, 3CH3); 13C NMR (CDCl3) δ ppm: 153.8, 143.6, 132.2, 129.5, 127.5, 80.0, 45.6, 43.4, 28.0, 21.2; MP: 190-1910C. Yield: 88%.
1-[(4-methylphenyl)sulfonyl]piperazine (6): 1H NMR (CDCl3) δ ppm: 8.94 (s, 1H, NH), 7.66-7.64 (d, 2H, J = 7.5 Hz, Ar-H), 7.49-7.47 (d, 2H, J = 7.3 Hz, Ar-H), 3.18-3.17 (t, 4H, H2C-N-CH2), 3.08-3.06 (t, 4H, H2C-N-CH2), 2.40 (s, 3H, Ar-CH3); 13C NMR (CDCl3) δ ppm: 144.46, 131.4, 130.1, 127.8, 43.0, 42.3, 20.9; LCMS: 241.3 (M+1). MP: 211-213 oC; Yield 42 %.
1-(3-chlorobenzyl)-4-tosylpiperazine (8a): IR (KBr, cm-1) S=O 1302.64 (Asymmetric), 1152.19 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.77-7.75 (d, 2H, J = 7.5 Hz, Ar-H), 7.47 (s, 1H, Ar-H), 7.45 – 7.42 (d, 2H, J = 7.3 Hz, Ar-H), 7.32-7.29 (m, 1H, Ar-H), 7.25-7.23 (d, 1H, J = 7.1 Hz, Ar-H), 7.21-7.0 (d, 1H, J = 7.1 Hz, Ar-H), 3.66 (s, 2H, -CH2-), 3.10-3.06 (t, 4H, H2C-N-CH2), 2.42-2.39 (t, 4H, H2C-N-CH2), 2.34 (s, 3H, -CH3);. 13C NMR (CDCl3) δ ppm: 143.3, 137.6, 136.9, 134.0, 132.2, 129.3, 128.3, 127.3, 126.9, 126.0, 63.0, 53.6, 48.8, 21.1. Elemental Analysis Calculated (Found) % C: 59.20 (59.22), H: 5.76 (5.74), N: 7.67 (7.65).
1-(2,6-dichlorobenzyl)-4-tosylpiperazine (8b): IR (KBr, cm-1) S=O 1312.24 (Asymmetric), 1155.79 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.76-7.74 (d, 2 H, J = 7.5 Hz, Ar-H), 7.53- 7.50 (d, 2H, J= 6.8, Ar-H), 7.45 - 7.39 (m, 3H, Ar-H), 3.65 (s, 2H, -CH2-), 3.11-3.05 (t, 4H, H2C-N-CH2), 2.40-2.37 (t, 4H, H2C-N-CH2), 2.35 (s, 3H, Ar-CH3). 13C NMR (CDCl3) δ ppm: 143.3, 137.6, 137.1, 135.5, 129.3, 129.0, 128.3, 126.6, 53.6, 52.2, 48.8, 21.2. Elemental Analysis Calculated (Found) % C: 54.14 (54.12), H: 5.05 (5.03), N: 7.00 (7.00).
1-(3-methylbenzyl)-4-tosylpiperazine (8c): IR (KBr, cm-1) S=O 1362.54 (Asymmetric), 1162.29 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.75-7.71 (d, 2H, J = 7.3 Hz, Ar-H), 7.47 (s, 1H, Ar-H), 7.43- 7.40 (d, 2H, J =7.5 Hz, Ar-H), 7.30 - 7.24 (m, 3H, Ar-H), 3.58 (s, 2H, -CH2-), 3.10-3.06 (t, 4H, H2C-N-CH2), 2.37-2.33 (t, 4H, H2C-N-CH2), 2.30 (s, 3H, -CH3); 13C NMR (CDCl3) δ ppm: 143.3, 138.5, 138.1, 137.6, 130.7, 129.3, 128.3, 127.5, 125.8, 64.7, 53.6, 52.60, 48.8, 21.6, 21.4; Elemental Analysis Calculated (Found) % C: 66.25 (66.23), H: 7.01 (6.99), N: 8.13 (8.11).
1-(2-chlorobenzyl)-4-tosylpiperazine (8d): IR (KBr, cm-1) S=O 1328.56 (Asymmetric), 1162.79 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.71-7.69 (d, 2H, J = 6.7 Hz, Ar-H), 7.65- 7.60 (m, 4H, Ar-H), 7.32 - 7.30 (d, 2H, J = 6.9 Hz, Ar-H), 3.64 (s, 2H, -CH2-), 3.05-3.02 (t, 4H, H2C-N-CH2), 2.33-2.29 (t, 4H, H2C-N-CH2), 2.34 (s, 3H, -CH3). 13C NMR (CDCl3) δ ppm: 143.3, 137.6, 136.3, 134.1, 130.2, 129.3, 128.6, 128.3, 126.5, 57.3, 53.6, 48.8, 21.0. Elemental Analysis Calculated (Found) % C: 59.23 (59.21), H: 5.79 (5.77), N: 7.68 (7.66).
