

Ayyadurai Brabakaran, Nallamuthu Thangaraju*
Algology Laboratory, CAS in Botany, University of Madras, Guindy Campus, Chennai-600 025, India
*Corresponding author
Dr. N. Thangaraju,
Assistant Professor,
Centre for Advanced studies in Botany, University of Madras, Guindy Campus, Chennai-600025 India
Abstracts
Objective: In the present study, methanolic extract of red seaweeds, Spyridia filamentosa, Grateloupia lithophila and Hypnea musciformis against α- amylase, glucosidase enzyme inhibitory activity were carried out. Materials and methods: Phytochemical constituents of methanolic extract were qualitatively determined. Antidiabetic activity was evaluated by inhibitory potential of methanolic extract against α-amylase and α- glucosidase by spectrophotometric assays. Results: The crude methanolic extract shows stronger inhibitor effect to α- amylase and α- glucosidase of S. filamentosa (IC50 = 58.02 and 66.06 µg/ml), G. lithophila (IC50 = 53.01 and 58.02 µg/ml) and H. musciformis (IC50 = 48.01 and 51.02 µg/ml), respectively. Phytochemical screening of red seaweed extracts revealed the presence of phenols, protein, lipids and carbohydrates. Conclusion: The result suggest that crude methanolic extract of
S. filamentosa have anti- diabetic potential through inhibition of α- amylase and α- glucosidase.
Keywords: Anti diabetic, Seaweeds, α- amylase, enzymes
Introduction
Hydrolysis of dietary starch is the major source of glucose in the blood, with α-amylase and α-glucosidase being the key enzymes involved in starch breakdown and an intestinal absorption. Enzymes can significantly decrease the postprandial increase of blood glucose level after a mixed carbohydrate diet and therefore can be an important strategy in the management of hyperglycaemia linked to type II diabetes (Kwon et al., 2008). A calcium metalloenzyme, α-amylase catalyses the cleavage of α-D-(1-4) glycosidic linkages of starch, amylose, amylopectin, glycogen and various maltodextrins in to shorter oligosaccharides (De Sales et al.,2012). Consequently, inhibitors of these hydrolytic enzymes suppress the influx of glucose from the intestinal tract to blood vessels resulting in a decrease in postprandial hyperglycaemia.
The uses of seaweeds were active components for the prevention and/or treatment of chronic diseases are based on the traditional medicine of various ethnic societies and on epidemiological data (Ribeiro et al.,2000). Marine algae are one of the richest sources of structurally diverse natural products. In recent years an increasing number of novel compounds have been isolated from marine algae against several biological activities (Miller et al.,1959). In the present study, to evaluate the phytochemical screening and enzymes activity of red seaweeds (S. filamentosa, G. lithophila, H. musciformis) through in vitro.
Materials and Methods
Collection of Seaweeds and preparation of extracts
Seaweeds (S. filamentosa, G. lithophila and H. musciformis) (Figure 1) were collected from Rameshwaram coast of Tamil Nadu, India. Seaweeds specimen were identified by the authentic algal taxonomist. The seaweeds were washed with sea/tap waters and shades dried at 27 °C for within week and were coarsely powdered in a powdering machine. The powdered biomass were prepared using methanol, as solvent using a soxhlet apparatus. The crude extracts were filtered and then concentrated in a rotary evaporator at a temperature 40 ͦC and further used.
 Figure 1. Photographs showing red seaweeds (A) Spyridia filamentosa, (B) Grateloupia lithophila and (C) Hypnea musciformis
Figure 1. Photographs showing red seaweeds (A) Spyridia filamentosa, (B) Grateloupia lithophila and (C) Hypnea musciformis
Phytochemical analysis of methanloic extract
Preliminary phytochemical screening of methanolic extracts of S. filamentosa, G. lithophila and H. musciformis were carried out according to standard procedure Harborne (1998).
α- amylase inhibition assay
The α-amylase was premixed with the methanolic extract at 50 to 250 µg/mL and starch as a substrate was added as a 0.5% starch solution to start the reaction. This was carried out at 37 oC for 5 min and terminated by addition of 2 mL of DNS (3,5-dinitrosalicylic acid) reagent. The reaction mixture was heated for 15 min at 100 oC and diluted with 10 mL of distilled water in an ice bath . α-Amylase activity was determined by measuring spectrum at 540 nm. The IC50 value was defined as the concentration of α-amylase inhibitor to inhibit 50% of its activity under the assay conditions (Miller et al.,1959).
α-glucosidase inhibition assay
The inhibition of α-glucosidase activity was determined standard method 3 mM p-nitrophenyl α-D-glucopyranoside as a substrate was added to the reaction mixture. The reaction was incubated at 37 oC for 30 min and stopped by adding 2 mL of Na2CO3. The α-glucosidase activity was determined by measuring the p-nitrophenol release from pNPG at 400 nm. The IC50 value was defined as the concentration of α-glucosidase inhibitor to inhibit 50% of its activity under the assay conditions (Miller et al.,1959).

Data analysis
The α-amylase and α- glucosidase inhibitory activity were performed in triplicate and mean and standard deviation values were calculated.
