

Anita S. Wagh*, Santosh R. Butle
School of Pharmacy, S.R.T.M. University, Nanded, Maharashtra, India
*Address for Corresponding Author
Anita S. Wagh
Department of Pharmacognosy,
School of Pharmacy, S. R.T.M University, Nanded, Maharashtra, India.
Abstract
Objective: The aim of the present work was the phytochemical investigation and in vitro evaluation of different extracts of Spathodea campanulata P. Beauv. leaves for anticancer activity. Method: Chloroform, Ethyl acetate, Methanol and Aqueous extracts of Spathodea campanulata P. Beauv. were tested for their anticancer activity on three cancer cell lines i.e. MCF-7 (Breast), HL-60 (Leukemia), and HT-29 (Colon) at various concentrations using Sulphorhodamine B assay. Results: Phytochemical analysis of the plant extracts proves the presence of flavonoid, alkaloids, saponins, tannins and triterpenes. Chloroform, Ethyl acetate and Methanolic extracts showed dose dependent anticancer effect on leukemia (HL-60) cancer cells but these extracts were inactive against breast (MCF-7) and colon (HT-29) cancer cell lines. The activities of these different extracts were compared with the standard drug Adriamycin. Aqueous extract was inactive against all three cell lines. Conclusion: The results of in vitro anticancer activity showed that Spathodea campanulata P. Beauv. plant can be used in the treatment of leukemia.
Keywords: Spathodea campanulata P. Beauv., phytochemicals, anticancer activity, Sulphorhodamine B assay
Introduction
Cancer is a worldwide public health problem that is likely to affect almost every person on the planet, with the estimation that one in three people will develop cancer at some point during their lifetime. It is second only to heart disease as a leading cause of death in developed nations, and is among the three leading causes of death for adults in developing countries (Jackson, 2000). Cancer incidence increased from 12.7 million cases in 2008 to 14.1 million cases in 2012, with the number of died people up from 7.6 million people in 2008 to 8.2 million people in 2012. It is estimated that around 26 million people will suffer from cancer and 17 million people will die because of cancer in 2030 (IARC, 2012). Cancer begins with mutations in DNA, which instructs the cells how to grow and divide. Normal cells have the ability to repair most of the mutations in their DNA, but the mutation which is not repaired and causing the cells to grow becomes cancerous (Krishnamurthi, 2000).
The major causes of cancer are smoking, dietary imbalances, hormones and chronic infections leading to chronic inflammation (Ames et al., 1995). Although there are many therapeutic strategies, including chemotherapy to treat cancer, high systemic toxicity and drug resistance limit the successful outcomes in most cases. Accordingly, several new strategies are being developed to control and treat cancer (Gali et al, 2011).
Plants have been used for treating various diseases of human beings and animals since time immemorial. They maintain the health and vitality of individuals, and also cure diseases, including cancer without causing toxicity. According to the estimates of the WHO, more than 80% of people in developing countries depend on traditional medicine for their primary health needs (Murthy et al., 2007). Plants constituents are not only important sources of drugs used to treat cancers, but also are used as models for the development of novel anticancer chemotherapeutic drugs (Cragg et al., 2005). 60% of the commercially available anticancer drugs are from natural sources.
The National Cancer Institute collected about 35,000 plant samples from 20 different countries, and has screened round 114,000 extracts for anticancer activity. Treatment by herbal medicines may have some advantages over treatment by single purified chemicals (Vickers, 2002); as herbal medicine are the mixtures of more therapeutic or preventive components, and so might have more activity than single products alone. The antioxidant and anti-tumor effects of extracts from various herbs and medicinal plants have been proved experimentally and clinically. Several in vitro or in vivo studies have proved the anticancer potential of the extracts from several medicinal plants (Rajdeep, 2011).
