

Ramchandra Gupta1, Ashish Garg1, Prabhakar Sharma1, Prakash Pandey2
1Department of Pharmacy, Guru Ramdas Khalsa Institute of Science and Technology, Pharmacy, Barela, Jabalpur, M.P. 483001
2Department of Chemistry and Pharmacy, Rani Durgavati Vishwavidyalaya (RDVV), Jabalpur, M.P. 482001.
Corresponding author
Ashish Garg
Department of Pharmacy,
Guru Ramdas Khalsa Institute of Science and Technology, Pharmacy, Barela, Jabalpur, M.P. 483001 India
Abstract
Objective: The present study deals with the phytochemical and biological evaluation of Calliandra haematocephala leaves for its wound healing activity in rats. Materials and methods: Leaves of C. haematocephala were collected and used for size reduction to make powder. Powder leaves of plant (100 gm) were extracted with ethyl acetate by hot continuous extraction process using Soxhlet apparatus. Ethyl acetate extract was screened for chemical constituents and evaluated wound healing effect by incision and excision in vivo wound models. The various antioxidants were also estimated into the healed skin to confirm the antioxidant effect. Results: The ethyl acetate was found most chemical constituent i.e. glycosides, terpenoids, tannins and flavonoids. The 2.5 and 5% ethyl acetate extract ointment of Calliandra haematocephala were subjected for detailed study of wound healing activity using incision wound and excision wound models. Healing effect was confirmed by significant (P< 0.05) increase in tensile strength as well as hydroxproline and protein level of healed skin tissues. The histological studies also support the satisfactory wound healing activity of ethyl alcohol extract of Calliandra haematocephala. Conclusion: Calliandra haematocephala leaves found potent wound healing effect in rats and mechanism of healing can be correlated with its antioxidant property.
Keywords: Wound healing, Calliandra haematocephala, incision, excision, antioxidant
Introduction
Wound is a disruption of cellular and anatomical continuity of a living tissue. It may be produced by physical, chemical, thermal or microbial insult to the tissue. When skin is torn, cut, or punctured it is termed as an open wound and when blunt force trauma causes a contusion, it is called closed wound, whereas the burn wounds are caused by fire, heat, radiation, chemicals, electricity, or sunlight (Shuid et al., 2005; Jalalpure et al., 2008). Wound healing is a multifaceted development consists of various stages namely wound, wound closure and re-establishment of a functional barrier. The sequential phases of healing process are inflammation, proliferation and migration of connective tissue cells, making of extracellular matrix as well as collagen synthesis, epithelial cells migration and proliferation most important to new vessels formation of wounded tissue (Strodtbeck, 2001).
C. haematocephala, (family: Mimosaceae) is commonly known as red powder puff. The red powder puff flowers are attractive to butterflies and hummingbirds but only appear during November-April. It is a rambling shrub with height of 1.5-4 meters has branched pinnates and silky leaves (Kiritikar and Basu, 1999). The presence of antibacterial and insecticidal properties in leaves and seeds from former researches has been reported (Parekh and Chanda, 2007). C. haematocephala is a branched plant, height from 1 to 3 meters. Its branches are brown, cylindric and rough. Leaves are oblong, acute, with 5 to 10 pairs of glossy green leaflets. It can be grown in gardens and parks and found native to tropical America and India. Phytochemical investigations of the C. haematocephala have been extensively done in last few decades and were reported to presence of flavonoids, carbohydrates, alkaloids, glycosides, saponins, steroids and tannins as major category of compounds. This plant is traditionally used as anti-oxidant and as blood purifier (Moharran, 2006). Ethyl acetate fraction of C. haematocephala barks consist of p-hydroxybenzoic acid, protocatechuic acid, caffeic acid, astilbin, neo-isoastilbin lupeol and betulinic acid. The plant genus showed various pharmacological properties e.g. anticonvulsant, anti-inflammatory, immunomodulatory and antiulcerogenic activity. Its butanolic extract reported for gastroprotective effects in acute gastric lesions (A de Paula et al., 2012). It also used traditionally as antibacterial (Nia et al., 1999). Recently, C. haematocephala effervescent granules have been formulated for the treatment of gastric ulcer treatment (Gupta et al., 2013). The present study was aimed to evaluate wound healing and antioxidant effect of leaves of C. haematocephala on incision and circular in vivo wound models in rats.
