

Prashant Soni, Alok Pal Jain*
Department of Pharmacology,
RKDF College of Pharmacy, Sarvepalli Radha Krishnan University, Bhopal (M.P.) India
*Address for Corresponding Author
Dr. Alok Pal Jain
Department of Pharmacology,
RKDF College of Pharmacy, Sarvepalli Radha Krishnan University, Bhopal (M.P.) India
Abstract
Objective: The aim of present study was to investigate polyherbal tablet containing methanol extracts of Carissa carandus, Ocimum sanctum, Moringa oleifrea, Manilkara zapota for screening of antidiabetic effects. Material and methods: The dried extracts of all four plants were mixed with different excipient using wet granulation method to prepare tablets using compressing machine. Prepared herbal formulation was tested for in vivo studies for antidiabetic activity by using Alloxan induced diabetes in rats. Effect of formulation was assessed by measurement of biochemical parameters and also estimation of antioxidant level. Results: Preformulation study of the granules showed that all the evaluated parameters were within the acceptable limit. Prepared polyherbal tablet was tested for antidabetic effect in alloxan induced diabetic animals showed significant reduction in body weight, blood glucose level, and lipid profile when compared to the control animals. The serum total cholesterol, and Triglyceride level were significantly (P<0.01) decreases and restore near to the normal level. Polyherbal formulation treated animals reversed the effect of Alloxan tissue antioxidant level. This may be due to the antioxidant mechanism of the individual herbs present in the polyherbal formulation. Conclusion Results of the present study was confirmed that prepared formulation was safe, and effective for antidiabetic activity. It was confirmed that prepared formulation was able to restore lipid profile, blood glucose level and antioxidants level to the nearly of normal. These positive effects may attribute to the presence of various flavonoid constituents present in methanol extracts of all plants. Additionally this formulation also showed antioxidant effect that may another possible mechanism for antidiabetic effect of formulation.
Keywords: Antioxidant, Carissa carandus, Ocimum sanctum, Moringa oleifrea, Manilkara zapota, ployherbal tablets, antidiabetic, Alloxan
Introduction
The World Health Organization (WHO) estimates that 4 billion people, 80% of the world population presently use herbal medicine for some aspect of primary health care. Herbal medicine is a major component in all indigenous peoples’ traditional medicine and a common element in Ayurvedic, Homeopathic, naturopathic, traditional oriental, and Native American Indian medicine. It has been estimated that 70-90% of the world’s population relies on alternative therapies and practices. Herbals are used in the art of healing since the time immemorial. The primitive man through trial and error gained knowledge of herbal and passed it on to the next progeny. It is reasonable to assume that for ten thousands of year herbs were perhaps used for the magical power as well as for their medicinal values (Maiti et al., 2011).
Diabetes mellitus is a chronic metabolic disorder resulting from insulin deficiency, characterized by hyperglycaemia, altered metabolism of carbohydrates, protein and lipids, and an increased risk of vascular complication. The insulin deficiency may be absolute or relative and the metabolic abnormalities lead to the classic symptom of polyuria, polydipsia, polyphagia and fatigue. Long term complication of diabetes mellitus includes gangrene, polyneuropathy and uraemia (Fowler and Williams, 1995).
Both type1 (T1D) and type 2 diabetes (T2D) are associated with significantly accelerated rates of micro vascular complications such as diabetic nephropathy, neuropathy, and retinopathy, and macro vascular cardiovascular diseases such as atherosclerosis, hypertension, and stroke. Diabetic neuropathies are a family of nerve disorders caused by diabetes. People with diabetes can, over time, develop nerve damage throughout the body (Muller, 2003; Suh, 1999).
Diabetes mellitus is induced chemically, surgically or by genetic manipulations. Although, there are some reports that have been shown to use the larger animals to induce diabetes, it is very common to use rodents for the experiments of diabetes (Matsumoto et al., 2009). In other study, diabetes prone strains have been introduced to the animals in regard to induce diabetes. Here on the basis characterization results, we selected a polyherbal tablet formulation containing extracts of Carissa carandus, Ocimum sanctum, Moringa oleifrea, Manilkara zapota plants extracts for further pharmacological screening. Results of pharmacological screening for antidiabetic activity of polyherbal tablets were compared with standard antidiabetic drug.
Carissa carandas is a species of flowering shrub in the dogbane family, Apocynaceae. It produces berry-sized fruits that are commonly used as a condiment in Indian pickles and spices. It is a hardy, drought-tolerant plant that thrives well in a wide range of soils.