Many natural plants extracts and phytoconstituents have been reported to induce apoptosis in cancer cell lines. From the screening process, taxol from Taxas brevifolia was discovered and developed into one of the most successful plant based anti cancer drug. Other anticancer drugs originated from plants include vinblastine and vincristine derived from Catharanthus roseus and etoposide from Podophyllum peltatum. Identification of new cytotoxic compounds led the development of anticancer therapeutics for several decades. Compounds of natural origin have often provided new leads in the novelty of structures with anticancer activity (Md. OR et al., 2012).
Spathodea campanulata P. Beauv. tree is native of Africa and it is widely grown in tropical and subtropical regions outside Africa. This plant is also commonly found in India as an ornamental plant. Spathodea campanulata, ,a perennial tree belonging to family Bignoniaceae commonly known as African Tulip tree, fire ball, fountain tree, flame of the forest and Pichakari. Plant parts such as flowers, leaves stem, bark and roots have been reported for possessing anti-inflammatory, analgesic, cytotoxic, anti-solar anti anticonvulsant activity. Phytochemical screening shows the presence of alkaloids, tannins, flavonoids, glycosides and sterols (Wagh and Butle, 2018).
Based on the literature survey, it is evident that no work has been carried out on the evaluation of anticancer property of leaves extracts on the selected cell lines. Hence in this present study, anticancer activity of Spathodea campanulata P. Beauv. extracts were assessed by investigating the inhibition of cell growth of MCF-7 (Breast), HL-60 (Leukemia), and HT-29 (Colon) at various concentrations using Sulphorhodamine B assay.
Materials and methods
Plant materials and reagents
Leaves of Spathodea campanulata P. Beauv. were collected from Nanded District, Maharashtra and authenticated from Botanical Survey of India, Pune. (Plant Authentication No. BSI/WRC/100-1/ Tech./2017). All the reagents and chemicals were purchased from Merck chemicals Ltd.
Prepration of stem extract
The leaves were washed with distilled water, shade dried and powdered. Powdered drug material was subjected to successive solvent extraction using Chloroform, Ethyl acetate, Methanol and Water respectively. The extracts were filtered, evaporated and stored at 400C.
Phytochemical screening
Phytochemical screening was done to determine presence or absence of secondary metabolites such as tannins, alkaloids, flavonoids, saponins, sterols and phenolic compounds. This was done according to established procedure (Fawole et al., 2009; Ndam et al., 2014; Ngbede et al., 2008; Khandelwal, 2000).
Anticancer Activity
The cell lines were grown in RPMI 1640 medium containing 10% fetal bovine serum and 2 mM L-glutamine. For present screening experiment, cells were inoculated into 96 well microtiter plates in 100 µL at plating densities as shown in the study details above, depending on the doubling time of individual cell lines. After cell inoculation, the microtiter plates were incubated at 37°C, 5% CO2, 95% air and 100% relative humidity for 24 h prior to addition of experimental drugs.
Different extracts were initially solubilized in dimethyl sulfoxide at 100mg/ml and diluted to 1mg/ml using water and stored frozen prior to use. At the time of drug addition, an aliquote of frozen concentrate (1mg/ml) was thawed and diluted to 100μg/ml, 200μg/ml, 400μg/ml and 800 μg/ml with complete medium containing test article. Aliquots of 10µl of these different drug dilutions were added to the appropriate microtiter wells already containing 90µl of medium, resulting in the required final drug concentrations i.e.10μg/ml, 20μg/ml, 40μg/ml, 80μg/ml.
After compound addition, plates were incubated at standard conditions for 48 hours and assay was terminated by the addition of cold TCA. Cells were fixed in situ by the gentle addition of 50µl of cold 30 % (w/v) TCA (final concentration, 10 % TCA) and incubated for 60 minutes at 4°C. The supernatant was discarded; the plates were washed five times with tap water and air dried. Sulforhodamine B (SRB) solution (50µl) at 0.4 % (w/v) in 1 % acetic acid was added to each of the wells, and plates were incubated for 20 minutes at room temperature. After staining, unbound dye was recovered and the residual dye was removed by washing five times with 1 % acetic acid. The plates were air dried. Bound stain was subsequently eluted with 10 mM trizma base, and the absorbance was read on a plate reader at a wavelength of 540 nm with 690 nm reference wavelength.