Materials and methods
Identification and collection of plant materials
The fresh leaves of C. haematocephala were collected during early summer from young matured plant from the rural belt of Jabalpur (M.P.) and C. haematocephala plant material was authenticated by Tropical Forest Research Institute, Jabalpur. The leaves were washed under running tab water, cut in small pieces and dried in shade for 15 days. To ensure complete dryness plant leaves were kept in hot air oven at 40°C for 5 minutes.
Successive solvent extraction
The extraction of drug (solid extraction) represents a solid from solid separation, as solid components must be extracted from a solid substance. The collected, cleaned leaves of C. haematocephala were used for the extraction process. Powder leaves of plant (100 gm) were extracted successively with petroleum ether and ethyl acetate by hot continuous extraction process using Soxhlet apparatus and porcelain bits were added to avoid the bumping during extraction in round bottom flask (Lodhi et al., 2010; Lodhi and Singhai, 2013).
The extracts were filtered, while hot, to remove any impurities, if present. The extracts were concentrated by vacuum distillation to reduce the volume 1/10. The concentrated extracts were transferred to 100 ml beaker and the solvent were removed by evaporating on the water bath. Then the semisolid mass was obtained were collected and placed in desiccators to remove the excessive moisture. The dried extracts were packed and labeled in air tight container for the further studies such as a phytochemical screening.
Qualitative Analysis
After complete extraction the preliminary tests were carried out for the presence or absence of phytoconstituents like glycosides, flavanoids, saponins, alkaloids, carbohydrates, sterols, proteins, phenolic compounds. Phytochemical examinations were carried out for ethyl acetate extract as per the standard procedure (Roopashree et al., 2008; Obasi et al., 2010; Audu et al., 2007).
Animal protocol
Wistar albino rats (150-200 g) were selected for in vivo study. The animals were acclimatized to the standard laboratory conditions in cross ventilated animal house at temperature 25±2°C relative humidity 44 –56% and light and dark cycles of 12:12 hours, fed with standard pallet diet and water during experiment. The rats were used after acclimatization to the laboratory environment for about 7 day’s period prior experiment. Animals were divided in four groups each group containing six animals. The control group received ointment base only. Standard group received topically Povidone iodine ointment. Test group treated with topically 2.5 and 5% w/w ethyl acetate extract ointment.
Incision wound model
All the animals were anaesthetized with diethyl ether before wound creation and the back hair of the rats were shaved. Full thickness of skin of 1.5 cm long incision was made through the skin at dorsal portion of rat skin with sterile scalpel or bled (Lodhi and Singhai, 2011). The parted skin was kept together and stitched with surgical thread (No. 1) and a curved needle (No.22). Both wound edges were tightened for good closure of the wound and after stitching, wound was left undressed. The wounding day was considered day 0. The all extracts and the standard drug ointment were topically applied once a day for 9 days. All the sutures were removed on the 9th day of post wounding. On 10 day, breaking or tensile strength was measured using Tensinometer (Lee, 1968; Suntar et al., 2010).
Tensile Strength Measurement
Tensile strength is the resistance to breaking under tension. It indicates resistance of tissue to breaking under tension and may indicate in part the required tissue. The newly repaired tissue including scar was tested to measure the tensile strength by using an instrument known as Tensiometer. Before testing, the rats were anesthetized with diethyl ether and secured to the operating table. Once day before measurement of tensile strength, the sutures were removed from the stitched wounds of rat’s recovery. The animal was then placed on stack of paper towels on the middle of the board. The two clamps were then carefully applied on the skin 0.5 cm away from the edge of wound facing each other on opposite side of the incision wound. One of the clamp or forceps was fixed on stands, while the other was connected to a freely suspended light weight plastic (Polyethylene) bottle of volume 1000 ml through a string run over to a pulley. Water was allowed to flow continuously from the reservoir slowly and steadily into the container. The moment the wound just opened up, the amount of water in polyethylene bottle was weighed and consider to as the tensile strength of wound (Ilango and Chitra, 2010).
Excision wound model
The rats were anesthetized with Diethyl Ether before and during infliction of experimental wounds. The hairs on the dorsal thoracic area of the animals were shaved and ethanol was applied on the region before making the wound. Thereafter open circular wound of 500 mm2 area was produced in each rat by excising the skin. Rats wound were left undressed to the open environment. After 24 h of wound creation, the ointments/gel was applied gently to cover the wounded area once daily until complete healing achieved (Singh et al., 2016). Wound area and wound contraction, epithelialization period was observed (Mukherjee et al., 2000; Charde, et al., 2005).