The chemical investigations of C. carandas had led to the isolation of several substances including β-sitosterol, lupeol, ursolic acid and a new cardioactive substance; glucosides of odoroside-H (Rastogi et al., 1967). Bark, leaves and fruit contain an unnamed alkaloid. The leaves are reported to have triterpene, tannins and carissic acid (Siddiqui et al., 2033). Fruits of this plant have been reported to contain a mixture of volatile principles like 2-phenyl ethanol, linalool, β-caryophyllene, isoamyl alcohol and benzyl acetate (Chandra, 1972) and a novel (Carissol) triterpenic alcohol (Naim et al., 1985). The karonda fruit is a rich source of iron and contains a fair amount of vitamin C. It is ant scorbutic and very useful for cure of anemia.
Ocimum sanctum, commonly known as holy basil, tulasi or tulsi, is an aromatic perennial plant in the family Lamiaceae. It is native to the Indian subcontinent and widespread as a cultivated plant throughout the Southeast Asian tropics. The major chemical constituents are Eugenol, nerol, eugenol methyl ether, caryophyllene, terpinene-4-ol-decyladehyde, α-selinene, a and β-pinene, Camphor and carvacrol, Cineole, and linalool.
Different parts of Tulsi plant e.g. leaves, flowers, stem, root, seeds etc. are known to possess therapeutic potentials and have been used, by traditional medical practitioners, as expectorant, analgesic, anticancer, antiasthmatic, antiemetic, diaphoretic, antidiabetic, antifertility, hepatoprotective, hypotensive, hypolipidmic and antistress agents (Chopra and Nayer, 1956).
M. oleifera is a fast-growing, deciduous tree that can reach a height of 10–12 m (32–40 ft) and trunk diameter of 45 cm (1.5 ft). The bark has a whitish-grey colour and is surrounded by thick cork.
Benzyl isothiocyanate, β- sitosteryl oleate, Stigmasterol, Oleic acid, Octadecene. The leaves are the most nutritious part of the plant, being a significant source of B vitamins, vitamin C, provitamin A as beta-carotene, vitamin K, manganese, and protein, among other essential nutrients (Chopra et al., 1956; Wealth of India, 1976).
Manilkara zapota, commonly known as the sapodilla is a long-lived, evergreen tree native to southern Mexico, Central America and the Caribbean. The leaves of the sapodilla fruit can also be used as a medicine for inflammatory diseases. The leaves work as an oral anti-inflammatory agent. Take clean sapodilla fruit leaves and then boil them for about ten minutes. This boiled water which contains the extracts of Sapodilla can be used as medicine. It can be used for gargling as well (Wealth of India, 1976).
Sapodilla or chikoo is a natural sedative. It is used to relax the nerves and also to relieve stress. It is often suggested as a part of the diet of those struggling with insomnia and panic disorders.
Materials and methods
Collection and identification of plant materials
The proposed plant drug (Carrisa carandus, Ocimum sanctum, Moringa olefera, Manilkara zapota) were collected from nearest area of Bhopal (M.P.). The Leaves of all plant drugs were shade-dried, powdered into moderately coarse powder and stored in air tight container. Plant specimens were identified and authenticated in Department of Pharmacognosy, RKDF College of Pharmacy, Bhopal. The powder drug of all plants material was used for extraction.
Extraction and phytochemical studies
The powdered drug of Carrisa carandus plant (about 150 g) was defatted with petroleum ether and extracted with methanol (95%) in a soxhlet apparatus for 12 and 24 Hrs respectively. The solvent was removed under reduced pressure, with respect to dried plant material. The dried extract of was stored in a desiccator till further use.
Phytochemical investigation means to investigate the plant material in terms of its active constituents. It involves the identification of active constituents and to identify them qualitatively. Extract obtained from all plant drugs (Carrisa carandus, Ocimum sanctum, Moringa olefera, Manilkara zapota) were subjected to various qualitative tests for the identification of various plant constituents present in this species (Paech and Tracey, 1955; Sim, 1968; Kokate et al., 2002).
Acute oral toxicity study of extracts
The acute oral toxicity studies were carried out as per the guidelines of Organization for Economic Co-operation and Development (OECD), draft guidelines 423 received from Committee for the purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Social Justice and Empowerment, Government of India.
The principle is based on the stepwise procedure with the use of a minimum number of animals per step, sufficient information is obtained on the acute toxicity of the test substance to enable its classification. The test substance is administered orally to the group of experimental animals using the defined doses. The substance is tested using a stepwise procedure, each step consisting of three animals of a single sex (normally females). Absence or presence of extract related mortality of the animals dosed at one step determines the next step.
Animals
Wistar albino rats (150-200g, 8 to 12 weeks old) of either sex were selected for acute toxicity study. They were housed individually in well-ventilated, temperature controlled (26 ±2°C) animal room for seven days of period prior experiment. The animals were given the standard commercial pellet rodent diet (Hindustan Lever Pvt Limited, Bangalore, India) and water ad libitum. The procedures were reviewed and approved by the Institutional Animal Ethics Committee (Reg. No. 1546/PO/E/S/11/CPCSEA).
