

Susan Kurian*, Leenamma Joseph, V. S. Josekumar
Department of Zoology, Mar Ivanios College (Autonomous), Thiruvananthapuram-695015, Kerala, India
*Address for Corresponding author
Ms. Susan Kurian
Assistant Professor,
Department of Zoology,
Mar Ivanios College, Thiruvananthapuram, Kerala- 695015. India.
Abstract
Objective: The objective of present study was to evaluate the antimicrobial and phytochemicals successive extracts of the leaves petiole of Artocarpus heterophyllus Lam. Methods: The hexane (AHH), ethyl acetate (AHE) and methanol (AHM) extracts of A. heterophyllus were quantitatively analysed for phytochemicals, viz. total phenolics, flavonoids, alkaloids and carbohydrates following standard method. The antimicrobial activity and minimal inhibitory concentration (MIC) of the plant extracts were examined against five bacterial and two fungal strains using agar well diffusion method and two fold serial dilution methods, respectively. Results: The quantitative phytochemical studies of the crude extracts showed methanol extract with highest phenolic, flavonoids, alkaloids and carbohydrates content which was followed by AHE and AHH extracts. There exists significant mean difference in the microbial activities among various extracts (P> 0.01). The maximum activity was exhibited by methanol extract. The GC-MS analysis of methanol extract identifies different phytocomponents which are biologically active. Conclusion: The present study reveals the presence of components highest in AHM that contribute to the pharmacological activity of the plant.
Keywords: Artocarpus heterophyllus, leaf petiole, phytochemical evaluation, Antimicrobial activity, GC-MS
Introduction
Medicinal plants have been used by humans in traditional medicines due to their therapeutic potential since ages (Vital and Rivera, 2009). About 80% of the world’s population relies on traditional medicine for their primary healthcare. Hence there arises a need to screen medicinal plants for bioactive compounds or further pharmacological studies (Hasan et al., 2007). Plants are a rich source of secondary metabolites with various biological activities (De Fatima et al., 2006).
Artocarpus heterophyllus Lam. (Jackfruit) belonging to the family: Moraceae is a native to Western Ghats of India, Malaysia and also found in central and Eastern Africa and many Pacific Islands. Various parts of the plant are reported to possess antidiabetic (Fernando, 1991), antibacterial (Khan, 2003), anti-inflammatory (Fang et al., 2008), antioxidant (Ko et al., 1998), antifungal (Trindade, 2006) and immune modulatory properties (Kabir, 1998). All the parts of the plant are used either as food or as medicine. Traditionally the decoction of the mature leaf petiole of jackfruit is used in various ailments. It possesses anti-inflammatory effect. Many studies have been reported on the leaves of the plant. But no attempt has been reported regarding the scientific studies on the leaf petiole of Artocarpus sps. Hence in the present study the estimation of the phytochemicals and antimicrobial activity of hexane, ethyl acetate and methanol extract of the leaf petiole of the A. heterophyllus has been done. The leaf petiole of the plant could be a valuable resource with many pharmacological activities especially as an antimicrobial agent.
Materials and methods
Collection of plant materials
Artocarpus heterophyllus Lam. leaves were collected from Mannanthala, Thiruvananthapuram, Kerala, India. The plant specimen was authenticated by Dr. G. Valsaladevi, Curator, Department of Botany, University of Kerala, Kariavattom, Thiruvananthapuram, Kerala. The voucher specimen has been deposited in the Herbarium of Department of Botany, University of Kerala with voucher no KUBH -6027 for future reference.
Preparation of the plant extracts
The petiole of mature leaves of A. heterophyllus was collected, washed and shade dried. The dried plant materials were grinded to fine powder using an electric grinder. The dried and powdered plant material was successively extracted with hexane (AHH), ethyl acetate (AHE) and methanol (AHM) in the order of their increasing polarity in soxhlet apparatus until it became colourless according to the standard methods (Harbone,1973).The extracts were concentrated using rotary vacuum evaporator (Buchi Rotavapor) and stored in refrigerator for further analysis.