Results
Phytochemical constituents of the methanolic extracts of S. filamentosa, G. lithophila and H. musciformis shows in table 1. The most an abundant compounds in S. filamentosa, extract were phenols, flavanoids, protein, lipids and carbohydrates (Table 1). Table 1 showed that G. lithophila and H. musciformis methanolic extracts flavanoids, saponins were not present.
Table. 1 Phytochemical constitutes of methanolic extracts of red seaweeds
|
Constitutes |
Methanolic extracts |
||
|
Spyridia filamentosa |
Grateloupia lithophila |
Hypnea musciformis |
|
|
Phenols |
+ |
+ |
+ |
|
Flavanoids |
+ |
_ |
_ |
|
Saponins |
+ |
_ |
_ |
|
Proteins |
+ |
+ |
+ |
|
Lipids |
+ |
+ |
+ |
|
Carbohydrates |
+ |
+ |
+ |
α-amylase inhibition activity
The α-amylase inhibition activity of methanolic extracts at various concentrations 50, 100, 150, 200, 250 µg/ml. The inhibition S, filamentosa (28%, 33%, 39%, 44% and 58%, ),G. lithophila (25%, 28%, 33%, 41% and 53%) and H. musciformis (22%, 25%, 29%, 32% and 48%) at 50, 100, 150, 200, 250 µg/ml, respectively (Figure 2). The standard acarbose 38%, 47%, 59%, 65% and 76% of α-glucosidase inhibition, respectively (Figure 2).
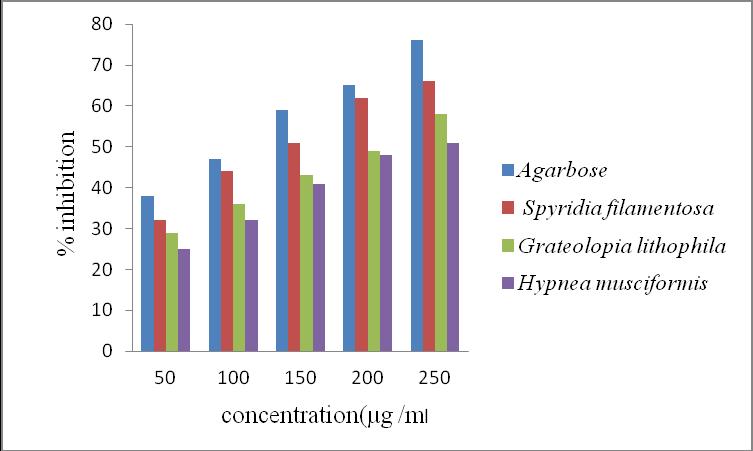 Figure 2. α- amylase inhibition activity of methanolic extracts of selected red seaweeds
Figure 2. α- amylase inhibition activity of methanolic extracts of selected red seaweeds
α-glucosidase inhibition activity
Figure 3 showed that α-glucosidase inhibition activity of methanolic extracts at various concentrations 50, 100, 150, 200, 250 µg/ml. The maximum inhibition S, filamentosa (66.6%), G. lithophila (58.4%) and H. musciformis (51.1%) at 250 µg/ml. The minimum inhibition S, filamentosa (32%), G. lithophila (29%) and H. musciformis (25%) at 50 µg/ml, 24hrs (Figure 3). The various range 50 to 250 µg/ml from standard acarbose showed 38% and 76% of α-glucosidase inhibition respectively (Figure 3).
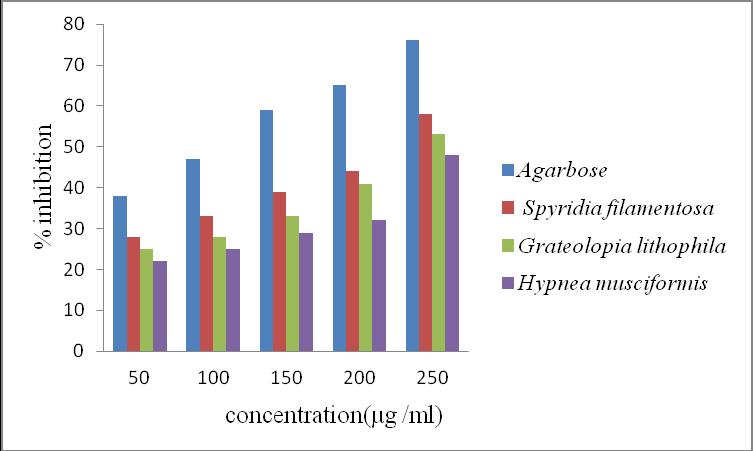 Figure 3. α-glucosidase inhibition activity of methanolic extracts
Figure 3. α-glucosidase inhibition activity of methanolic extracts
Discussion
Seaweeds products presents in vitamins, minerals, trace elements, proteins and bioactive substances (Dharmesh et al., 2014). Several polysaccharides are recovered from seaweeds, with the most important of them being agar, alginic acid, laminarine, fucoidin, etc., (Saritha et al., 2013). In the present investigation, phytochemical screening of red seaweeds (S. filamentosa,
G. lithophila and H. musciformis) showed that presence of phenols, flavanoids. proteins, lipids, and carbohydrates, in all extracts were tested. Flavanoids as absent in the methanolic extracts of G. lithophila and H. musciformis.