Wound area and wound contraction and epithelialization time
The ointment was topically applied once daily until the wounds were completely healed. Wound area was measured at day zero (450mm2) and once every 3 days post wounding. Excision wound margins were traced using transparent paper. The tracing was then shifted to graph paper, from which the wound surface area was obtained. The measured surface area at different days of post wound creation was employed to calculate the percentage of wound contraction, taking initial size of wound as 100 %, according to reported method (Shenoy et al., 2011).
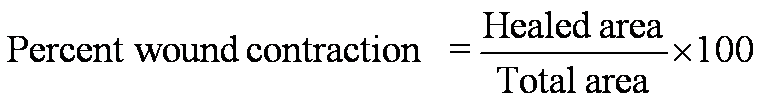
Hydroxyproline and protein estimation
The wound tissue samples were analyzed for hydroxyproline content, which is a basic constituent of collagen. Tissues were dried at 60-70 0C to constant weight in a hot air oven. After drying tissues were hydrolysed in 6 N HCl at 130 0C for 4 hour in sealed tubes. After neutralization of hydrolyze tissue to pH 7 then subjected to Chloramine-T oxidation for 20 minutes (Woessner, 1961). The reaction was terminated by addition of 0.4M perchloric acid and developed color with Ehlrich reagent at 60 0C was read at 557 nm in UV (Agilant Technology) Spectrophotometer.
Tissue protein was estimated from tissue lysate by treatment with a mixture of sodium tartrate, copper sulphate and sodium carbonate. The mixture was left to stand for 10 minutes and then treated with Folin-Ciocalteau reagent that resulted in a bluish color in 20-30 minutes. The absorbance was measured using standard Spectrophotometry techniques 660 nm (Lowry et al., 1951).
Estimation of antioxidants level in skin tissues
The granuloma tissues sample were collected from wounded site and tested for antioxidants assay. Superoxide dismutase (SOD) assay was based on the inhibition of epinephrine autoxidation by the enzyme (Misra and Fridovich, 1972). Catalase was estimated following the breakdown of hydrogen peroxide (Beers and Sizer, 1952). Reduced glutathione (GSH) level was determined by method of Moron et al., (1979). Tissue homogenates were immediately precipitated with 0.1 ml of 25% TCA and separated by centrifugation. The assay of free-SH groups in 3 ml of sample done by the addition of 2 ml of 0.6 mM DTNB and 0.9 ml 0.2 mM sodium phosphate buffer (pH 8.0) to 0.1 ml of the supernatant and the absorbance was read at 412 nm using a UV spectrophotometer.
Histopathological studies
Wound tissue specimens were collected from wounded site of excision wound and after usual processing. 6 mm thick sections were cut and stained with haematoxylin and eosin. Sections were qualitatively assessed under the light microscope and observed in respect of fibroblast proliferation, collagen formation and epithelialization.
Statistical analysis
Data were presented as the mean ± Standard Deviation. Treated groups were compared with the standard group. The results were analyzed statistically using Student’s t-test for the comparison. The data were considered significant at P < 0.05.
Results and discussion
Extraction and Phytochemical study
In the present study of Calliandra haematocephala (leaves) extract was subjected to phytochemical and pharmacological studies for evaluation of wound healing activity. The plant material was selected on the basis of traditional uses and phytochemical constituent present in its leaves. The powered plant material (leaves) was successively extracted with ethyl acetate. The percentage yields of all extract of Calliandra haematocephala was found to be 2.5% w/w. The phytochemical tests showed the presence of glycosides, terpenoids, tannins, and flavonoids.
Incision wound model
The tensile strength of the wounds treated with ethyl acetate extract of Calliandra haematocephala was 560±2.56 and reference ointment was 589±3.67 significantly (P< 0.01) higher than the control group (330±3.45) of animals (Table 1). Collagen imparts tensile strength and elasticity to healed skin. An increase in collagen in the treated wound correspond with significantly increased of tensile strength in treated groups of incision wound over that in the untreated wound. According to Singer and Clack (1999), wounds gain 20% of their final strength in the first 3 weeks post wounding. The tensile strength of wound can be related to its collagen formation and maturation.