Quantitative estimation of phytochemicals
Estimation of total phenolic content (TPC)
Total phenolic content of the extracts was measured using Folin-Ciocalteau spectrometric method (Singleton et al., 1999). Test samples of different concentration (10-500µg) dissolved in 1 % DMSO were mixed with 1 ml 95% ethanol. 0.25 ml of Folin-ciocalteau was added to each test tube. After 2 min, 0.75 ml of 20% sodium carbonate was added and the volume was made up to 5 ml with distilled water. The mixture was vortexed, left for 2h and the absorbance was measured at 700nm. A standard curve was plotted using 10-500µg of gallic acid in 95% ethanol. Total phenolic content was expressed as mg of gallic acid equivalent (GAE)/g of leaf extract.
Estimation of total flavonoid content
Flavanoid content was determined by aluminium–chloride colorimetric method (El Far & Taie, 2009). The method is based on the formation of the acid stable, flavanoid–aluminium complex. 0.5 ml of each extract in ethanol (10mg/ml) was mixed with 1.5ml methanol, 0.1 ml aluminium chloride (10%), 0.1ml potassium acetate solution and 2.8ml distilled water were added sequentially and mixed well. A blank sample was prepared in a similar way by replacing AlCl2 with distilled water. The test solution was vigorously shaken. Absorbance was recorded at 415 nm after 40min of incubation. A standard calibration plot was generated at 415nm using known concentrations of quercetin. The concentrations of flavonoid in the test samples were calculated from the calibration plot and expressed as mg quercetin equivalent/g of sample. All the determinations were carried out in triplicate.
Estimation of total alkaloids
The alkaloids estimation was performed by the spectrophotometric method of dragendorff’s reagent as it was described by Sreevidya and Mehrotra (2003). 10 mg amount of each crude extract was centrifuged over 10 min (3000 rpm) to remove residual suspended particles.0.5 ml extract was mixed with 1 ml of 0.1N HCl. 0.25 ml of dragendroff’s reagent was added and the precipitate formed was centrifuged. The precipitate was further washed with 0.25ml of ethanol. The filtrate was discarded and the residue was then treated with 0.25ml of 1 % disodium solution. The brownish black precipitate formed was the centrifuged (5 min, 3000 rpm). This residue was dissolved in 0.2ml of concentrated nitric acid and 0.1 ml was then pipette out and mixed with 0.5 ml of 3% thiourea solution. The absorbance was measured at 435 nm against a blank containing nitric acid and thiourea. A standard calibration curve was plotted using 20-100µg of caffeine. Total alkaloids were expressed as mg of caffeine equivalent/g of leaf extract.
Estimation of total carbohydrates
The total carbohydrates content was estimated by Hedge and Hofreiter (1962) anthrone method. The method is based on the formation of hydroxyl methyl furfural in an acidic medium which gives a green coloured product with anthrone having an absorption maximum at 630 nm. 10 mg of sample was hydrolyzed with 0.5 ml of 2.5 N HCl in a boiling water bath for three hours and cooled to room temperature. Neutralized it with sodium carbonate until the effervesces ceases. Made the volume to 10 ml and centrifuged. The supernatant was collected and 1 ml aliquot was taken for analysis. Standards were prepared by taking 0.2-1.0 ml of the working standard (glucose). 1 ml of water served as a blank. All the tubes are made up to 1.0ml with distilled water. 4.0ml of anthrone reagent was added, heated for eight minutes in a boiling water bath, cooled rapidly and read the absorbance at 630 nm. A standard curve was plotted using 20-100µg of glucose.