1-(2-methylbenzyl)-4-tosylpiperazine (8e): IR (KBr, cm-1) S=O 1313.34 (Asymmetric), 1167.89 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.73-7.70 (d, 2H, J = 7.2 Hz, Ar-H), 7.40- 7.34 (d, 2H, J =7.5 Hz, Ar-H), 7.32 - 7.30 (m, 4H, Ar-H), 3.60 (s, 2H, -CH2-), 3.03-3.01 (t, 4H, H2C-N-CH2), 2.30-2.28 (t, 4H, H2C-N-CH2), 2.30 (s, 3H, -CH3); 13C NMR (CDCl3) δ ppm: 143.3, 13.6, 130.1, 129.9, 129.3, 128.3, 127.1, 125.4, 59.9, 53.6, 52.02, 48.5, 21.5, 18.8; Elemental Analysis Calculated (Found) % C: 66.22 (66.24), H: 7.03 (7.01), N: 8.13 (8.13).
1-tosyl-4-(2-(trifluoromethyl)benzyl)piperazine (8f): IR (KBr, cm-1) S=O 1378.14 (Asymmetric), 1192.49 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.68-7.65 (d, 2H, J = 7.5 Hz, Ar-H), 7.53- 7.40 (d, 2H, J =7.8Hz, Ar-H), 7.19 - 7.13 (m, 4H, Ar-H), 3.59 (s, 2H, -CH2-), 2.89-2.85 (t, 4H, H2C-N-CH2), 2.31-2.28 (t, 4H, H2C-N-CH2), 2.27 (s, 3H, -CH3). 13C NMR (CDCl3) δ ppm: 144.3, 136.6, 131.4, 130.6, 129.2, 128.4, 127.6, 127.4, 125.9, 123.2, 58.9, 54.6, 47.8, 20.2. Elemental Analysis Calculated (Found) % C: 57.27 (57.23), H: 5.32 (5.28), N: 7.01 (7.01).
1-(3-methoxybenzyl)-4-tosylpiperazine (8g): IR (KBr, cm-1) S=O 1333.11 (Asymmetric), 1166.11 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.69-7.65 (d, 2H, J = 7.5 Hz, Ar-H), 7.40- 7.37 (d, 2H, J = 5 Hz, Ar-H), 7.01 (s, 1H, Ar-H), 6.80 – 6.77 (m, 3H, Ar-H) 3.83 (s, 3H –O-CH3), 3.69 (s, 2H, -CH2-), 3.59-3.55 (t, 4H, H2C-N-CH2), 2.30-2.27 (t, 4H, H2C-N-CH2), 2.27 (s, 3H, -CH3); 13C NMR (CDCl3) δ ppm: 160.3, 144.5, 141.9, 137.7, 129.3, 129.1, 128.3, 121.1, 112.9, 112.9, 64.7, 55.8, 53.6, 46.8, 22.1; Elemental Analysis Calculated (Found) % C: 63.32 (63.28), H: 6.71 (6.69), N: 7.77 (7.75).
1-(3,4-difluorobenzyl)-4-tosylpiperazine (8h): IR (KBr, cm-1) S=O 1372.44 (Asymmetric), 1162.59 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.72-7.69 (d, 2H, J = 5.5 Hz, Ar-H), 7.40- 7.36 (d, 2H, J = 5.1 Hz, Ar-H), 7.16 - 7.10 (d, 1H, J = 8.0 Hz, Ar-H), 6.92-6.80 (d, 1H, J =7.4 Hz, Ar-H), 6.75 (s, 1H, Ar-H), 3.57 (s, 2H, -CH2-), 3.49-3.45 (t, 4H, H2C-N-CH2), 2.33-2.29 (t, 4H, H2C-N-CH2), 2.20 (s, 3H, -CH3). 13C NMR (CDCl3) δ ppm: 149.2, 148.0, 143.3, 137.5, 132.9, 129.2, 128.5, 126.0, 117.4, 116.6, 64.5, 53.5, 46.3, 22.8. Elemental Analysis Calculated (Found) % C: 59.00 (59.00), H: 5.50 (5.46), N: 7.64 (7.62).
