

Abdul Haseeb M.1*, Mohammed Ziauddin Sarkhil1, Mohammed Fayazuddin2, Farida Ahmad3
1Department of Pharmacology, Kannur Medical College, Kannur - 670612 Kerala, India
2Department of Pharmacology, Raichur Institute of Medical Sciences, Raichur, Karnataka 584102 India
3Department of Pharmacology, Jawaharlal Nehru Medical College, AMU, Aligarh (Utter Pradesh) India
*Address for corresponding author
Abdul Haseeb M.
Department of Pharmacology, Kannur Medical College, Kannur - 670612 Kerala, India
Abstract
Moringa oleifera Lam. commonly known as the drumstick tree, is widely used in Indian cuisine with a number of medicinal properties. Objective: The objective of this study was to evaluate the peripheral analgesic activity of Moringa oleifera seeds. Material and methods: The Peripheral analgesic activity was studied using Acetic acid induced writhing method in Swiss Albino mice of either sex weighing 20-40g at doses of EEMO 50 mg/kg, 100mg/kg, 200mg/kg and AEMO 250 mg/kg, 500mg/kg, 750 mg/kg. Statistical significance was calculated using One-way analysis of variance (ANOVA) followed by post hoc dunnett's test. Results: EEMO at all three doses 50 mg/kg, 100 mg/kg and 200 mg/kg exhibited peripheral analgesic activity by significantly decreasing the number of writhes, when compared to control group. AEMO at doses of 500 mg/kg and 750 mg/kg decreased the number of writhes significantly in comparison to control group. Conclusion: Moringa oleifera seeds exhibits Peripheral analgesic activity in Swiss Albino mice.
Keywords: Acetic acid induced writhing method, Moringa oleifera, peripheral analgesic activity
Introduction
Pain is one of the most common presenting complaints of patients for which they seek medical advice. Pain is a critical component of body’s defence system that guides to move away from noxious stimuli. Pain prevents further damage after an initial insult and promotes the healing process of an injured body part (Woolf, 1995).
Pain is defined as, "an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage" by International Association for the Study of Pain (IASP) (Bonica, 1990).
Pain generation occurs when either of the two components are involved namely central or peripheral. Peripheral pain is most commonly associated with Inflammation (Schaible, 2007). Prostaglandins being inflammatory mediators induce hyperalgesia by inducing transducing property of free nerve endings. NSAIDS block the pain sensitizing mechanism induced by prostaglandins, bradykinin, TNFα, ILs, and other algesic substances. Hence they are more effective against inflammation associated pain (Ikoma et al., 2006).
Moringa oleifera is a well-known medicinal plant distributed in Indian sub-continent. Moringa oleifera leaves and pods are used in Indian cuisine for their nutritious value (Fuglie, 1999). Moringa oleifera is evaluated for its antifungal (Chuang et al., 2007), antioxidant (Verma et al., 2009), antibacterial (Rahman et al., 2009), immunomodulatory (Gupta et al., 2010) hepatoprotective (Chattopadhyay et al., 2011) and antidiabetic (Adisakwattana et al., 2011) activities. There are very few studies reporting the anti-inflammatory activity and one study reporting the analgesic activity of the Moringa oleifera seeds (Caceres et al., 1992; Guevara et al., 1996; Sudha et al., 2010). Hence the study was conducted for the scientific evaluation of the Moringa oleifera seeds using in vivo peripheral analgesic models.
Material and methods
Extraction of Plant material
The pods of Moringa oleifera were collected, seeds were separated and shade dried. The seeds were identified and authenticated by botanist Dr. Athar Ahmad Khan, Assistant Professor Department of Botany, Aligarh Muslim University. Specimen was deposited and voucher number (SC-0130/11) was obtained. The seeds were finely powdered in a grinder, and were stored in air-tight bottles till further use. 100 g of seed powder was extracted separately with 200 ml of distilled water and ethanol for Aqueous and Ethanolic extract with the help of Soxhlet’s apparatus. The extract obtained was filtered. Then the filtrate was collected in Petri dishes and evaporated till dryness at 40 ºC in an autoclave. The semisolid mass so obtained was weighed, their yield calculated, sealed with aluminium foils (airtight) and then stored at 4 ºC for further experimental work.
Drugs and Chemicals
Diclofenac sodium (Novartis, India), Propylene glycol (BDH, Mumbai) Ethanol (Merck Laboratories, Mumbai India) and were used in the study.