Gas Chromatography – Mass Spectroscopy (GC-MS) Analysis
Methanol extract of Artocarpus heterophyllus was analyzed for their chemical composition by using gas chromatography –mass spectrometry (GC 3800/ms Saturn 2200). A vent free (VF) -5 ms capillary column which uses GC liquid as injection mode was equipped with Turbo molecular pump (58.0 L/Sec for He). Helium was the carrier gas used which is having a flow rate 1 ml/mint. The injector and detector temperature were set at 250°C and 280°C respectively. The injections were performed in split less mode at 280° C. The oven temperature was programmed at 280°C. The mass spectrum of compounds in the samples was obtained by electron ionization at 70 eV. The total running time was 36 minutes. The extract was dissolved in methanol and filtered through a millipore syringe and were injected by the auto sampler into the capillary column through the GC injection port. The identity and quantity of the measured active compounds was correlated with that present in the database.
Identification of the phytoconstituents
The sample molecules were analyzed according to their mass-to-charge ratios. The chemical constituents of the components in the extract was determined by comparing the retention indices with their mass spectral fragmentation pattern of the known compounds stored in the National Institute of Standards and Technology (NIST) research library. The phytochemical compounds are identified based on peak area, molecular weight and molecular formula. The relative percentage amount of each component was calculated by comparing its average peak area to the total areas. The name, molecular weight and the structure of the components of the extracts were ascertained.
Selection of microbial strains
Five bacterial strains [Staphyllococcus aureus (ATCC 25923), Sreptococcus mutans (MTCC 890), Escherichia coli (ATCC 25922), Klebsiella pneumonia (ATCC 13883) and Pseudomonas aeroginosa (ATCC 27853)] and two fungi [Candida albicans (ATCC 10231), and Aspergillus niger (ATCC 16404)] were used. American type culture collection (ATCC) strains were purchased from Himedia while S. mutans was obtained from Institute of Microbial Technology (MTECH), Chandigarh.
Antibacterial activity
The antimicrobial activity of the different extracts of the plant was assayed by agar-well diffusion method as described in NCCLS, (1993). Petri plates containing 20ml nutrient agar medium was seeded with bacterial strains. Wells of approximately 10 mm was bored using a well cutter. Plant extracts were prepared in DMSO (stock: 1mg/ml DMSO). The plant extracts of 25, 50, and 100µl concentrations were added. Streptomycin (20µl) and DMSO (100µl) were used as positive and negative controls respectively. The plates were then incubated at 37 °C for 24 h. The antimicrobials present in the plant extract are allowed to diffuse out into the medium and interact with the test organisms in the freshly seeded plate. The diameter of the zone of inhibitions was measured in millimetres after 24h.
Antifungal activity
The potato dextrose agar plates were prepared and inoculated with a fungal culture. Wells of approximately 10mm was bored using a well cutter and samples of different concentration was added. The zone of inhibition was measured in millimetres after overnight incubation and compared with that of standard antimycotic (Clotrimazole) (10µl) which was used as positive control and DMSO (10%) as the negative control.
The percentage of inhibition (% I) was calculated by using the following formula:

The diameter of the test sample of 100 µl concentration of various plant extracts was taken for calculating the inhibition %.
Minimal inhibitory concentration (MIC)
Minimal inhibitory concentration (MIC) of the ethyl acetate and methanol extracts was determined by using two-fold serial dilution methods in 96-well plates against S. mutans, E. coli, P. aeroginosa, K. pneumoniae, C. albican and A. niger. Samples were dissolved in DMSO to a final concentration of 1 mg/ml and added in increasing concentration such as 6.25, 12.5, 25, 50, 100µg/ml respectively. Solvent control was prepared with DMSO (10%), and blank control was prepared from virgin media. The plates were prepared in triplicates and incubated overnight at 37 ˚C. Growth was observed by visual inspection and by measuring the optical density (OD) at 620 nm using microplate reader (ERBA, Germany, Model: ELISCAN FT3, 1x96 wells). The OD was measured immediately after the visual reading. The growth inhibition for the test wells at each plant extract dilution was determined by the formula:
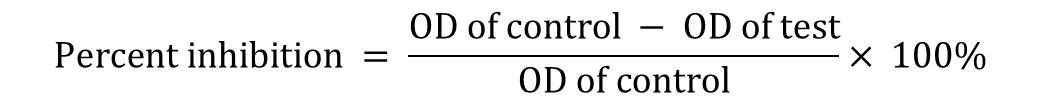
Spectrophotometric MICs were calculated based on the density of the growth control and the lowest drug concentrations that resulted in a 50% reduction in growth was compared with that of the drug free growth control (NCCLS, 1997).