1-(4-chlorobenzyl)-4-tosylpiperazine (8i): IR (KBr, cm-1) S=O 1326.14 (Asymmetric), 1123.10 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.80-7.78 (d, 2H,J = 6.5 Hz, Ar-H), 7.41- 7.32 (d, 2H, J = 6.5 Hz, Ar-H),7.21-7.0 (d, 2H, J = 7.5 Hz, Ar-H), 6.90-6.88 (d, 2H, J = 6.5 Hz, Ar-H), 3.57 (s, 2H, -CH2-), 3.10-3.06 (t, 4H, H2C-N-CH2), 2.36-2.33 (t, 4H, H2C-N-CH2), 2.36 (s, 3H, -CH3). 13C NMR (CDCl3) δ ppm: 143.5, 137.6, 136.7, 132.8, 131.2, 129.8, 128.6, 128.3, 69.1, 55.4, 44.8, 23.3. Elemental Analysis Calculated (Found) % C: 59.20 (59.22), H: 5.80 (5.88), N: 7.68 (7.66).
1-(2,4-difluorobenzyl)-4-tosylpiperazine (8j): IR (KBr, cm-1) S=O 1322.23 (Asymmetric), 1143.19 (Symmetric); 1H NMR (CDCl3) δ ppm: 7.74-7.71 (d, 2H, J = 6.3 Hz, Ar-H), 7.43- 7.38 (d, 2H, J = 6.1 Hz, Ar-H), 6.89 (m, 2H, Ar-H), 6.56 (m, 1H, Ar-H), 3.58 (s, 2H, -CH2-), 3.09-3.05 (t, 4H, H2C-N-CH2), 2.39-2.36 (t, 4H, H2C-N-CH2), 2.37 (s, 3H, -CH3); 13C NMR (CDCl3) δ ppm: 162.9, 159.7, 143.7, 137.7, 132.2, 129.3, 128.4, 121.5, 110.8, 104.4, 55.6, 53.9, 48.9, 21.3. Elemental Analysis Calculated (Found) % C: 58.99 (58.97), H: 5.48 (5.46), N: 7.60 (7.62).
In vitro biological activity
Compounds (8a-j) showed significant inhibitory action against tested strains. All the derivatives showed inhibition against Escherichia coli and Staphylococcus aureus. Initially different concentrations of 5, 15, 25, 35 and 50 µg/50µL have been tried. At 50 µg/50µL minimum inhibitory concentration (MIC) was observed. Data present in Table 2a showed that the derivatives are active at and above 50 µg/50µL. Among all derivatives of the series evaluated for antibacterial activity, 8a, 8b, 8e, and 8f presented substantial activity against Escherichia coli and Staphylococcus aureus and all others were moderately active against tested strains compared to standard Chloramphenicol.
Biological Activities
Antibacterial activity: Compounds 8a-j were screened for their antibacterial activity. Bacterial strains were acquired from CSIR-National Chemical Laboratory (NCL) Pune and Microbial Type Cell culture (MTCC), Chandigarh. Agar well diffusion technique (Mohan et al., 2014) was used for the study of bacterial strains broth culture. In the sterilized condition, from the working culture, a small amount of culture was taken into 10–15 ml sterile normal saline (0.9% NaCl solution) and was gently mixed. Approximately 0.5 ml of inoculums and melted agar cooled to 45 °C was added to the sterilized Petri dish and was allowed to solidify. Bacterial strains incubated for 24 h and were uniformly smeared on sterile nutrient agar medium in each petri dishs using sterile L-Shaped glass rod. Five uniform wells of 6 mm diameter were bored using cork borer to accommodate 50 µl of the solution in each well. Test samples were dissolved in dimethyl sulfoxide, a negative control and Chloramphenicol (10 µg/50 µL) a positive control was taken as a standard drug, purchased from Himedia, Mumbai. The concentration of 50 µg/50 µL per well was used to assess the activity. Sterile micropipette tips were used to load the wells with the right amount of sample, control, and standard. After inoculation plates were incubated at 37 °C for 36 h. After the incubation period, zone of inhibition diameter for each well was measured in mm. The MIC and experiment performed in triplicates the average values are tabulated in Table 2a and 2b.