Table 1. Effect of Calliandra haematocephala extract ointment on tensile strength of incision wound in rats
|
Treatment groups |
Tensile strength (gm/cm2) |
|
Control (Base) |
205±2.05 |
|
Reference ointment |
576±2.40 |
|
Ethyl acetate extract ointment (5%w/w) |
540±2.20 |
n = 6 albino rats per group, value represent Mean ± S.D.; P< 0.01, when compared each treated group with control group
The tensile strength indicates how much the required tissue resists to breaking under tension and may indicate in part the quality of repaired tissue. The results of the measurement of tensile strength on day 10th were shown in Table 1. The tensile strength of animals treated with ethyl acetate extract and reference ointment was significantly greater than that of control. In case of Calliandra haematocephala the significant difference (P<0.01) was observed with the ethyl acetate extract.
Excision wound model
The percentage wound contraction was determined at two days interval. The wound margins were traced and measured to calculate the non healed area which then subtracted from the original wound area to obtain the healed area. The wound contraction percentage was found as observed on 16th day in the ethyl acetate extract ointment (92.39±3.31) and reference ointment (95.40±3.57) was significantly higher than that of control group of animals. The mean epithelialization time was also comparatively less in these groups compared to control group of animal (Table 2 and figure 1).
Table 2. Effect of prepared ointment and reference ointment on percentage of wound contraction area of excision wound in rats
|
Treatment groups |
Post wounding days (Percent wound contraction) |
Epithelializa-tion period |
|||||||||
|
2 |
4 |
6 |
8 |
10 |
12 |
14 |
16 |
18 |
20 |
||
|
Control (Base) |
8.05± 0.67 |
12.49±1.87 |
20.62± 0.83 |
26.37± 2.46 |
34.52±1.47 |
46.24±1.39 |
55.38±2.32 |
69.82±2.57 |
76.35±3.48 |
83.64±0.87 |
22 |
|
Ethyl acetate extract ointment (5%w/w) |
12.49± 0.56 |
15.38±1.38 |
25.30± 0.35 |
37.94± 1.32 |
48.41±1.51 |
67.04±2.47 |
81.29±2.45 |
92.39±3.31 |
100.00 |
|
18 |
|
Reference ointment |
13.64± 0.87 |
19.93±2.57 |
31.24± 2.87 |
40.47± 1.74 |
52.74±1.63 |
69.93±2.25 |
84.48±2.87 |
95.40±3.57 |
100.00 |
|
18 |
n = 6 albino rats per group, value represents Mean ± S.D.; *P < 0.01, when compared each treated group with control group
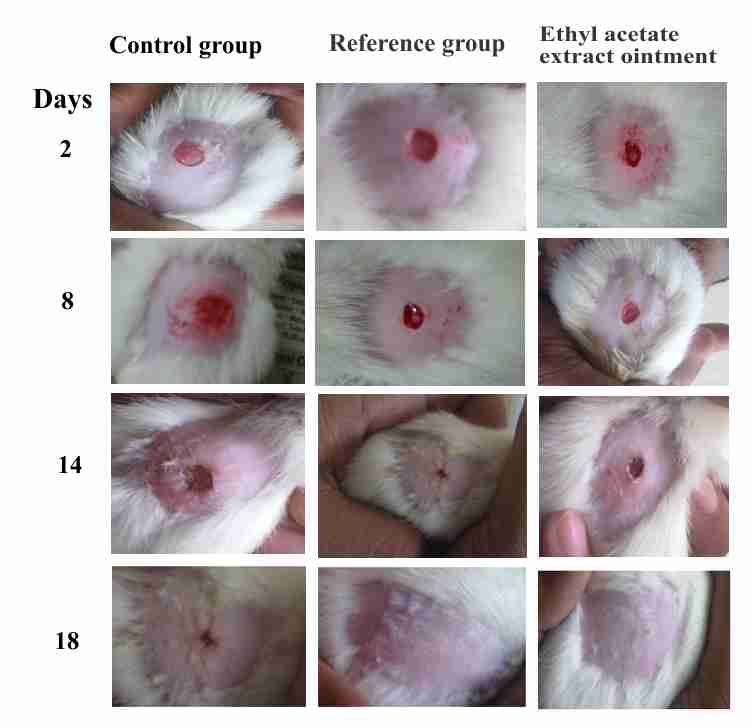
Figure 1. Wound contraction of different treatment groups: (a) Control group; (b) Ethyl acetate extract ointment group; (c) Reference group
The hydroxyproline content represents to the formation of collagen fibers in to the tissues. The protein content found as higher significantly in the group treated with ethyl acetate extract ointment and reference ointment groups. The hydroxyproline content of ethyl acetate extract ointment group found as 54.17±4.61 which is very close to the reference group 55.62±4.32. It means the prepared ointment was effective in wound healing. The protein content of 5% w/w extract ointment treated groups was showed higher than the control group. The results were comparable to the reference ointment group (Table 3). The higher protein content suggests that ethyl acetate extract through an unknown mechanism, stimulate cellular proliferation.