Animal Protocol
Swiss Albino mice of either sex (20-40 g) were procured from the Central Animal House, JNMC, Aligarh Muslim University. Mice were housed in polypropylene cages which were bedded with paper strips in the Pharmacology section of Central Animal House. The animal room was well ventilated and maintained under standard conditions throughout the experiment (temperature 27 ± 2°C, 12 hour light/dark cycles), and fed with standard rodent diet and water ad libitum. Mice were acclimatized to the experimental conditions for 1 week prior to use.
The study protocol was approved by the Institutional Animal Ethics Committee (IAEC) on 19.04.2011. Animal experiments were done according to the guidelines of CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) and rules and regulations of IAEC.
Analgesic activity by Acetic acid induced Writhing method
This method is used for the screening of peripherally acting analgesic agents. Swiss Albino mice weighing 20-40g were used. Pain is induced by intraperitoneal injection of 0.6% acetic acid (10ml/kg). The animals react with a characteristic stretching behaviour called writhing (Koster et al., 1959). All the drugs were administered 30 minutes before the injection of acetic acid. The mice were then placed individually into glass beakers and five min were allowed to elapse. The mice were then observed for a period of 20 min and the numbers of writhes are recorded for each animal. For scoring purposes, a writhe was counted as stretching of the abdomen along with stretching of at least one hind limb. Propylene glycol served as the control whereas Diclofenac sodium served as the standard drug. Mice were divided into eight experimental groups as shown in Table 1.
The formula for computing percent inhibition is
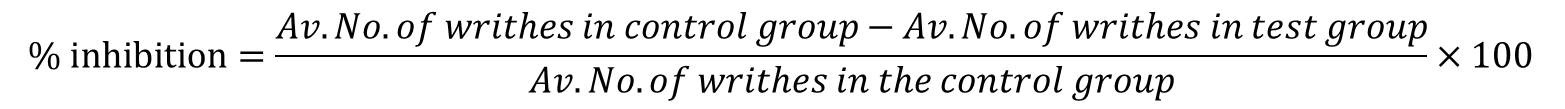
Table 1. Experimental groups for screening method
|
Groups |
Name |
Treatment received |
|
|
Normal Control |
Propylene glycol 2ml/kg, Orally. |
|
|
Standard Control |
Diclofenac sodium (5mg/kg). |
|
|
Test group- 1 |
Ethanolic extract 50mg/kg, Orally. |
|
|
Test group-2 |
Ethanolic extract 100mg/kg, Orally. |
|
|
Test group-3 |
Ethanolic extract 200mg/kg, Orally. |
|
|
Test group-4 |
Aqueous extract 250mg/kg, Orally. |
|
|
Test group-5 |
Aqueous extract 500mg/kg, Orally. |
|
|
Test group-6 |
Aqueous extract 750mg/kg, Orally. |
Statistical analysis
Each group comprised of 6 mice. Values were expressed as Mean ± SEM. SPSS-17 software was used for statistical tests. One way ANOVA followed by post hoc Dunnett’s multiple comparison test was employed for statistical significance. p <0.05 was taken as statistically significant.
Results
Plant extracts
The aqueous and ethanolic extracts of Moringa oleifera seeds were prepared by Soxhlet extraction using Distilled water and Ethanol respectively.
Ethanolic extract- It was a green coloured semi solid mass of oily consistency. Yield obtained was 7.54%.
Aqueous extract – It was a brown coloured semi solid mass. Yield obtained was 16.2%.
Analgesic activity by Acetic acid induced writhing method
The analgesic activity was evaluated by recording the number of writhes in test and control groups. All the values are expressed as Mean ± SEM in Table 2 and 3 and Graphically represented in Figures 1 and 2.
Table 2 shows number of writhes and % inhibition in control, Diclofenac sodium and ethanolic extract treated groups. In Diclofenac sodium 5mg/kg treated group, there was maximum inhibition of writhes compared to control group (p<0.001). All EEMO treated groups demonstrated dose depended decrease in number of writhes.
EEMO in doses of 50, 100 and 200 mg/kg significantly (p<0.001) decreased number of writhes when compared to control. EEMO inhibited the number of writhes by 31.05%, 41.31%, and 55.55% at 50, 100 and 200mg respectively.
Table 2. Analgesic effect of Ethanolic extract of Moringa oleifera seeds by Acetic acid induced writhing test
|
Groups |
Number of writhes |
% Inhibition |
|
Control |
58.5±1.99 |
--- |
|
Diclofenac sodium (5mg/kg) |
17.8±1.64** |
69.57 |
|
EEMO (50mg/kg) |
40.33±2.49** |
31.05 |
|
EEMO (100mg/kg) |
34.33±1.76** |
41.31 |
|
EEMO (200mg/kg) |
26±1.57** |
55.55 |
Figure 1. Shows the changes in No. of writhes occurring in various groups after administration of the Ethanolic extract and control drugs.