Statistical analysis
Experimental results were expressed in mean±standard error mean (SEM) of the triplicates. Statistical analysis was performed by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test (DMRT) using SPSS software package (version 16). P value < 0.01 was considered as significant at 1% level.
Results
Phytochemical estimation
The results of the phytochemical analysis of various phytoconstituents of hexane, ethyl acetate and methanol extract of A. heterophyllus are given in the figure 1. The total phenolic content of AHH, AHE and AHM measured by folin ciocalteau reagent in terms of gallic acid equivalent is 7.13 ± 0.85, 16.1 ± 0.15, 24.23 ±1.04 mg/g respectively from the standard curve (y=0.0256x+0.475,R2+0.9031). Total flavonoid content of the plant extract was evaluated as quercetin equivalent from the standard curve (y=0.0001x+0.011; R2=0.988). Higher flavonoid content was observed for AHE and AHM as 3.7 ± 0.13 and 3.5 ± 0 .07mg/g of dry plant extract respectively. Total alkaloid content of AHH, AHE and AHM was evaluated as 2.34 ± 0.15, 2.34 ± 0.12, 3.55 ± 0.07 mg/g of dry plant extract respectively from the standard curve of caffeine (y=0.0113x+0.0324; R2=0.9978). Highest quantity of total carbohydrate content was in AHM (85.15 ± 0.45) which is followed by AHE (25.12 ± 0.62) and (2.02 ± 0.6) from the standard curve of glucose (y=0.0012+0.867, R2=0.9945).
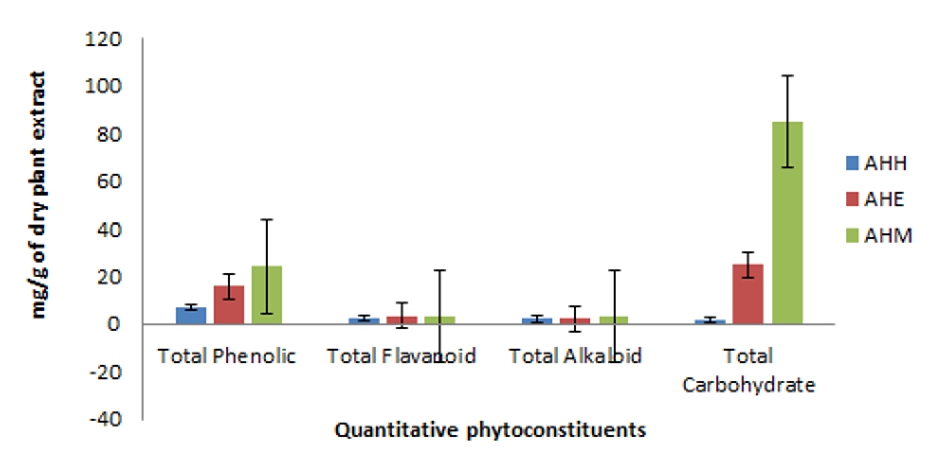
Figure 1. Phytochemical constituents of A. heterophyllus
GC-MS analysis
Identification of Components
GC-MS technique helps to identify the volatile matter, long chain, branced chain hydrocarbon, alcohols, acids, esters etc. present in the extract. The GC-MS analysis of the methanol extract of A. heterophyllus reveals the presence of twenty phytochemical constituents that could contribute to the medicinal property of the plant. The phytochemicals with their Retention time (RT), molecular formula, molecular weight and peak area in percentage are given in the table. 1. The chemical nature and the biological activity of the phytochemicals relevant to the present study with high peak area are listed in the table. 2. The biological activities listed are mainly based on Dr. Duke’s Phytochemical and Ethanobotanical Databases created by Dr. Jim Duke of the Agriculture Research Service/USDA. The chromatogram of the methanol extract of A. heterophyllus is given in the figure 2.