Alpha amylase inhibition of seaweeds methanolic extracts of Chlorodesmis sp., (green) (Unnikrishnan et al., 2015) and acetone extracts of Spatoglossum schroederi (brown) by Teixeira et al (2007). Lakshmana Senthil (2013) studied on α-amylase inhibitory activity using methanol, acetone and ethyl acetate extracts of Gracilaria edulis (57±0.9, 53±0.1 and 60±1.2), respectively. Ethanol extracts of Ascophyllum nodosum have been strongest a-amylase inhibitory (IC50 = 44.7 µg/ml) (Lordan et al., 2013). In the present study, we noted the α-amylase inhibitory activity than acarbose, methanolic extracts of red seaweeds (S. filamentosa, G. lithophila and
H. musciformis).
Sun and Chen (2012) evaluated the inhibitory effects of green algae Chollrella Pyrenoidasa sp., against α-amylase and α- glucosidase inhibitory activity and fulfilled an careful as a potential functional food for diabetic patients. The cold water and ethanol extracts of Fucus vesiculosus were found to be potent inhibitors of a-glucosidase IC50 values of 0.32 and 0.49 µg/ml (Lordan et al., 2013). Reka et al (2017) reported that aqueous and ethanol extracts of Ulva reticulate, Ulva lactuca, Gracilaria edulis, Gracilaria corticata against α-amylase and α-glucosidase at eight hours. The α- glucosidase inhibition activity of U. lactuca was 65.71% in ethanol extract.
Conclusion
The methanolic extracts of S. filamentosa may be attributed to the presence of phyto-chemicals analysis and inhibitory activity. The marine algae showed significant inhibition activity. Further compound isolation, purification and characterization which is responsible for inhibitory activity has been done usage of anti-diabetic agent.
Acknowledgement
The Director, Centre for Advanced studies in Botany, University of Madras would like to thank for their support and encouragement for UGC- BSR Fellowships in Sciences, University Grants Commission, New Delhi – 110002.
Conflicts of interest:
We declare that we have no conflict of interest.
References
De Sales PM, de Souza PM, Simeoni LA, Magalhaes PO, Silveira D. 2012. α-Amylase inhibitors: A review of raw material and isolated compounds from plant source. Journal of Pharmacy and Pharmaceutical Sciences, 15: 141–183.
Dharmesh R, Stalin K, Ramavatar M, Siddhanta AK, 2014. Antioxidant activity and phytochemical analysis of a few Indian seaweed species. Indian Journal of Geo-Marine Sciences, 43: 507-518.
Harborne JB, 1998. Methods of extraction and isolation. In: Phytochemical Methods (3rd Ed.). Chapman and Hall, London; pp: 60–66.
Kwon YI, Apostolidis E, Shetty K. 2008. In vitro studies of eggplant (Solanum melongena) phenolics as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. Bioresource Technology, 99: 2981–2988.
Lakshmana Senthil S, Vinothkumar T, Geatharamani D, Maruthupandi T. 2013. Screening of seaweeds collected from a south- east coastal area of India for α-amylase inhibitory activity, antioxidant activity and biocompatibility. International Journal of Pharmacy and Pharmaceutical Sciences, 5:240-4.
Lordan S, Smyth TJ, Soler-Vila A, Stanton C, Ross RP. 2013. The α-amylase and α-glucosidase inhibitory effects of Irish seaweed extracts. Food Chemistry, 141: 2170-2176.
Miller GL. 1959. Use of dinitrosalicyclic acid reagent for determination of reducing sugar. Analytical Chemistry, 31: 426-428.
Reka P, Thahira Banu A, Seethalakshmi M. 2017. Elemental composition of selected edible seaweeds using SEM- energy dispersive spectroscopic analysis. International Food Research Journal, 24(2): 600-606.
Ribeiro RA, Vale ML, Thomazzi SM, Aschoalato AB, Poole S, Ferreira SH, Cunha FQ. 2000. IK Involvement of resident macrophages and mast cells in the writhing nociceptive response induced by zymosan and acetic acid in mice. European Journal of Pharmacology, 387(1):111-118.
Saritha K, Mani AE, Priyalaxmi M, Patterson J. 2013. Antibacterial activity and biochemical constituents of seaweed U. lactuca. Global Journal of Pharmacology, 7: 276-282.
Sun Z, Chen F. 2012. Evaluation of the green algae Chlorella pyrenoidosa for the management of diabetes. Journal of Food and Drug Analysis, 20: 246-9.
Teixeira VL, Rocha FD, Houghton PJ, Auxiliadora M, Kaplan C, Pereira RC. 2007. α- amylase inhibitors from Brazilian seaweeds and their hypoglycemic potential. Fitoterapia, 78: 35–6.
Unnikrishnan PS, Suthindhiran K, Jayasri MA. 2015. α-amylase inhibition and antioxidant activity of marine green algae and its possible role in diabetes management. Pharmacognosy Magazine, 11:511–5.