Figure 1. Analgesic effect of EEMO by acetic acid induced writhing test
Table 3 shows number of writhes and % inhibition in control, Diclofenac sodium and Aqueous extract treated groups. In Diclofenac sodium 5mg/kg treated group, there was maximum inhibition of writhes compared to control group (p<0.001).
There was no significant decrease in number of writhes in AEMO 250mg/kg treated group as compared to control group. AEMO in doses of 500 and 750mg/kg significantly (p<0.001) decreased number of writhes in dose dependent manner when compared to control group. AEMO inhibited the number of writhes by 34.18% and 40.19% at the dose of 500 and 750mg/kg respectively.
Table 3. Analgesic effect of aqueous extract of Moringa oleifera seeds on Acetic acid induced writhing test
|
Groups |
Number of writhes |
% Inhibition |
|
Control |
58.5±1.99 |
---- |
|
Diclofenac sodium (5mg/kg) |
17.8±1.64** |
69.57 |
|
AEMO (250mg/kg) |
58.16±2.37 |
0.58 |
|
AEMO (500mg/kg) |
38.5±2.29** |
34.18 |
|
AEMO (750mg/kg) |
35±1.88** |
40.19 |
AEMO: Aqueous extract of Moringa oleifera seeds, n = 6 in each group; *indicates p<0.05, ** indicates p <0.001 when compared to the control group.
Figure 2. Shows the changes in No. of writhes occurring in various groups after administration of the aqueous extract and control drugs.
Figure 2. Analgesic effect of AEMO by acetic acid induced writhing test
Discussion
Pain is a common symptom with which a health care professional deals in clinical practice. Analgesic therapies for acute and chronic pain conditions currently rely on three major classes of drugs: Nonsteroidal anti-inflammatory drugs (NSAIDs), Opioids, and a group of drugs with diverse pharmacological actions such as antidepressants, anticonvulsants, local anaesthetics.
NSAIDS are nonselective inhibitors of the enzyme cyclooxygenase (COX-1 and COX-2) which is responsible for the production of prostaglandins. Prostaglandins do not cause pain as such, but they potentiate the analgesic effect of other agents such as 5-hydroxytryptamine or bradykinin. They sensitize nerve terminals to other agents partly by inhibiting potassium channels and partly by facilitating-through second messenger mediated phosphorylation reactions the cation channels opened by noxious agents. NSAIDS block the pain sensitizing mechanism induced by bradykinin, TNFα, ILsand other algesic substances. Hence they are more effective against inflammation associated pain.However their adverse effect profile is well known which prompts to search for newer peripheral analgesics (Samad et al., 2002)
The peripheral analgesic activity was studied by acetic acid induced writhing method. It is most commonly used method for evaluating peripheral analgesic activity of a test compound.It is a chemical method where in an irritant compound like acetic acid is injected in peritoneal cavity to induce pain of peripheral origin. Writhe is a response to injection acetic acid which induces pain via nociceptors. It is noted by stretching of the abdomen with simultaneous stretching of at least one hind limb. Analgesic activity of the test compound is measured by decrease in the number of writhes (Shivaji and Gawade, 2012).
Both Ethanolic and Aqueous extracts of the Moringa oleifera seeds exhibited dose dependent significant (p<0.001) decrease in number of writhes at the doses of 50mg/kg, 100 mg/kg and 200 mg/kg of EEMO and AEMO at the dose of 500mg/kg and 750mg/kg (Table 2 and 3). But the Ethanolic extract showed greater analgesic effect than aqueous extract. The peripheral analgesic activity produced by EEMO 200mg/kg (55.55%) was comparable with standard drug Diclofenac sodium (69.57%) (Table 2).
In the present study it can be suggested that Moringa oleifera seeds possess peripheral analgesic activity. The mechanism of peripheral analgesic activity may be due inhibition of cyclooxygenase in the prostaglandin pathway. The analgesic activity of Moringa oleifera seeds can be attributed to various phytochemicals like flavonoids, saponins and alkaloids present in the seeds (Sanchez et al., 2006). Semi purified flavanoids have demonstrated analgesic activity in acetic acid induced writhing method (Cristal et al., 2018). Flavanoids from leaves of Juniperus sabina caused a significant dose-dependent inhibition on acetic acid induced writhing response in mice (Jun Zhao et al., 2018).
References
Adisakwattana S, Chanathong B. 2011. Alpha-glucosidase inhibitory activity and lipid-lowering mechanisms of Moringa oleifera leaf extract. European Review for Medical and Pharmacological Sciences, 15(7):803-808.