Table 1. Phyto- chemical components identified in methanol extract of A.heterophyllus by GC-MS
|
S. No. |
R. Time |
Name of the Compounds |
Molecular Formula |
Molecular Weight |
Area % |
|
1 |
13.34 |
Quinoline, 7-propyl- |
C12H13N |
171.24 |
0.49 |
|
2 |
13.73 |
o-Aminobenzohydroxamic acid |
C7H8N9 |
152.15 |
0.556 |
|
3 |
13.93 |
1,3-Benzodioxole, 2-methoxy- |
C8H8O3 |
152.14 |
1.25 |
|
4 |
14.31 |
N,N-Dimethyl-6-benzyloxyhexylamine |
C15H25NO |
235.36 |
1.74 |
|
5 |
14.46 |
6,9-Octadecadiynoic acid, methyl ester |
C19H30O2 |
290.44 |
0.051 |
|
6 |
14.62 |
10H-Dibenzo[b,E]thiopyran-10-one,2-ethy |
C13H9 |
195.21 |
0.325 |
|
7 |
14.80 |
1,4-Pentanediamine, N1,N1-diethyl- |
C9H22N2 |
158.28 |
0.049 |
|
8 |
14.91 |
Androst-5-ene-11,17-dione, 3- |
C19H26O2 |
286.41 |
0.11 |
|
9 |
15.259 |
Propane, 1-(2,2-dichloro-1,3,3-trimethyl |
C3H6Cl2 |
112.98 |
0.086 |
|
10 |
15.384 |
2-Methoxy-4-vinylphenol |
C9H10O2 |
150.18 |
1.784 |
|
11 |
15.66 |
Methyl N-acetylalanine, 3,3'-dithiobis- |
C6H11NO3 |
144.17 |
7.391 |
|
12 |
15.77 |
Oxazolidinecarboxylic acid, |
C4H7NO3 |
117.1 |
8.754 |
|
13 |
16.58 |
3-Buten-2-one, 1-(2,3,6-trimethylphenyl) |
C4H6O |
70.09 |
0.171 |
|
14 |
17.07 |
Coumarin, 3,4-dihydro-4,4,7-trimethyl- |
C12H14O2 |
190.24 |
0.252 |
|
15 |
17.86 |
9-Aminofluorene |
C13H11N |
181.23 |
1.086 |
|
16 |
18.05 |
Megastigmatrienone |
C13H18O |
190.28 |
0.411 |
|
17 |
18.44 |
Formic acid, 2-bromomethyl-4,4-dimethyl- |
C10H7CH2Br |
221.09 |
0.333 |
|
18 |
19.34 |
Phenol, 4-(3-hydroxy-1-propenyl)-2-metho |
C9H10O |