Table 2a. Antibacterial activity Minimum Inhibitory Concentration (MIC) values of sulfonyl piperazines (8a-j)
|
Bacterial Stains |
Conc. (µg /50µL) |
Compound Codes (8a-j) |
|
Bacterial Growth |
||
|
Escherichia coli |
5 |
+ |
|
15 |
+ |
|
|
25 |
+ |
|
|
35 |
+ |
|
|
50 |
- |
|
|
Staphylococcus aureus |
5 |
+ |
|
15 |
+ |
|
|
25 |
+ |
|
|
35 |
+ |
|
|
50 |
- |
|
+: Presence of Growth; -: Absence of Growth
Table 2b. Antibacterial activities Zone of Inhibition of compounds (8a-j)
|
Code |
Conc. (µg 50 µL-1) |
E. coli (Mean±SE) |
S. aureus (Mean±SE) |
|
8a |
50 |
7.38±0.55** |
4.22±0.30** |
|
8b |
50 |
7.08±0.34** |
4.80±0.33** |
|
8c |
50 |
6.08±0.62** |
1.92±0.15** |
|
8d |
50 |
4.32±0.27** |
4.00±0.41** |
|
8e |
50 |
5.38±0.24** |
5.08±0.42** |
|
8f |
50 |
7.72±0.30** |
3.15±0.16** |
|
8g |
50 |
5.38±0.23** |
1.95±0.20** |
|
8h |
50 |
6.00±0.41** |
2.22±0.29** |
|
8i |
50 |
5.78±0.76** |
3.28±0.25** |
|
8j |
50 |
5.65±0.37** |
2.80±0.16** |
|
CAM |
10 |
13.00±0.58 |
15.17±0.44 |
|
Control |
- |
- |
- |
(Values are the mean ± SEM of the clear zone. Symbols represent statistical significance, *P˂ 0.05, ** P˂0.01 as compared with the control group). Standard CAM: Chloramphenicol
Antifungal Activity: Antifungal activities of all piperazine derivatives towards two mold fungi were studied, viz. Candida albicans (human pathogen) Aspergillus flavus (mold). Poisoned food technique method was used to assess the antifungal activity of the synthesized compounds (Mohan N R et al., 2014) and Nystatin (10µg/disc) as a standard fungicide. Basal medium used for test fungi was Potato Dextrose Agar (PDA) it was prepared by using Potato, Dextrose, and Agar. First, crushed potatoes were boiled in water to get potato soup. The filtered soup has been mixed up with Dextrose and Agar. It is then sterilized by autoclave at 121oC for 20 minutes and was cooled (~ 45 oC). 15 mL of sterilized melted PDA medium was poured into each sterilized petri dish. After solidification, small portions of the mycelium of each fungus were inoculated carefully on each PDA plate with the help of sterilized L-shaped rod. Plates were incubated at (25 ± 2) oC for five days. Five uniform wells with 6 mm diameter were bored using sterile cork borer to accommodate 50 µL of the solution in each. Samples were dissolved in dimethyl sulfoxide (DMSO) a negative control, Nystatin a positive control was taken as a standard drug, purchased from Himedia, Mumbai. The concentration of 50µg/50µL per well was used to assess the activity. Sterile micropipette tips were used to load the wells with the right amount of sample, control, and standard. The plates were then kept at 4 oC for 24 h to provide sufficient time to diffuse over a considerable area of the plates. After 24h plates were incubated at 280 C for 72h. After the incubation period, Diameter of the zone of inhibition in mm was measured for each well. The MIC and experiment performed in triplicates the average values are tabulated in Table 3a and 3b.
Table 3a. Antifungal activity Minimum Inhibitory Concentration (MIC) values of sulfonyl piperazines (8a-j)
|
Bacterial Stains |
Conc. (µg /50µL) |
Compound Codes (8a-j) |
|
Bacterial Growth |
||
|
Candida albicans |
5 |
+ |
|
15 |
+ |
|
|
25 |
+ |
|
|
35 |
+ |
|
|
50 |
- |
|
|
Aspergillus flavus |
5 |
+ |
|
15 |
+ |
|
|
25 |
+ |
|
|
35 |
+ |
|
|
50 |
- |
|
Table 3b. Antifungal activities Zone of Inhibition of compounds (8a-j)
|
Code |
Conc. (µg 50 µL-1) |
C. albicans (Mean±SE) |
A. flavus (Mean±SE) |
|
8a |
50 |
13.92±0.24** |
16.44±0.63** |
|
8b |
50 |
20.67±0.42** |
19.56±0.52** |
|
8c |
50 |
18.75±0.17** |
17.87±0.13** |
|
8d |
50 |
- |
- |
|
8e |
50 |
15.24±0.11** |
18.55±0.26** |
|
8f |
50 |
- |
- |
|
8g |
50 |
12.67±0.25** |
8.78±0.43** |
|
8h |
50 |
13.30±0.51** |
14.34±0.79** |
|
8i |
50 |
14.10±0.62** |
12.23±0.23** |
|
8j |
50 |
9.52±0.29** |
13.20±0.11** |
|
NYS |
10 |
29.33±0.13 |
21.33±0.23 |
|
Control |
- |
- |
- |
(Values are the mean ± SEM of the clear zone. Symbols represent statistical significance, *P˂ 0.05, ** P˂0.01 as compared with the control group). Standard NYS: Nystatin
Anthelmintic activity: Anthelmintic activity of compounds 8a-j were evaluated using Pheretima posthuma (Indian Earthworm), which were maintained under the normal vermicomposting system for about three weeks with a suitable source of nutrition and water. Adult earthworms of about 4 cm in length and 0.3 - 0.4 cm in width were chosen. Concentrations of 50 mg of samples were evaluated as per the standard method reported (Mohan et al., 2014). Five sets each with six earthworms were taken. Each worm was washed individually with normal saline and was placed into a 20 ml of normal saline. I Group earthworms were placed in 20 ml saline in a sterile petri plate and II Group earthworms were placed in 20 ml saline with standard drug piperazine citrate (50 mg/ml). Similarly, III to XXII Group's worms was placed in a 20 ml saline containing 50 mg/ml of test samples respectively. Time is taken for paralysis and time taken for death as objective and was documented in minutes. Nonrevival body state in the normal saline medium was analyzed paralysis time. Total loss of motility with faded body color was concluded as death based on observation results are shown in Table 4a and 4b.