Table 3. Effect of prepared formulations and reference ointment on different biochemical parameters of excision wound in rats
|
Treatment groups |
Hydroxyproline (mg/g tissue) |
Protein content (mg/g tissue) |
Antioxidants level |
||
|
SOD (μg/50 mg tissue) |
CAT (μmol/50 mg tissue) |
GSH (μmol/50 mg tissue) |
|||
|
Control (Base) |
37.26±2.63 |
46.24±3.25 |
12.47±1.25 |
17.64±1.67 |
11.63±1.35 |
|
Ethyl acetate extract ointment (5%w/w) |
54.17±4.61* |
75.62±5.17* |
26.31±1.84* |
41.82±3.56* |
25.01±1.87* |
|
Reference Ointment |
55.62±4.32* |
81.20±7.45* |
28.52±2.01* |
45.10±4.26* |
27.61±2.14* |
n = 6 albino rats per group, value represents Mean ± S.D.; *P < 0.01, when compared each treated group with control group
The antioxidant enzymes levels in the granuloma tissue found significantly increased during wound healing process. SOD and CAT activity in granulation tissue were resulted significantly increased in case of rats treated with ethyl acetate extract ointment when compared with control group. GSH level in granulation tissue was also significantly increased in rats treated with extract ointment.
Histological studies
Histological studies of the tissue obtained from the treated group showed significant increase in collagen deposition, and more fibroblasts. It was approximately equal to the animals treated with reference ointment (Povidone-iodine Ointment USP). It was observed that ethyl acetate extract of Calliandra haematocephala ointment and reference ointment showed significant moderate deposition of collagen and less macrophages than control group of animals (Figure 2).
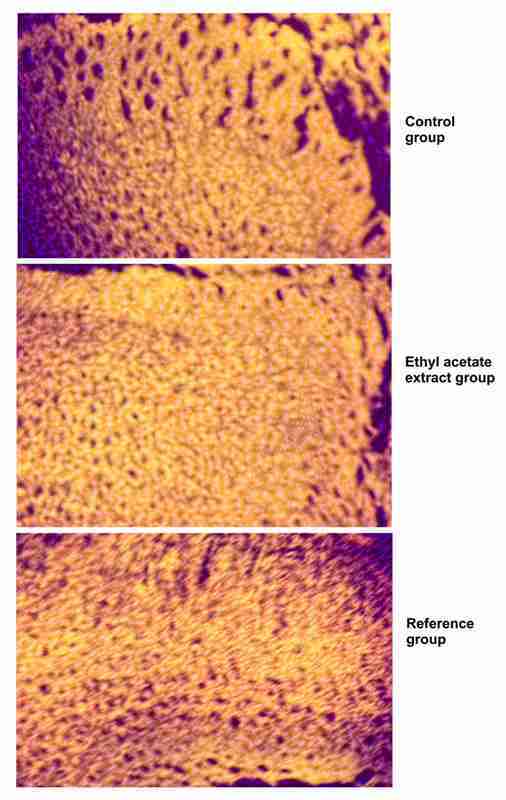
Figure 2. Histological observations of different treatment groups: (a) Control group; (b) Ethyl acetate extract ointment group; (c) Reference group
Conclusion
The leaves of Calliandra haematocephala confirmed the presence of glycosides, flavonoids and terpenoids in ethyl acetate extract. This extract was significantly improving healing effect with improvement of antioxidant levels in wounded tissues. Possible healing mechanism can be antioxidant mechanism of Calliandra haematocephala.
Conflict of interest
Authors did not have any conflict of interest.
References
Audu SA, Mohammed I, Kaita HA. 2007. Phytochemical screening of the leaves of Lophira lanceolata (Ochanaceae). Life Science Journal, 4: 75-79.
Beers RF, Sizer IW. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. The Journal of Biological Chemistry, 1952; 195: 133–140.
Charde MS, Fulzele SV, Satturwar PM, Joshi SB, Kasture AV, Dorle AK. 2005. Study on wound healing activity of Mulathiaadi Ghrita. Journal of Pharmaceutical Research, 4: 08‐12.
Gupta R, Sharma P, Garg A, Shukla A, Jain AP. 2013. Investigation of in-vitro Anthelmintic activity of Ficus elastica leaves. Journal of Drug Discovery and Therapeutics, 1(5): 01-03.