150.17 |
0.445 |
|
19 |
19.48 |
2,6-Piperidinedione, 4-[[2-butyl-4a,5,6, |
C9H15NO2 |
169.221 |
0.193 |
|
20 |
19.79 |
1-Methyl-3,5-diisopropoxybenzene |
C13H20O2 |
208.29 |
1.022 |
|
21 |
20.51 |
Pentadecanoic acid, 14-methyl-, methyl |
C17H34O2 |
270.45 |
0.213 |
|
22 |
20.82 |
Hexadecanoic acid, octadecyl ester |
C34H68O2 |
508.90 |
1.846 |
|
23 |
21.94 |
9-Octadecenoic acid (Z)-, 2-hydroxy-1- |
C18H34O2 |
282.46 |
0.793 |
|
24 |
22.63 |
Ethyl 5,8,11,14,17-icosapentaenoate |
C22H34O2 |
330.50 |
0.081 |
|
25 |
24.25 |
Bis (2-ethylhexyl) phthalate |
C24H38O2 |
390.55 |
4.025 |
|
26 |
31.36 |
gamma.-Sitosterol |
C29H50O |
414.70 |
0.058 |
Table 2. GC-MS analysis showing phytochemical compounds, their nature and their biological activities of the methanol extract of A.heterophyllus
|
S. No. |
Retention time |
Area % |
Name of compounds |
Compounds nature |
Activity |
|
1 |
15.77 |
8.754 |
1,3,Oxazolidinecarboxylic acid, |
Carboxylic acid |
Acidifier |
|
2 |
15.66 |
7.391 |
Methyl N-acetylalanine, 3,3'-dithiobis- |
Dimethyl ester |
Anticancer, NO-Scavenger, Neuroprotector |
|
3 |
24.25 |
4.025 |
Bis (2-ethylhexyl) phthalate |
Organic compound |
Plasticizer |
|
4 |
20.82 |
1.846 |
Hexadecanoic acid, octadecyl ester |
Palmitic acid ester |
Antioxidant, hypocholesterolemia, nematicide, pesticide, antiandrogenic,,hemolytic, Acidifier |
|
5 |
15.384 |
1.784 |
2-Methoxy-4-vinylphenol |
Aromatic substance |
Flavoring agent |
|
6 |
14.31 |
1.74 |
N,N-Dimethyl-6-benzyloxyhexylamine |
Aliphatic amines |
Antitumor, Neuritoxic, Neurostimulant, |