The animals were randomly selected, marked to permit individual identification, and kept in their cages for 5 days prior to dosing to allow for acclimatization to the laboratory condition.
Animal protocol and administration of doses
All the extracts were prepared as a suspension by triturating with 2% Tween-80. The test substances were administered in a single dose using a gastrointestinal tube. Animals were fasted prior to dosing. Following the fasting period, the animals were weighed and test substance was administered, food was withheld for a further 3-4 hours of post dosing period in rats.
The acute toxicity of all extracts was determined in albino rats. The animals were fasted overnight prior to the experiment. Animals were divided into 8 groups and the extracts were administered orally to various groups of rats in doses ranging from 1000, 1200, 1400, 1600, 18000, 2000, 2200 and 2500 mg/kg for the acute toxicity study (Ghosh, 1971). There was no lethality in any of the groups after 7 days of treatment.
Preparation of Polyherbal tablets
The dried plant extracts of all proposed plants were mixed with different excipient using wet granulation method to prepare later solid pharmaceutical forms. These prepared granules of each form were compressed into tablets using Compressing machine.
Table 1. Composition of Polyherbal tablet formulation
|
Ingredients |
Quantity per tablet (mg) |
|||
|
|
F1 |
F2 |
F3 |
F4 |
|
Carissa Carandus |
100 |
100 |
100 |
100 |
|
Ocimum Sanctum |
100 |
100 |
100 |
100 |
|
Moringa Oleifera |
100 |
100 |
100 |
100 |
|
Manilkara Zapota |
100 |
100 |
100 |
100 |
|
Ethyl Cellulose |
50 |
40 |
40 |
30 |
|
Microcrystalline Cellulose |
40 |
40 |
40 |
40 |
|
Dibasic calcium phosphate |
30 |
40 |
30 |
50 |
|
PEG 400 |
20 |
10 |
20 |
20 |
|
Methyl paraben |
10 |
20 |
20 |
10 |
|
Weight per tablet |
550 mg |
550 mg |
550 mg |
550 mg |
Characterization of Polyherbal tablets
Particle size measurement
Size affects the average weight of tablet. The method used for determination of particle size is Sieving.
Loose and tapped bulk density
Apparent bulk density was determined by pouring a weighed quantity of blend into graduated cylinder and measuring the volume and weight (Aulton, 2002).
LBD = Weight of the powder / volume of the packing
It was determined by placing a graduated cylinder, containing a known mass of drug excipient blend. The cylinder was allowed to fall under its own weight on to a hard surface from the height of 10 cm at two second intervals (Banker and Anderson, 1987). The tapping was continued until no further change in volume was noted.
TBD = Weight of the powder/vol of the tapped packing
Compressibility index
The Compressibility index of the blends was determined by Carr’s compressibility index (Mohsin et al., 2010).
Compressibility index (%) = (TBD-LBD) x 100/TBD
Hausner ratio
It is the measurement of frictional resistance of the drug. The ideal range should be 1.2-1.5 (Hamid et al., 2006). It is determined by using the following formula:
Hausner ratio= TBD / LBD
Color and appearance
Colour and taste can be made more appealing by adding coloring and flavouring agents. The externally added coloring and flavoring agents should match with the colour and taste of medicament. The compressed tablets were examined for their color and appearance (Lakade and Bhalekar, 2008).
Weight variation test
With a tablet designed to contain a specific amount of drug in a specific amount of tablet formula, the weight of the tablet being made is routinely measured to ensure that a tablet contains the proper amount of drug. The average weight was determined by randomly selecting and weighing 20 tablets. Each tablet was also weighed individually. The deviation from the average weight in each case was Calculated and expressed as a percentage (Banker and Anderson, 1987).
Hardness and Friability test
Tablets require a certain amount of strength or hardness and resistance friability, to withstand mechanical shocks of handling in all processes. The hardness and friability were tested for the tablets by using calibrated hardness tester (Monsanto) and Roche friabilitor (4 minute at 25 rpm) tests respectively.
The friability of tablets for all formulations, were determined by Roche friabilator. The tablets was weighed and put into the plastic chamber and operated for 100 revolutions. Then, the tablets were dusted and reweighed. The percentage friability was calculated for each formulation and the values were given in table 4. The tablets passed the acceptable limit less than 1% (Lachman et al., 1987).