Percent growth was calculated on a plate-by-plate basis for test wells relative to control wells. Percent Growth was expressed as the ratio of average absorbance of the test well to the average absorbance of the control wells * 100 (Vanicha et al., 2006; Skehn et al., 1990).
Using the six absorbance measurements [time zero (Tz), control growth (C), and test growth in the presence of drug at the four concentration levels (Ti)], the percentage growth was calculated at each of the drug concentration levels. Percentage growth inhibition was calculated as:
[Ti/C] x 100 % (Ref)
Results and discussion
The phytochemicals present in the leaves of Spathodea campanulata P. Beauv. was found to be alkaloids, saponins, tannins, flavonoid, steroids and phenolic compounds. The result of preliminary phytochemical investigation is presented in table 1.
Table 1. Phytochemical analysis of different extracts of Spathodea campanulata P. Beauv. leaves
|
Phytochemicals |
Different extracts of Spathodea campanulata P. Beauv. leaves |
|||
|
Chloroform |
Ethyl Acetate |
Methanol |
Aqueous |
|
|
Alkaloids (Dragendorff’s Test) |
+ |
+ |
+ |
- |
|
Phenols (Lead acetate solution) |
+ |
+ |
+ |
- |
|
Tannins (5% FeCl3 solution) |
+ |
+ |
+ |
+ |
|
Flavonoids (Shinoda Test) |
+ |
+ |
+ |
+ |
|
Saponins (Foam Test) |
+ |
+ |
+ |
+ |
|
Steroids (Salkowski Test) |
+ |
+ |
- |
- |
|
Carbohydrates (Molish’s Test) |
- |
- |
- |
+ |
|
Proteins (Millons Test) |
- |
- |
- |
+ |
Where, + = Present, - = Absent
In the present study chloroform, ethyl acetate, methanol and aqueous extracts of leaves Spathodea campanulata P. Beauv. were evaluated for anticancer activity on three cancer cell lines i.e. MCF-7 (Breast), HL-60 (Leukemia), and HT-29 (Colon) at various concentrations using SRB assay. The activities of the different extracts were compared with the standard drug Adriamycin (Doxorubicin). The result was found that chloroform, ethyl acetate and methanol extracts were active on Human Leukemia Cell Line HL-60 and GI50 value for chloroform, ethyl acetate, methanol extracts was found to be <10μg/ml except aqueous extract >80μg/ml. These extracts were inactive on Human Breast cancer cell line MCF-7 and Human Colon cancer cell line HT-29 and GI50 value of chloroform, ethyl acetate, methanol and aqueous extracts for MCF-7 and HT-29 cell line was found to be >80μg/ml. Aqueous extract was inactive against all the three selected cell lines. Doxorubicin served as a positive control (Tables 2, 3 and 4). The results of in vitro cancer activity showed that Spathodea campanulata plant can be used in the treatment of leukemia.