Jalalpure SS, Agrawal N, Patil MB, Chimkode R, Tripathi A. 2008. “Antimicrobial and wound healing activities of leaves of Alternanthera sessilis Linn”. International Journal of Green Pharmacy, 2: 141–144.
Kiritikar KR, Basu BD. 1999. Indian Medicinal Plants; International book distributors’ booksellers and Publisher. pp 749.
Lee KH. 1968. Studies on mechanism of action of salicylates: retardation of wound healing by aspirin. Journal of Pharmaceutical Sciences. 57: 1042–1043.
Lodhi S, Pawar RS, Jain AP, Jain A, Singhai AK. 2010. Effect of Tephrosia purpurea (L) Pers. on partial thickness and full thickness burn wounds in rats. Journal of Complementary and Integrative Medicine, 7(1): 13.
Lodhi S, Singhai AK. 2011. Preliminary Pharmacological evaluation of Martynia annua Linn leaves for Wound healing. Asian Pacific Journal of Tropical Biomedicine, 1(6): 421-427.
Lodhi S, Singhai AK. 2013. Wound healing effect of flavonoid rich fraction and luteolin isolated from Martynia annua Linn. on streptozotocin induced diabetic rats. Asian Pacific Journal of Tropical Medicine, 6 (4): 253-259.
Lowry OH, Rosenbrough NJ, Farr AL, Randall BJ. 1951. Protein measurement Folin phenol reagent. The Journal of Biological Chemistry, 193: 265-275.
Misra HP, Fridovich I. 1972. The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. The Journal of Biological Chemistry, 247: 3170–3175.
Moharram FA, Marzouk MS, Ibrahim MT, Mabry TJ. 2006. Antioxidant Galloylated Flavonol Glycosides from Calliandra haematocephala; Natural Product Research, 20(10): 927 – 934.
Moron MA, Depierre JW, Mannervick B. 1979. Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochimica et Biophysica Acta, 582: 67–78.
Mukherjee PK, Verpoorte R, Suresh B. 2000. Evaluation of in‐vivo wound healing activity of Hypericum patulum (Family: Hypericaceae) leaf extract on different wound model in rats. Journal of Ethnopharmacology, 70: 315‐321.
Nia R, Adesanya SA, Okeke IN, Illoh HC, Adesina SJ. 1999. Antibacterial Constituents of Calliandra haematocephala. Nigerian Journal of Natural Products and Medicine, 3:58-60.
Obasi NL, Egbuonu ACC, Ukoha PO, Ejikeme PM. 2010. Comparative phytochemical and antimicrobial screening of some solvent extracts of Samanea saman pods. African Journal of Pure and Applied Chemistry, 4 (9): 206-212.
Parekh J. Chanda SV. 2007. In vitro antimicrobial activity and phytochemical analysis of some Indian Medicinal Plants. Turkish Journal of Biology, 31: 53-58.
Roopashree TS, Dang R, Rani SRH, Narendra C. 2008. Antibacterial activity of anti-psoriatic herbs: Cassia tora, Momordica charantia and Calendula officinalis. International Journal of Applied Research in Natural Products. 1(3): 20-28.
Shenoy RR, Sudheendra AT, Nayak PG, Paul P, Kutty NG, Rao CM. 2011. Normal and delayed wound healing is improved by sesamol, an active constituent of Sesamum indicum (L.) in albino rats. Journal of Ethnopharmacology, 133: 608–612.
Shuid AN, Anwar MS, Yusof AA. 2005. The effects of Carica papaya Linn. latex on the healing of burn wounds in rats. Jurnal Sains Kesihatan Malaysia, 3(2): 39–47.
Singer AJ, Clark RA. 1999. Cutaneous wound healing. New England Journal of Medicine, 341: 738–746.
Singh S, More PK, Shukla R. 2016. Ameliorative effect of flavonoids rich extract from Plantago lanceolata for cutaneous wound healing with antioxidant mechanism. Advance Pharmaceutical Journal, 1(1): 21-25.
Strodtbeck F. 2001. Physiology of wound healing. Adv Skin Wound Care, 1: 43-52.
Suntar IP, Akkol EK, Yilmazer D, Baykal T, Kirmizibekmez H, Alper M, Yesilada E. 2010. Investigationsonthe In-vivo wound healing potential of Hypericum perforatum L. Journal of Ethnopharmacology, 127 468–477.
Woessner JF. 1961. The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Archieves of Biochemistry and Biophysics, 93: 440-447.