Disintegration test for tablets
The disintegration time of tablet for all formulations was determined by using the IP disintegration apparatus (Indian Pharmacopoeia, 1996). The 900 ml of 0.1N hydrochloric acid was the disintegration medium and the time to disintegrate completely was noted. A glass of plastic tube 80-100 mm long with an internal diameter of about 28 mm and external diameter 30-31 mm fitted at the lower end with a disc of rust proof wire gauge. Six tablets were placed in the tube, raise and lower the tube in such a manner that the complete up and down movement is repeated 28 to 32 per minute. The tablets are disintegrated when no particles remains above the gauge, which readily pass through mesh (10 mesh screen).
Thickness and diameter
The thicknesses and Diameter of the tablets were evaluated by using Vernier calipers.
In vitro drug release
The drug release testing of the formulated tablet was conducted by using USP paddle apparatus. It was carried out for 08 hrs with 900 ml 1.2 pH buffer acid solution maintained at 37±0.5◦C and agitated at 75 rpm. 5 ml of dissolution medium was withdrawn, filtered and diluted at regular intervals to determine the percentage drug release. The drug concentration was determined by calibration curve equation.
Antidiabetic effect of polyherbal formulation in Alloxan induced diabetes
Alloxan monohydrate (150mg/kg i.p) was dissolved in normal saline and injected i.p. after 18 hr fasting of animals to induce hyperglycemia. After 1hr of alloxan administration, the animals were fed on standard pellets and water ad libitum. The blood glucose level was monitored, samples collected under mild anesthesia and blood sugar level was measured with an autoanalyzer by using Accu Check advantage II glucose kit. Surviving rats after 72 hr, the rats having Blood Glucose Level (BGL) above 150mg/dl were selected for the investigation and divided into 5 groups (I-V) of 6 rats each. The vehicle and reference drug Metformin (250 mg/kg) were administered orally to animals of group I, II respectively and III group representing the control receiving only alloxan. Group No. IV and V received the polyherbal tablet formulation at a dose of 550 mg/kg, for 12 consecutive days and BGL was monitored after 1, 3, 6 and 12 days.
Determination of biochemical parameters
The effects of herbal formulation in normal and diabetic rats were assessed by measuring blood glucose level, Total cholesterol, Triglyceride levels and changes in body weight. Finally the one animal from each group was select to sacrifice by diethyl ether anesthesia, and liver and kidney tissues were separate for biochemical analysis.
Statistical analysis
The results of the study were subjected to analysis of variance followed by Dunnett’s t-test for multiple comparisons. Values with P<0.01 were considered to be significant.
Results and discussion
Extraction of different plant materials
The moderately coarse powder of the leaves of Carissa Carandus (150g) was subjected to extraction with petroleum ether and methanol by increasing order of polarity from non-polar to polar. The yields were found to be 2.8g (1.86% w/w of crude drug) of petroleum ether extract with semisolid mass of brown colour, 12.2g (8.13% w/w of crude drug) of methanolic extract with blackish brown colour semisolid mass, was observed. The moderately coarse powder of the leaves of Ocimum Sanctum (150g) was subjected to extraction with petroleum ether and methanol by increasing order of polarity from non-polar to polar.
Phytochemical investigation was performed for petroleum ether and methanolic extracts of the all proposed plants (Carissa carandus, Ocimum sanctum, Moringa oleifrea, Manilkara zapota). Petroleum ether extract of Carissa carandus showed the presence of Sterols while methanolic extract of Carissa carandus showed the presence of flavonoids and Tannins. Petroleum ether extract of Ocimum sanctum showed the presence of Proteins and amino acids while methanolic extract of Ocimum sanctum showed the presence of Alkaloids, flavonoids and saponins. Petroleum ether extract of Moringa oleifrea showed the presence of Sterols, Acidic compounds and Resins while methanolic extract of Moringa oleifrea showed the presence of Alkaloids flavonoids and Tannins. Petroleum ether extract of Manilkara zapota showed the presence of Sterols and amino acids while methanolic extract of Manilkara zapota showed the presence of Flavonoids, Phenolic compounds, Carbohydrates and Tannins.
Acute toxicity Study
Animals were observed at regular time intervals. In all the cases no death was observed within first 24 hours. Additional observations like behavioral changes in skin, fur, eyes, mucous membranes, respiratory, autonomous and central nervous systems and somatic motor activity and behavioral pattern. Attention was also given to observation of tremors and convulsions. The toxicity studies were carried out as per the OECD guidelines. The polyherbal formulation did not show any mortality or adverse event up to 2000 mg/kg. Hence, the study was carried out at the dose levels of 550 mg/kg.
Preparation and Characterization of polyherbal tablets
Polyherbal tablet formulations were prepared using the four herbal drug extracts. The various combinations of dried granules of powdered extracts of Carissa carandus, Ocimum sanctum, Moringa oleifrea, Manilkara zapota were prepared and characterized on the basis of pre-formulation studies including parameters like loose bulk density, tapped bulk density, compressibility index and hausner ratio etc. Preformulation study of the granules showed that all the evaluated parameters were within the acceptable limit.