Table 2. In vitro anticancer activity on MCF -7 Cell lines
|
Extracts |
% Growth Control at different concentration (μg/ml) |
GI50 |
|||
|
10 |
20 |
40 |
80 |
||
|
Chloroform extract (SCE) |
97.6 |
103.8 |
92.1 |
71.2 |
>80 |
|
Ethyl acetate extract (SEE) |
82.4 |
86.5 |
58.7 |
36.4 |
59.6 |
|
Methanolic extract (SME) |
93.0 |
101.5 |
84.0 |
68.8 |
>80 |
|
Aqueous extract (SAE) |
105.6 |
118.3 |
117.1 |
111.4 |
>80 |
|
ADR |
-64.3 |
-64.0 |
-50.1 |
-45.9 |
<10 |
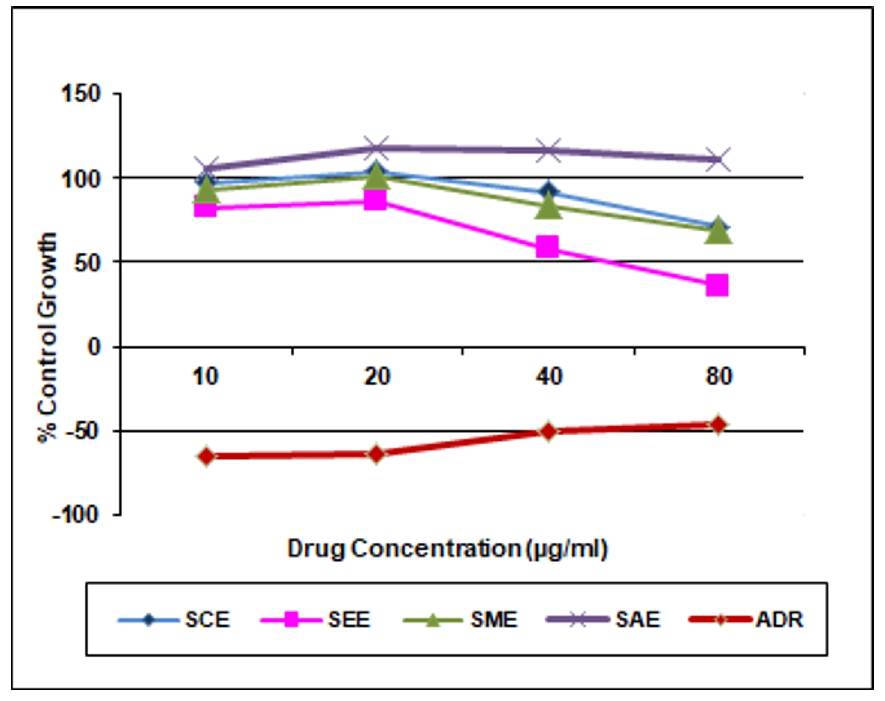 Figure 1. Growth curve: Human breast cancer cell line
Figure 1. Growth curve: Human breast cancer cell line
Table 3. In vitro anticancer activity on HT-29 Cell lines
|
Extracts |
% Growth Control at different concentration (μg/ml) |
GI50 |
|||
|
10 |
20 |
40 |
80 |
||
|
Chloroform extract (SCE) |
103.7 |
109.2 |
106.5 |
106.3 |
>80 |
|
Ethyl acetate extract (SEE) |
102.4 |
105.9 |
98.5 |
85.5 |
>80 |
|
Methanolic extract (SME) |
107.2 |
111.4 |
117.8 |
118.6 |
>80 |
|
Aqueous extract (SAE) |
107.5 |
109.5 |
118.1 |
117.4 |
>80 |
|
ADR |
-16.1 |
-22.5 |
-21.3 |