|
7 |
13.93 |
1.25 |
1,3-Benzodioxole, 2-methoxy- |
Ether compound |
No activity reported |
|
8 |
17.86 |
1.086 |
9-Aminofluorene |
Nitrogen compound |
Insecticide, Psychotic effect, Neurotoxic effect |
|
9 |
19.79 |
1.022 |
1-Methyl-3,5-diisopropoxybenzene |
Benzene compound |
Antibacterial, Antiviral, Antiseptic |
|
10 |
21.94 |
0.793 |
9-Octadecenoic acid (Z)-, 2-hydroxy-1- |
Monoglyceride |
Antimicrobial, Anticancer, Diuretic, Anti- inflammatory, Acidifier |
|
11 |
13.73 |
0.556 |
o-Aminobenzohydroxamic acid |
Anhydride |
Anticancer, Osmoregulator, Osteolcalcin stimulant. |
|
12 |
13.34 |
0.49 |
Quinoline, 7-propyl- |
Alkaloid |
Antibacterial agent |
|
13 |
19.34 |
0.445 |
Phenol, 4-(3-hydroxy-1-propenyl)-2-metho |
Phenolic compound |
Testosterone –hydroxylase -Inducer |
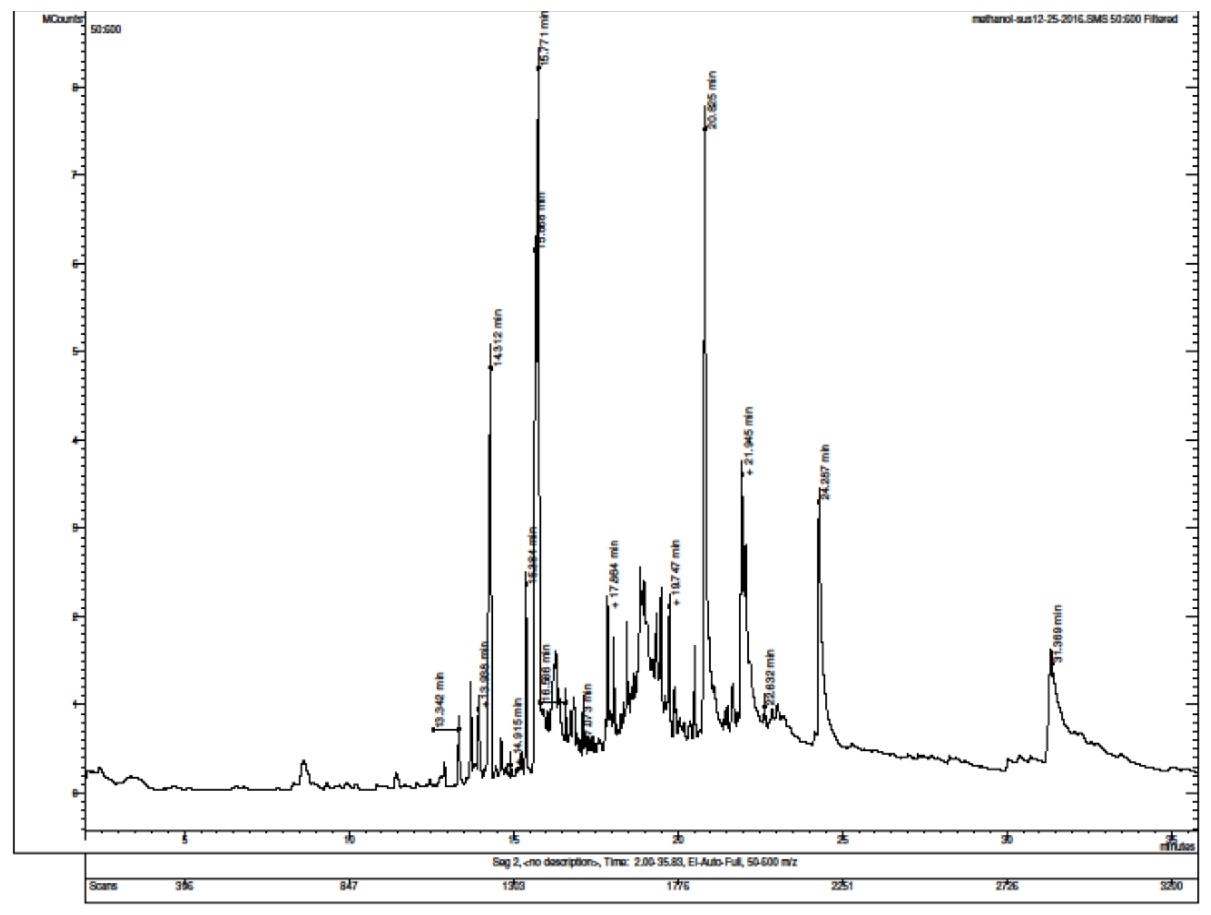 Figure 2. Chromatogram of GC-MS Analysis of methanol extracts of Artocarpus heterophyllus
Figure 2. Chromatogram of GC-MS Analysis of methanol extracts of Artocarpus heterophyllus
Antimicrobial activity
The in vitro antimicrobial activity of the different extracts of A. heterophyllus under different concentration with the standard are given in the table 3. At 25µg/ml concentration of the plant extracts, no antimicrobial activity was shown. At 50µg/ml and 100µg/ml, the methanol extract of the plant showed antimicrobial activity against most of the test organisms. While ethyl acetate extract (AHE) showed activity at 100µg/ml concentration. While the hexane extract showed the least activity.