The results of bulk density, Compressibility Index and Hausner’s ratio were indicated that the polyherbal powder mixture possess good flow properties and good packing ability (Figure 1 and 2). After a formulation by a direct compression method using automated punching machine, developed polyherbal tablets were subjected to measuring of post compression parameters like uniformity of weight, uniformity of content, hardness, friability, thickness, and disintegration time of the tablets. All the parameters of the test products are complied with the Pharmacopeial requirements.
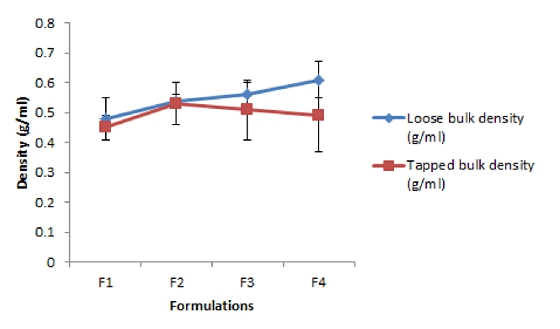
Figure 1. Loose bulk density and Tapped bulk density of granulated powder
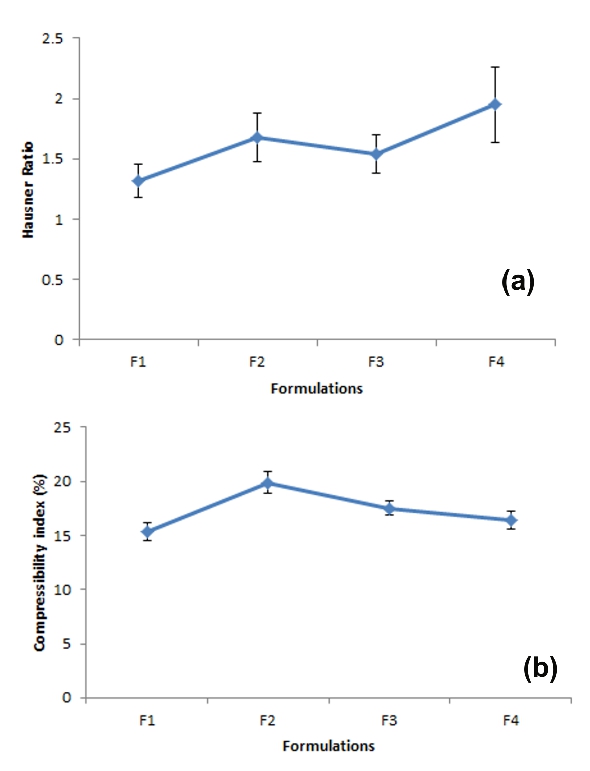
Figure 2. (a) Hausner Ratio and (b) Compressibility index (%) of powdered mixture
In the weight variation test the percentage weight variation in all the tablet formulations was within the pharmacopoeial limit. The variation was in range of 1.85±0.21 to 2.65±0.31% indicating the maximum percentage variation in F4 and minimum in F1. Thus all the formulations pass the test (Table 3).
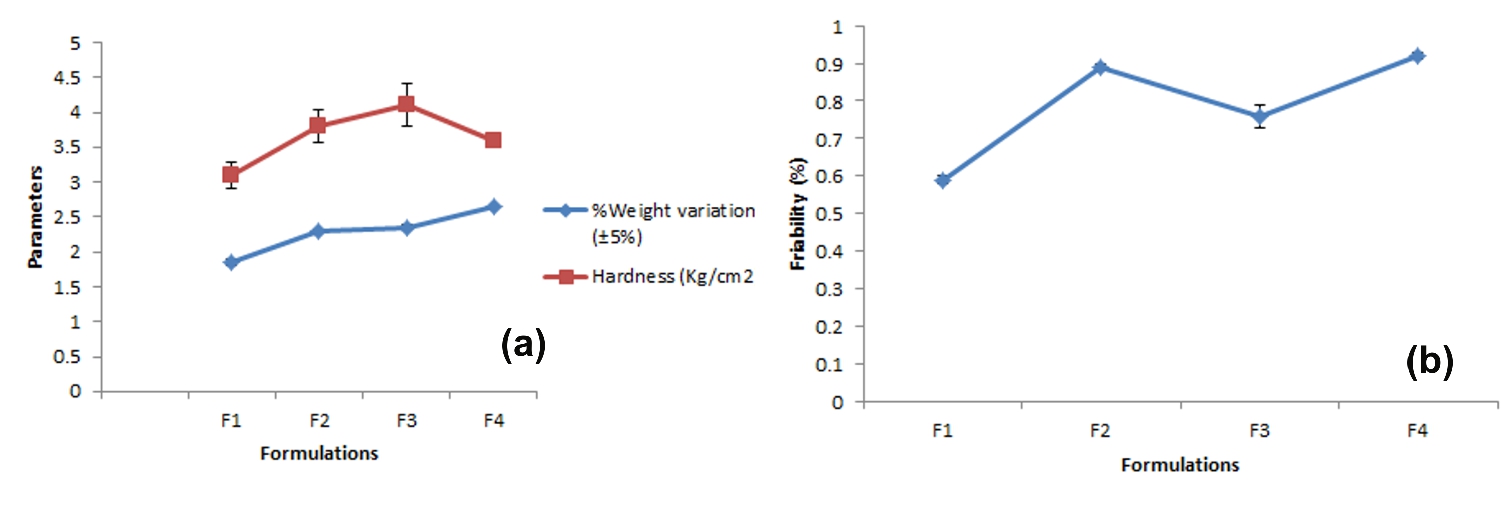
Figure 3. (a) Weight variation and Hardness (Kg/cm2) (b) Friability (%) results of prepared polyherbal tablet
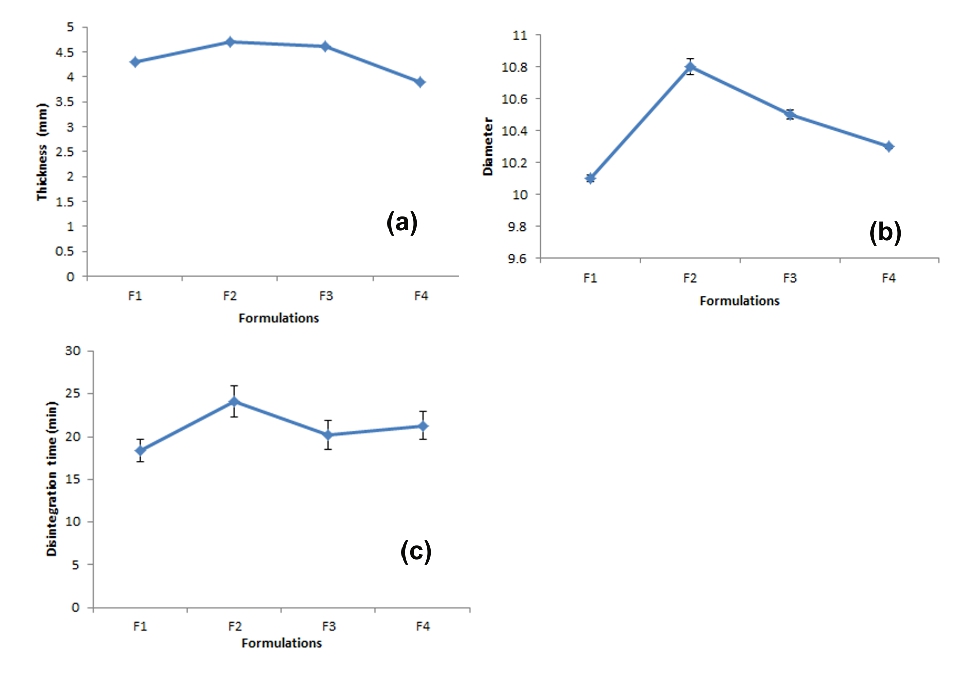
Figure 4. (a) Thickness, (b) diameter and (c) disintegration time of formulated tablets
The hardness of formulation was measured in kg/cm2 with the help of Monsanto tester. Amongst all the formulations prepared, F1 has been found to be the most acceptable one in terms of weight variation and in vitro disintegration time. This formulation showed appreciable hardness characteristics, which facilitated its fast disintegration. The friability of formulation indicated that the tablets were mechanically stable. As the average weight of tablets was 550 mg, the acceptable weight variation range is ±5%. Hence the entire formulated tablet passed the weight variation test. The disintegration time of formulation was not more than 25 Minutes (Figure 4).
The tablets require a certain amount of strength or hardness and resistance friability to withstand mechanical shocks of handling in all processes. The hardness of tablets was determined by Monsento hardness tester. The hardness of tablets was in range of 3.1 ±0.02 to 4.1± 0.03 indicating maximum in F3 and minimum in F1. The hardness of tablets was within the Pharmacopoeial limit. Thus all the tablets were pass the hardness test.
The friability of tablets was determined by Roche friabilator. The percentage friability was in range of 0.59 ± 0.01 to 0.92 ± 0.01 indicating maximum with F4 and minimum with F1. The range of percentage friability was within the pharmacopoeial limit. Thus all the formulations passed the friability test.