Table 4a. Anthelmintic activity Minimum Concentration (MCs) values of sulfonyl piperazines (8a-j)
|
Test model |
Conc. (mg mL-1) |
Compound Codes (8a-j) |
|
Activity |
||
|
Pheretima posthuma |
15 |
IA |
|
25 |
IA |
|
|
35 |
IA |
|
|
45 |
IA |
|
|
50 |
AC |
|
AC: Active; IA: Inactive
Table 4b. Anthelmintic activity of sulfonyl piperazine derivatives (8a-j)
|
Test Samples Concentration (50 mg mL-1) |
Time taken for paralysis (min) |
Time taken for death (min) |
|
Control |
142.33±0.49 |
167.17±0.87 |
|
PC |
49.17±0.48** |
47.00±0.58** |
|
8a |
61.00±1.46** |
101.00±0.86** |
|
8b |
60.33±1.23** |
87.17±0.60** |
|
8c |
70.83±0.95** |
94.50±1.26** |
|
8d |
77.17±0.60** |
111.67±0.95** |
|
8e |
64.17±0.48** |
97.33±0.67** |
|
8f |
71.33±1.48** |
102.17±0.87** |
|
8g |
61.33±1.33** |
97.33±0.67** |
|
8h |
66.50±0.76** |
97.83±0.48** |
|
8i |
74.67±1.50** |
104.33±1.56** |
|
8j |
61.001.06** |
101.17±0.48** |
(Values are the mean ± SEM of three earthworms. Symbols represent statistical significance, *P˂ 0.05, ** P˂0.01 as compared with the control group) Standard PC: Piperazine citrate
Anti-inflammatory activity: Effect of compounds 8a, 8b, 8c, 8e and 8f on carrageenan-induced paw edema (Mohan et al., 2014) on Albino Wistar rats of either sex having the weight of 90 to 120 g were studied. The animals are maintained under standard laboratory condition for a week. The animals are fed with filtered water and standard pelleted food. Studies conducted were approved by the institutional Animal Ethics Committee (IAEC) of Sree Siddaganga College of Pharmacy, Tumkur (Ref No: SSCPT/IAEC.Clear/141/2012-13). Indomethacin (standard drug) was procured from Fine Chem. Industries Research Lab. The concentration of test samples used was 100 mg/kg of body weight and indomethacin 10 mg/kg of body weight. 1% carboxymethyl cellulose (CMC) was used as a vehicle for the samples and fed to animals through the oral route. Six animals in each set of seven groups were prepared fastened overnight. Group, I aided as control and received carboxymethyl cellulose vehicle only. Indomethacin (10 mg/kg) was fed to Group II through the oral route. III to VII Groups was administered with compounds of 8a, 8b, 8c, 8e and 8f (100 mg/kg). After administration of samples, test models were observed 30 minutes for a clinical sign. After the period suspension of Carrageenan (0.1 ml of 1% w/v) was injected into the subplantar region of the right hind paw of each testing model. Instantly after inoculation, the paw size was measured using digital plethysmometer. Again it was repeated at 30, 60, 120 and 180 minutes time intervals and are reported in Table 5.