-22.4 |
<10 |
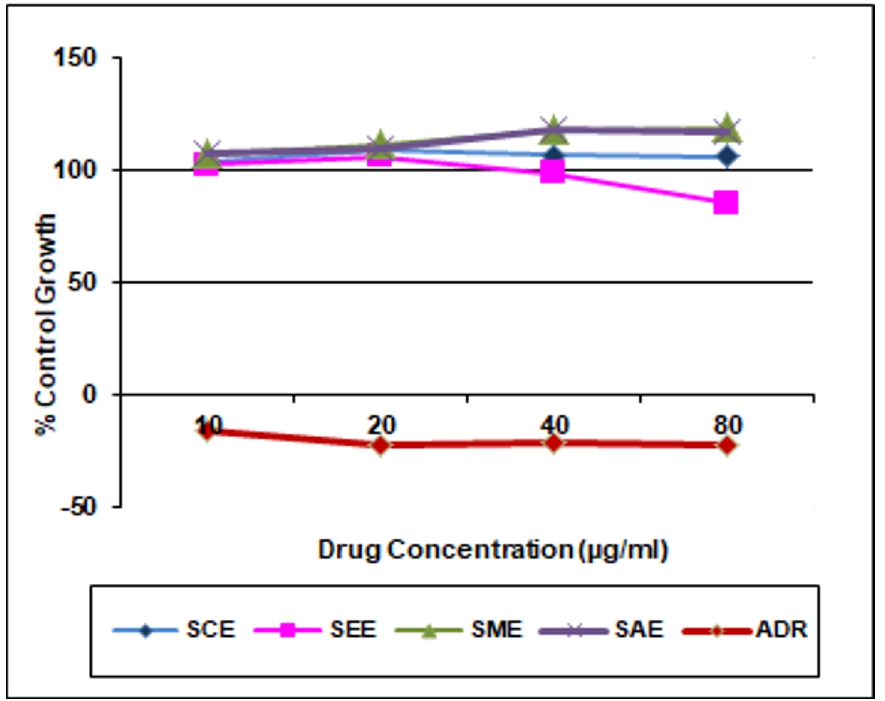
Figure 2. Growth curve: Human colon cancer cell line
Table 4. In vitro anticancer activity on HL-60 Cell line
|
Extracts |
% Growth Control at different concentration (μg/ml) |
GI50 |
|||
|
10 |
20 |
40 |
80 |
||
|
Chloroform extract (SCE) |
-5.8 |
-3.5 |
-10.7 |
-18.6 |
<10 |
|
Ethyl acetate extract (SEE) |
6.0 |
27.6 |
40.3 |
6.4 |
<10 |
|
Methanolic extract (SME) |
20.5 |
37.4 |
29.5 |
45.0 |
<10 |
|
Aqueous extract (SAE) |
133.0 |
91.3 |
99.3 |
72.6 |
>80 |
|
ADR |
-64.3 |
-68.7 |
-72.6 |
-73.5 |
<10 |
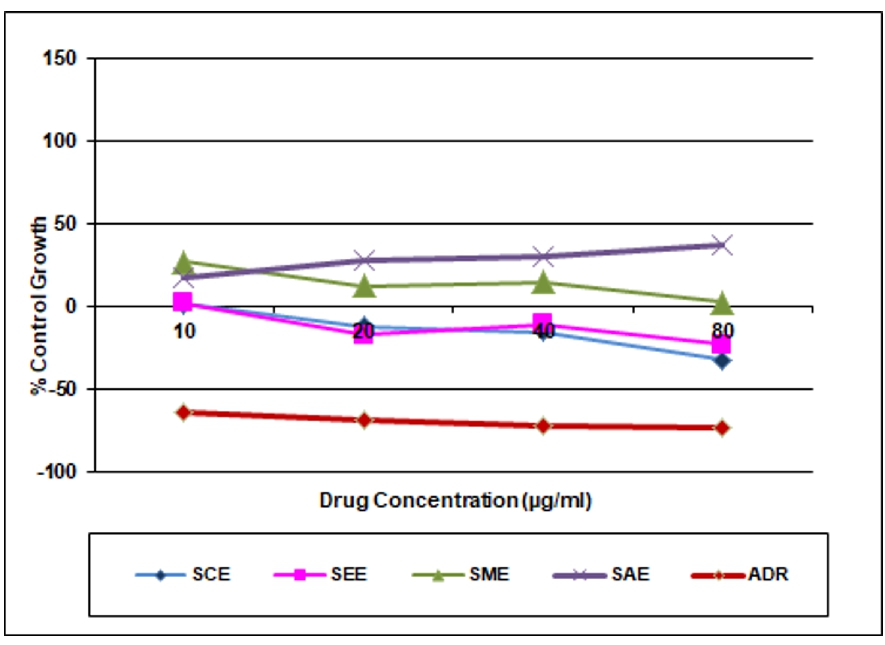
Figure 3. Growth curve: Human leukemia cancer cell line
Conclusion
From the present study, it was concluded that the Spathodea campanulata P. Beauv plant was active against Human leukemia cancer Cell lines (HL-60) which may be due to synergistic effect of various phytochemicals present in the extracts. Isolation of phytoconstituents responsible for anticancer activity is going in the laboratory. This research will give an idea for the development of anticancer drugs.