The disintegration time of tablets of all the formulations was determined using IP disintegration test apparatus. The time required to disintegrate the tablets was in range of 18.40±1.27 to 24.15±1.83 min. indicating maximum with F2 and minimum with F1. The range of disintegration time was within the pharmacopoeial limit. Thus all the formulation passed the disintegration test. The formulations F1-F4 were evaluated for in vitro drug release. The formulation F1 was found suitable which gave 100% drug release in 12hrs.
Antidiabetic effect of polyherbal formulation in Alloxan induced diabetes
The mechanism of action of alloxan includes the reactive oxygen species (ROS). The dismutation of free radicals causes great increase of calcium concentration inside the cell which leads to rapid destruction of the pancreatic beta cells. Dialuric acid is a reduction product of alloxan which helps to generate ROS in a cyclic reaction (Winterbourn and Munday, 1989; Winterbourn et al., 1989). The dose of alloxan needed to induce diabetes is less compared to STZ. Additionally, there is high possibility of death if it is slightly overdosed (Liang et al., 2010). Moreover, the tubular cell necrosis in kidney has been reported to be observed due to high dose of ALX induction. Since, the correct dosage regime for the induction of diabetes in animals is very important matter for both the STZ and ALZ.
Alloxan-induced diabetes in rats is characterized by particularly elevated blood glucose level, plasma triglycerides and total cholesterol levels. In contrast, Polyherbal tablet treated and metformin treated rats revealed decreased levels of triglycerides, cholesterol and also normal blood glucose level. These indicated beneficial effect of prepared polyherbal tablets in improving lipid profile in diabetic rats. Moreover, alloxan administration has also been reported to induce weight loss in rats (Chougale et al., 2007). The reduction of visceral fat in the diabetic rats and herbal tablet treated rats might be a reflection of the decreased body weight experienced by those rats.
Body weight changes in Alloxan induced model was lesser compared to STZ induce diabetic model. This indicates that in this model, partially prevented body weight loss as a result of its hypoglycemic effect of polyherbal tablets (Table 2).
The rats treated with polyherbal tablets and metformin showed significant (P > 0.01) reduction in blood glucose level measured after 21 days. Blood glucose level of both groups was found near to the normal control group of animals (Table 3).
Table 2. Effect of Poly herbal tablet on body weight of Alloxan induced diabetic rats
|
Groups (n = 6) |
Body weight (g) in different days |
|||
|
Day 0 |
Day 07 |
Day 14 |
Day 21 |
|
|
Control |
180.40±12.73 |
181.67±17.34 |
181.34±14.23 |
183.21±16.34 |
|
Diabetic Control |
195.28±17.28 |
201.23±18.26 |
205.93±18.06 |
205.43±18.73 |
|
Diabetic treated with standard |
176.53±13.67 |
179.64±14.61 |
178.67±14.31 |
178.07±14.81 |
|
Treated with Polyherbal tablet |
179.26±15.08 |
182.42±14.28 |
180.76±16.24 |
180.24±16.76 |
Data shown as mean±Standard deviation (SD). *P<0.01 compared to control group
Table 3. Effect of Poly herbal tablet on blood glucose of Alloxan induced diabetic rats
|
Groups (n = 6) |
Blood glucose level |
|
|
Day 0 |
Day 21 |
|
|
Control |
138.29±5.83 |
137.43±7.68 |
|
Diabetic Control |
341.28±25.19 |
338.25±28.19 |
|
Diabetic treated with standard |
324.58±24.89 |
142.39±12.36 |
|
Treated with Polyherbal tablet |
317.53±26.40 |
140.46±15.28 |
Values are mean ± SD; * P > 0.01
An increase in serum total cholesterol, and Triglyceride level were found in diabetic condition, but after treatment with polyherbal tablets and metformin it was significantly (P<0.01) reduced and restored near to the normal level (Table 4). In diabetic condition, total cholesterol and triglyceride level was 127.64±11.67 and 97.36±7.45, respectively. After treatment with polyherbal tablets, it was significantly reduced to 130.60±11.38 and 102.87±8.64, respectively. These lowered values were comparable to the standard treated group.
After treatment with polyherbal tablets and standard metformin, there was a significant improvement was observed in antioxidant markers level (Table 5). The polyherbal formulation treated animals inhibited the hyperglycemia induced by Alloxan, which may be due to the free radical scavenging properties of the individual herbs present in the herbal tablets.