Table 5. Effect of 8a-j in Carrageenan-Induced Paw Edema on Wistar Rats
|
Treatment (Dose mg kg-1 body weight) |
Values are expressed in Time in Minutes (MEAN± S.E.M) |
||||
|
0 min |
30 min |
60 min |
120 min |
180 min |
|
|
Carrageenan (1%, 0.1 ml) |
0.01 ± 0.10 |
0.21 ± 0.100 |
0.42 ± 0.02 |
0.53 ± 0.02 |
0.49 ± 0.02 |
|
8a (100 mg kg-1 of b.w) |
0.02 ± 0.02 |
0.32 ± 0.065 |
0.18 ± 0.03*** |
0.07 ± 0.00*** |
0.05 ± 0.04 |
|
8b (100 mg kg-1 of b.w) |
0.01 ± 0.06 |
0.23 ± 0.028 |
0.12 ± 0.03*** |
0.06 ± 0.04*** |
0.02 ± 0.01 |
|
8c (100 mg kg-1 of b.w) |
0.05 ± 0.01 |
0.44 ± 0.062 |
0.50 ± 0.07** |
0.38 ± 0.06** |
0.22 ± 0.03 |
|
8d (100 mg kg-1 of b.w) |
0.02 ± 0.07 |
0.35 ± 0.04 |
0.18 ± 0.03*** |
0.07 ± 0.01*** |
0.06 ± 0.02 |
|
8e (100 mg kg-1 of b.w) |
0.06 ± 0.02 |
0.48 ± 0.074 |
0.54 ± 0.01** |
0.42 ± 0.03** |
0.26 ± 0.02 |
|
8f (100 mg kg-1 of b.w) |
0.04 ± 0.02 |
0.35 ± 0.042 |
0.18 ± 0.04*** |
0.10 ± 0.02*** |
0.04 ± 0.03 |
|
8g (100 mg kg-1 of b.w) |
0.03 ± 0.02 |
0.49 ± 0.07 |
0.53 ± 0.01** |
0.41 ± 0.01** |
0.20 ± 0.02 |
|
8h (100 mg kg-1 of b.w) |
0.02 ± 0.04 |
0.34 ± 0.04 |
0.20 ± 0.03*** |
0.09 ± 0.00*** |
0.05 ± 0.04 |
|
8i (100 mg kg-1 of b.w) |
0.06 ± 0.04 |
0.41 ± 0.01 |
0.16 ± 0.03*** |
0.07 ± 0.02*** |
0.07 ± 0.04 |
|
8j (100 mg kg-1 of b.w) |
0.01 ± 0.02 |
0.44 ± 0.06 |
0.48 ± 0.07** |
0.06 ± 0.03*** |
0.04 ± 0.03 |
|
INDO (10 mg kg-1 of b.w) |
0.03 ± 0.01 |
0.26 ± 0.02 |
0.12 ± 0.02 |
0.04 ± 0.06 |
0.01 ± 0.06 |
(Change in paw size values are expressed in mm (MEAN± S.E.M). for six animals in each group. ANNOVA followed by Dunnett’s Multiple Comparison Test. Values are statistically **p˂0.01 and ***p˂0.001 when compared with carrageenan control). Standard: INDO Indomethacin
Statistical analysis
The data of antimicrobial and anthelmintic was expressed as Mean±S.E of triplicates and six Pheretima posthuma in each group respectively. The difference in values at **p≤0.01 was considered as statistically significant. The analysis of variance (ANOVA) was performed using ezANOVA (version 0.98) software to determine the mean and standard error of the inhibition zone in antimicrobial activity and standard error of paralysis and death time of earthworms. Change in paw size values were calculated using the following equation,
VMM³ = VML ×1 000
Results and discussion
Initially Piperazine (1) undergoes substitution reaction with di tertiary butyl di carbonate (2) in the presence of potassium carbonate in acetonitrile to get tertiary–butyl piperazine–1–carboxylate (3), which is then treated with 4–methyl–benzene sulfonyl chloride (4) in the presence of triethyl amine in methylene di chloride to acquire tertiary–butyl–4[(4–methyl phenyl)sulfonyl]piperazine–1–carboxylate (5). Deprotection of Boc group from a secondary amine of compound (5) was done by using trifluoroacetic acid in the presence of methylene dichloride to get 1-[(4–methyl phenyl)sulfonyl]piperazine (6). Equimolar quantities of 1-[(4-methyl phenyl) sulfonyl] piperazine (6), different substituted benzyl bromide (7a-j) and K2CO3 taken in MDC. Stirred the reaction mixture for 8h, TLC technique was used to was used to monitored completion of the reaction, reaction mixture was filtered, concentrated to get crude compound which was purified by column chromatography to get alkylated piperazine containing sulfonyl nucleus (8a-j) in good yield. Pure compounds were used to carry out the biological evaluation work.
In vitro biological activity
All the derivatives (8a-j) showed significant inhibition against Escherichia coli and Staphylococcus aureus. Initially different concentrations of 5, 15, 25, 35 and 50 µg/50µL have been tried. At 50 µg/50µL minimum inhibitory concentration (MIC) was observed. Data present in Table 2a showed that the derivatives are active at and above 50 µg/50µL. Among all derivatives of the series evaluated for antibacterial activity, 8a, 8b, 8e, and 8f presented substantial activity against Escherichia coli and Staphylococcus aureus and all others were moderately active against tested strains compared to standard Chloramphenicol.
Sulfonyl piperazines (8a-j) showed inhibitory action against C. albicans and A. flavus except for 8d and 8f. Data present in Table 3a showed that the derivatives are active at Minimum Inhibitory Concentration (MIC) 50 µg/50µL. Among all derivatives tested for antifungal potency compounds 8b, 8c and 8e showed significant activity, whereas all other samples observed moderate to good activity against tested molds compared to standard Nystatin.