Bonica JJ. 1990. Definitions and taxonomy of pain. In: Bonica (Ed 2). The Management of Pain, pp. 18-27, Philadelphia, Lea & Febiger.
Cáceres, Amarillis Saravia, Sofia Rizzo, Lorena Zabala, Edy De Leon, Federico Nave. 1992. Pharmacologie properties of Moringa oleifera. 2: Screening for antispasmodic, anti-inflammatory and diuretic activity. Journal of Ethnopharmacology, 36(3):233-237.
Chattopadhyay S, Maiti S, Maji G, Deb B, Pan B, Ghosh D. 2011. Protective role of Moringa oleifera (Sajina) seed on arsenic-induced hepatocellular degeneration in female albino rats. Biological Trace Element Research, 142(2):200-12.
Chuang, Lee CW, Chou JY, Murugan M, Shieh BJ, Chen HM. 2007. Anti-fungal activity of crude extracts and essential oil of Moringa oleifera Lam. Bioresource Technology, 98:232–236.
Cristal Mae B Pedong, Mary Regielynn P Ilao, Sarah L De Lima, Shayne V Manarang, April B Lingat, Mariel Niña M Esguerra, Billy K Singian. 2018. Evaluation of the analgesic property of the semi-purified flavonoids from the fronds of Diplazium esculentum ethanolic extract. International Journal of Research in Pharmacy and Pharmaceutical Sciences, 3(1):154-159.
Fuglie LJ. 1999. The Miracle Tree. In: Fuglie LJ, The Multiple Attributes of Moringa, pp, 172, Dakar, Church World Service.
Guevara AP, Vargas C, Sakurai H, Fujiwara Y, Hashimoto K, Maoka T, Kozuka M, Ito Y, Tokuda H, Nishino H. 1999. An antitumor promoter from Moringa oleifera Lam. Mutation Research, 440: 181–188.
Gupta A, Gautam MK, Singh RK, Kumar MV, Rao ChV, Goel RK, Anupurba S. 2010. Immunomodulatory effect of Moringa oleifera Lam. extract on cyclophosphamide induced toxicity in mice. Indian Journal of Experimental Biology, 48(11):1157-60.
Ikoma A, Steinhoff M, Ständer S, Yosipovitch G, Schmelz M. 2006. The neurobiology of itch. Nature Reviews Neuroscience, 7 (7):535–47.
Jun Zhao, Aosimanjiang Maitituersun, Chenyang Li, Qian Li, Fang Xu, Tao Liu. 2018. Evaluation on Analgesic and Anti-Inflammatory Activities of Total Flavonoids from Juniperus Sabina. Evidence-Based Complementary and Alternative Medicine. 7965306.
Koster R, Anderson M, De Beer EJ. 1959. Acetic acid analgesic screening. Federation Proceedings 18, 412-417.
Rahman MM, Islam Sheikh, Sharmin SA, Islam MS, Rahman A, Mizanur Rahman M, Alam MF. 2009. Antibacterial Activity of Leaf Juice and Extracts of Moringa against some Human Pathogenic Bacteria. Chiang Mai University Journal of Natural Sciences, 8(2):219-227.
Samad TA, Sapirstein A, Woolf CJ. 2002. Prostanoids and pain: unravelling mechanisms and revealing therapeutic targets. Trends in Molecular Medicine, 8:390-396.
Sanchez MDI, Lopez CJ, Rios V. 2005. High-performance liquid chromatography method to measure ∝- and γ-tocopherol in leaves and fresh beans from Moringa oleifera . Journal of Chromatography A, 1105(1-2):111–114.
Schaible HG. 2007. Peripheral and central mechanisms of pain generation. Handbook of Experimental Pharmacology, 177:3-28
Shivaji P Gawade. 2012. Acetic acid induced painful endogenous infliction in writhing test on mice. Journal of Pharmacology and Pharmacotherapeutics, 3(4):348.
Sudha P, Asdaq SM, Dhamingi SS, Chandrakala GK. 2010. Immunomodulatory activity of methanolic leaf extract of Moringa oleifera in animals. Indian Journal of Physiology and Pharmacology, 54(2):133-40.
Verma M. Vijayakumar, Chandra S. Mathela, Chandana V. Rao. 2009. In vitro and in vivo antioxidant properties of different fractions of Moringa oleifera leaves. Food and Chemical Toxicology, 47(9):2196-2201.
Woolf CJ. 1995. Somatic pain-pathogenesis and prevention. British Journal of Anaesthesia, 75:169-176.