Table 4. Effect of Poly herbal tablet on Lipid profile of Alloxan induced diabetic rats
|
Groups (n = 6) |
Lipid parameters on Day 21 |
|
|
Total Cholesterol (TC) (mg/dL) |
Triglyceride (TG) (mg/dL) |
|
|
Control |
127.64±11.67 |
97.36±7.45 |
|
Diabetic Control |
216.82±19.34 |
217.64±19.73 |
|
Diabetic treated with standard |
134.23±11.48 |
112.37±9.34 |
|
Treated with Polyherbal tablet |
130.60±11.38 |
102.87±8.64 |
Values are mean ± SD; *P > 0.01
Table 5. Effect of tablet formulations and reference drug on enzymes and non enzymatic level of tissues in Alloxan induced diabetes model in rats
|
Groups |
Enzymatic and non-enzymatic assay |
||
|
SOD(μg/50 mg tissue) |
CAT(μmol/50 mg tissue) |
GSH(μmol/50 mg tissue) |
|
|
Control |
42.07±3.80 |
37.19±2.49 |
54.27±4.16 |
|
Diabetic Control |
22.36±1.97 |
21.84±1.86 |
24.67±2.85 |
|
Diabetic treated with standard |
40.52±3.26 |
35.62±±2.46 |
53.69±3.47 |
|
Treated with Polyherbal tablet |
41.28±3.60 |
34.24±2.95 |
50.61±4.25 |
n = 6 albino rats per group, value represents Mean ± S.D. *P< 0.01, when compared each treated group with control group
Conclusion
Observation of the present study was confirmed that prepared formulation was safe, and effective for antidiabetic activity. It was confirmed that prepared formulation was able to restore lipid profile, blood glucose level and antioxidants level to the nearly of normal. These positive effects may attribute to the presence of various flavonoid constituents present in methanol extracts of all plants. Additionally this formulation also showed antioxidant effect that may another possible mechanism for antidiabetic effect of prepared formulation.
Conflicts of interest: Not declared.
References
Aulton ME, Pharmaceutics-The science of dosage form design. ed 2, London: ELBS/ Churchill Livingstone, 2002.
Banker GS, Anderson NR. Tablets In: Lachman N, Liberman HA, Kanig JL, editors. The theory and practice of industrial pharmacy, ed 3, Bombay, Varghese Publication House, 1987.
Chanda SV, Nagani KV. 2010. Antioxidant capacity of Solanum Virginianum L.leaves extracts evaluated by four in vitro method. Nature and Science, 10:260-266.
Chopra RN, Nayer SL, Chopra IC. Glossary of Indian Medicinal Plants. New Delhi, India: Council of Scientific and Industrial Research 1956.
Fowler. LM, ME Williams. 1995. Use of unproven therapies by people with alzheimers disease. Journal of the American Geriatrics Society 43:747-750.
Ghosh S, Rai P. Investigation on aerial parts of Canscora decussate. Journal of Pharmacy Sciences, 28:144-147.
Indian Pharmacopeia. 1996. Ghaziaad, India: The Indian Pharmacopoeia Commission, ISBN 81-903436-6-1.
Kokate CK, Purohit AP, Gokhale SB. 2002. Pharmacognosy 13th edition, Nirali Prakashan, Pune.
Lachman L, Lieberman HA, Kanig JL. The Theory and Practice of Industrial Pharmacy. 3rd ed. Varghese Publishing House; 1987:67.
Lakade SH, Bhalekar MR. 2008. Formulation and evaluation of sustained release matrix tablets of anti-anginal drug, influence of combination of hydrophobic and hydrophilic matrix former. Research Journal of Pharmacy and Technology, 1(4); 410-413.
Maiti B, Nagori BP, Singh R. 2011. Recent trends in herbal medicine: review. International Journal of Drug Research and Technology, 1(1):17-25.
Mohsin AA, Nimbalakr NE, Sanaullah S, Aejaz A. 2010. Formulation and evaluation of mouth dissolving tablets of amitryptyline hydrochloride by direct compression technique. International Journal of Pharmaceutical Science, 2(1):204-210.
Muller K. 2003. Characterization & treatment of large sensory fiber Peripheral neuropathy in mice BS, Emory University 2003.
Naim Z, Khan M, Nizami S. 1985. Isolation of a new triterpenic alcohol from of Carissa carandas. Pakistan journal of Scientific and industrial Research, 28 (6):378-381.
Paech K, Tracey MV. Modern Methods of plant Analysis" Vol. 4, Springer Verlog, Berlin, 1955.
Rastogi, RC, Rastogi RP, Dhar ML. 1967. Studies on Carissa carandas Linn. II. Polar glycosides. Indian Journal of Chemistry, 5(5):215-221.
Siddiqui S, Ghani U, Ali S, Usmani S, Begum S. 2003. Triterpenoidal Constituents of the leaves of Carissa carandas. Natural Products Research 31(11):753-755.
Sim SK. Medicinal Plant Glycosides 2nd edition, University of Toronto Press, 1968.