Anthelmintic activity of the sulfonyl piperazines (8a-j) against Pheretima posthuma (Indian earthworm) showed moderate outcomes with that of standard Piperazine citrate. Earthworms beeing in to control group showed paralysis time at 142.33±0.49 min and death time at 167.17±0.87 min. The concentration of 50 mg/mL was chosen for the study depending the initial different concentrations checked at 15, 25, 35 and 45 mg/mL as given in Table 4a. Standard drug piperazine citrate 50 mg/mL exhibited 49.17±0.48 and 47.00±0.58 min for paralysis and death. Examination of anthelmintic activity revealed that Piperazine derivative 8b, 8c, and 8e showed significant activity against P. posthuma reported in Table 4b
After administration of carrageenan (1%, 0.1ml), increase in right hind paw size was observed after administration of carrageenan (1%, 0.1ml), an increase in appendage size was observed at time intervals 0, 30, 60, 120, and 180 minutes. Samples 8a, 8b, 8c, 8e, 8f, 8g and 8j administered orally (100 mg/kg of body weight) to test system, significantly repressed the paw edema. After 30 minutes break, no change was shown for all the tested samples. But at 60 minute time interval 8a, 8b and 8f showed good activity and compound 8c and 8e showed moderate activity. After 120-minute interval 8a, 8b and 8f showed very good activity compared to 8c, 8e, 8g and 8j. Finally, results obtained at 180 minutes interval all compounds were comparable to standard Indomethacin as reported in Table 5.
Conclusion
In this study new series of compounds were synthesized and evaluated for their antibacterial, antifungal, anthelmintic and anti-inflammatory activity. Out of all tested compounds 8a, 8b, 8c and 8f shows noteworthy antibacterial activity against Escherichia coli and Staphylococcus aureus and 8b, 8c and 8e showed antifungal and other piperazine derivatives 8b, 8c, 8e showed comparable anthelmintic activity compared to standard. The compounds 8a, 8b, and 8f showed significant anti-inflammatory. It can be concluded that aryl sulphonyl piperazine derivatives show great promise as antibacterial, antifungal, anthelmintic and anti-inflammatory agents.
Conflict of interest
The author(s) confirm that this article content has no conflict of interest.
Acknowledgements
The authors are thankful to Tumkur University and University College of Science for the laboratory facilities. Vivek Chandra Mohan wishes to thank the Management, Principal, Director, and Head of the department of the Siddaganga Institute of Technology, Tumkur, Karnataka, India. The authors also appreciate KBITS for their support in providing them with the required computational resources for carrying out this work. This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
References
Abou-Gharbia M, Moyer JA, Nielsen ST, Webb M, Patel U. 1995. New Antihistamines: Substituted Piperazine and Piperidine Derivatives as Novel H1-Antagonists. Journal of Medicinal Chemistry, 38 (20): 4026–4032.
Al-ghorbani M, Bushra Begum A, Zabiulla, Mamatha SV, Khanum SA. 2015. Piperazine and Morpholine : Synthetic Preview and Pharmaceutical Applications. Journal of Chemical and Pharmaceutical Research, 7 (5): 281–301.
Amandeep Kaur Kahlon, Sudeep Roy, Ashok Sharma. 2010. Molecular Docking Studies to Map the Binding Site of Squalene Synthase Inhibitors on Dehydrosqualene Synthase of Staphylococcus aureus. Journal of Biomolecular Structure and Dynamics, 28(2): 201-210.
Chaudhary P, Kumar R, Verma AK, Singh D, Yadav V, Chhillar AK, Sharma GL, Chandra R. 2006. Synthesis and Antimicrobial Activity of N-Alkyl and N-Aryl Piperazine Derivatives. Bioorganic & Medicinal Chemistry, 14(6): 1819–1826.
Deprez Poulain R, Melnyk P. 2005. 1,4-Bis(3-Aminopropyl)Piperazine Libraries: From the Discovery of Classical Chloroquine-Like Antimalarials To the Identification of New Targets. Combinatorial Chemistry and High Throughput Screening, 8(1): 39–48.
Fang W, Robinson DA, Raimi OG, David BE, Harrison JR, Lockhart DEA, Torrie Leah S, Ruda GF, Wyatt PG, Gilbert IH, Van Aalten DMFN. 2015. Myristoyltransferase Is a Cell Wall Target in Aspergillus fumigatus. ACS Chemical Biology, 10(6): 1425-1434.
Francesco Affortunato, Saverio Florio, Renzo Luisi, Biagia Musio. 2008. a-vs Ortho Lithiation of N-Alkylaryl aziridines: Probing the Role of the Nitrogen Inversion Process. Journal of Organic Chemistry, 73: 9214–9220.
Fu X, Tan PZ, Kula NS, Baldessarini R, Tamagnan G, Innis RB, Baldwin RM. 2002. Synthesis, Receptor Potency, and Selectivity of Halogenated Diphenylpiperidines as Serotonin 5-HT2a Ligands for PET or SPECT Brain Imaging. Journal of Medicinal Chemistry, 45 (11): 2319–2324.