Acknowledgement
Authors are thankful to the Director, School of Pharmacy, S R T M University, Nanded for providing necessary facilities.
Conflict of interest statement
We declare that we have no conflict of interest.
References
Ames BN, Gold LS, Willett WC. 1995. The causes and prevention of cancer. Proceeding of National Academy of Sciences, 92:5258-5265.
Cragg GM, Newman DJ. 2005. Plants as a source of anticancer agents. Journal of Ethnopharmacology, 100:72-79.
Fawole OA, Ndhlala AR, Amoo SO, Finnie JF, Van-Staden J. 2009. Anti-inflammatory and phytochemical properties of twelve medicinal plants used for treating gastrointestinal ailments in South Africa. Journal of Ethnopharmacology, 123:237-243.
Gali K, Ramakrishnan G, Kothai R, Jaykar B. 2011. In-vitro Anti-Cancer activity of Methanolic extract of leaves of Argemone mexicana Linn. International Journal of PharmTech Research, 3:1329-133.
IARC. 2012. World Cancer Report. Geneva: International Agency for Research on Cancer (IARC).
Jackson BG. 2000. Mechanism based target identification and drug discovery in cancer research. Science 287:1969.
Khandelwal KR, Practical Pharmacognosy. 2005. Nirali Prakashan, IIIrd Edition, pp. 149-53.
Krishnamurthi K. 2000. Screening of natural products for anticancer and antidiabetic properties. Health Administrator. XX (1&2):69.
Md OR, Syed MT, Afrina B, Md KA, Haque A, Md SA. 2012. Evaluation of Antitumor Activity of Leea indica (Burm.f.) Merr. extract against Ehrlich Ascites Carcinoma (EAC) Bearing Mice. American Journal of Biomedical Sciences, 4(2):143-152.
Murthy NS, Agarwal UK, Chaudhry K, Saxena S. 2007. A study on time trends in incidence of breast cancer –Indian scenario. European Journal of Cancer Care, 16:185-6.
Ndam LM, Mih AM, Fongod AGN, Tening AS, Tonjock RK, Enang JE, Y. Fujii. 2014. Phytochemical screening of the bioactive compounds in twenty (20) Cameroonian medicinal plants. International Journal of Current Microbiology and Applied Sciences, 3(12):768-778.
Ngbede J, Yakubu RA, Nyam DA. 2008. Phytochemical screening for active compounds in Canarium schweinfurthii (atile) leaves from Jos North, Plateau State, Nigeria. Research Journal of Biological Sciences, 3(9):1078-1078.
Rajdeep K, Karan K, Harpreet K. 2011. Plants as a source of anticancer agents. Journal of Natural Products and Plant Resources, 1:19-124.
Skehn P, Storeng R, Scudiero A, Monks J, McMohan D, Vistica D, Jonathan TW, Bokesch H, Kenney S, Boyd MR 1990. New colorimetric cytotoxicity assay for anticancer drug screening. Journal of the National Cancer Institute, 82-1107.
Vanicha V, Kanyawim K. 2006. Sulforhodamine B colorimetric assay for cytotoxicity screening Nature Protocols: 1112 –16.
Vickers A. 2006. Botanical medicines for the treatment of cancer: rationale, Overview of current data, and methodological considerations for phase I and II trials. Cancer Invest 20:1069-1079.
Wagh AS, Butle SR. 2018. Plant Profile, Phytochemistry and Pharmacology of Spathodea Campanulata P. Beauvais (African Tulip Tree): A Review. International Journal of Pharmacy & Pharmaceutical Sciences, 10:1-6.