Harman CA, Turman MV, Kozak KR, Marnett LJ, Smith WL, Garavito RM. 2007. Structural Basis of Enantioselective Inhibition of Cyclooxygenase-1 by S-α-Substituted Indomethacin Ethanolamides. Journal of Biological Chemistry, 282(38): 28096 -28105.
Jacquelyn G Black, William DO. 1993. Microbiology: Principles and Applications. Prentice Hall, Englewood Cliffs, New Jersey, 360.
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings, WC. 1996. Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature, 384(6610): 644-648.
L I, Askin D, Eng KK, Rossen K, Purick RM, Wells KM, Volante RP, Reider PJ. 1994. Highly Diastereoselective Reaction of a Chiral , Non-Racemic Amide Enolate Synthesis of the Orally Active HIV-L Protease with ( S ) -Glycidyl Tosylate . Tetrahedron Letters, 35 (5): 673–676.
Maloy K P, Gautam P, Srivastava K, Sunil KP. 2008. Design, synthesis and antimalarial activity of benzene and isoquinoline sulfonamide derivatives. Bioorganic & Medicinal Chemistry Letters, 18 (2): 776-781.
Mallesha L, Mohana KN, 2011. Synthesis, antimicrobial and antioxidant activities of 1‐(1,4‐benzodioxane‐2‐carbonyl)piperazine derivatives. European Journal of Chemistry, 2: 193-199.
Mohan NR, Sreenivasa S, Manojkumar KE, Madhu Chakrapani Rao T. 2013. Synthesis and invitro antibacterial activity of some novel Sulfonamide derivatives bearing 1,4-disbstituted-1,2,4-oxadiazole Moiety. Journal of Applied Chemistry, 2: 722-729.
Mohan NR, Sreenivasa S, Manoj Kumar KE, Anitha HC, Madhu Chakrapani Rao T, Thippeswamy BS, Vijay KG. 2013. Synthesis, Characterization And In-Vitro Antimicrobial Study Of Series Of 1-((Substituted Aryl/Alkyl)Sulfonyl)-4-Tosylpiperazines. Indo American Journal of Pharm Research, 3(12): 1513-1520.
Mohan NR, Sreenivasa S, Manojkumar KE, Rao TMC, Thippeswamy BS, Suchetan PA. 2014. Synthesis, Antibacterial, Anthelmintic and Anti-Inflammatory Studies of Novel Methylpyrimidine Sulfonylpiperazine Derivatives. Journal of the Brazilian Chemical Society, 25(6): 1012–1020.
Piera Trinchera, Biagia Musio, Leonardo Degennaro, Anna Moliterni, Aurelia Falcicchiob, Renzo Luisi. 2012. One-pot preparation of piperazines by regioselective ring-opening of non-activated arylaziridines. Organic & Biomolecular Chemistry,10: 1962-1967.
Paul P, Chowdhury A Das, Talukdar A, Choudhury MD. 2015. Homology modeling and molecular dynamics simulation of N-myristoyltransferase from Plasmodium falciparum: an insight into novel antimalarial drug design. Journal of Molecular Modeling, 21(3): 37-41.
Raghavamma, S.T.V, Rama Rao, N. In vitro Evaluation of Anthelmintic Activity of Nauclea orientalis Leaves. Indian Journal of Pharmaceutical Sciences, 2010, 72 (4): 520–521.
Rossen, K, Weissman, S. A, Sager, J, Reamer, R. A, Askin, D, Volante, R. P, Reider, P. J. Asymmetric Hydrogenation of Tetrahydropyrazines: Synthesis of (S)-Piperazine-2-Tert-Butylcarboxamide, an Intermediate in the Preparation of the HIV Protease Inhibitor Indinavir. Tetrahedron Letters, 1995, 36 (36): 6419–6422.
Saingar S, Kumar R, Joshi YC. 2011. Synthesis and biological activity of novel 1H-1,4-diazepines containing benzene sulfonyl piperazine moiety. Medicinal Chemistry Research, 20: 975-980.
Sogabe S, Masubuchi M, Sakata K, Fukami TA, Morikami K, Shiratori Y, Ebiike H, Kawasaki K, Aoki Y, Shimma N, D'Arcy A, Winkler FK, Banner DW, Ohtsuka T. 2002. Crystal structures of Candida albicans N-myristoyltransferase with two distinct inhibitors. Chemistry & Biology, 9(10): 1119-1128.
Sugimoto H, Ogura H, Arai Y, Limura Y, Yamanishi Y. 2002. Research and Development of Donepezil Hydrochloride, a New Type of Acetylcholinesterase Inhibitor. Japanese Journal of Pharmacology, 89 (1): 7–20.
Svedaite I, Udrenaite E, Lauzikiene N, Gaidelis P. 1999. Khim.-Pharm. Zh, 6 Winter FTY, R. EP 1982. Patent, 63: 544.