Table 3. Antimicrobial activity of Artocarpus heterophyllus extracts using disc diffusion assay
|
Microbial Strains |
Diameter of inhibition zone (mm) |
||
|
Escherichia coli (EC) |
50µg/ml |
100µg/ml |
Streptomycin (10µg/ml) |
|
Hexane |
Nil |
10.97±0.33 |
29.1± 0.12 |
|
Ethyl acetate |
Nil |
11.97±0.033 |
|
|
Methanol |
Nil |
13.00±0.00 |
|
|
Klebsiella pneumonia (KP) |
|||
|
Hexane |
Nil |
11.97±0.033 |
32.2± 0.2 |
|
Ethyl acetate |
10.03 ±0.03 |
10.97±0.033 |
|
|
Methanol |
11.10± 0.05 |
16.00±0.05 |
|
|
Pseudomonas aeroginosa (PA) |
|||
|
Hexane |
Nil |
13.83±0.16 |
34.2± 0.0.2 |
|
Ethyl acetate |
Nil |
10.1± 0.05 |
|
|
Methanol |
12.0±0.00 |
13.83±0.16 |
|
|
Staphyllococcus aureus (SA) |
|||
|
Hexane |
Nil |
11.00±0.00 |
30.12±0.01 |
|
Ethyl acetate |
Nil |
10.00±0.00 |
|
|
Methanol |
10.00± 0.00 |
11.83±0.16 |
|
|
Sreptococcus mutans (SM) |
|||
|
Hexane |
Nil |
Nil |
33.12± 0.01 |
|
Ethyl acetate |
Nil |
12.17±0.12 |
|
|
Methanol |
Nil |
12.17± 0.12 |
|
|
Candida albicans (CA) Clotrimazole(10µg/ml) |
|||
|
Hexane |
Nil |
Nil |
13.1±0.04 |
|
Ethyl acetate |
Nil |
11.00±0.00 |
|
|
Methanol |
10.1±0.58 |
12.00±0.00 |
|
|
Aspergillus niger (AN) |
|||
|
Hexane |
Nil |
Nil |
13.1±0.04 |
|
Ethyl acetate |
Nil |
Nil |
|
|
Methanol |
10.0±0.00 |
13.20± 0.153 |
|
Values are mean inhibition zone (mm) ± SE of three replicates.
Table 4. Comparison of antimicrobial activity of different extracts (100µg/ml) of Artocarpus heterophyllus with respect to various microbial strains (mean±SE)(n=3).
|
Microbial Strains |
Diameter of inhibition zone(mm) |
||||
|
AHH |
AHE |
AHM |
F |
P |
|
|
Escherichia coli (EC) |
10.97±0.033a |
11.97± 0.033b |
1 3.00±0.00c |
1395.50 |
0.000** |
|
Klebsiella pneumonia (KP) |
11.97±0.033a |
10.97±0.033b |
16.00±0.05c |
3834.200 |
0.000** |
|
Pseudomonas aeroginosa (PA) |
13.83±0.16a |
10.10±0.05b |
13.83±0.16a |
236.679 |
0.000** |
|
Staphyllococcus aureus (SA) |
11.00±0.00a |
10.00±0.00b |
11.83±0.16c |
91.000 |
0.000** |
|
Sreptococcus mutans (SM) |
Nil |
12.17±0.12a |
12.17±0.12a |
5124.038 |
0.000** |
|
Candida albicans (CA) |
Nil |
11.00±0.00a |
12.00±0.00b |
- |
0.00 |
|
Aspergillus niger (AN) |
Nil |
Nil |
13.20±0.153a |
7467.429 |
0.000** |
Statistical analysis: Test of significance (ANOVA followed by Duncan’s Test). **Significant at 1% level (p<0.01) [Values are mean±SE of triplicates; Mean ± SE with different alphabets denote significant pair-wise difference by Duncan’s Test]
ANOVA showed that there exist significant mean differences in antimicrobial activities among various extracts at 100 µg/ml concentration (P>0.01) (Table 4). Duncun’s test showed the maximum inhibition zone for methanol extract against E. coli (13.00 ±0.00), K. pneumonia (16.00±0.05), C. albicans (12.00±0.00) and A. niger (13.20+-±0.153) when compared to ethyl acetate (AHE) and hexane (AHH) extracts. Inhibition zone of the extract AHH and AHM showed similar reaction and higher zone of inhibition (12.17 ±0.12) against S. mutans. Only the methanol extract is having an inhibitory effect against the A. niger which is having a zone of inhibition as 13.2±0.153.
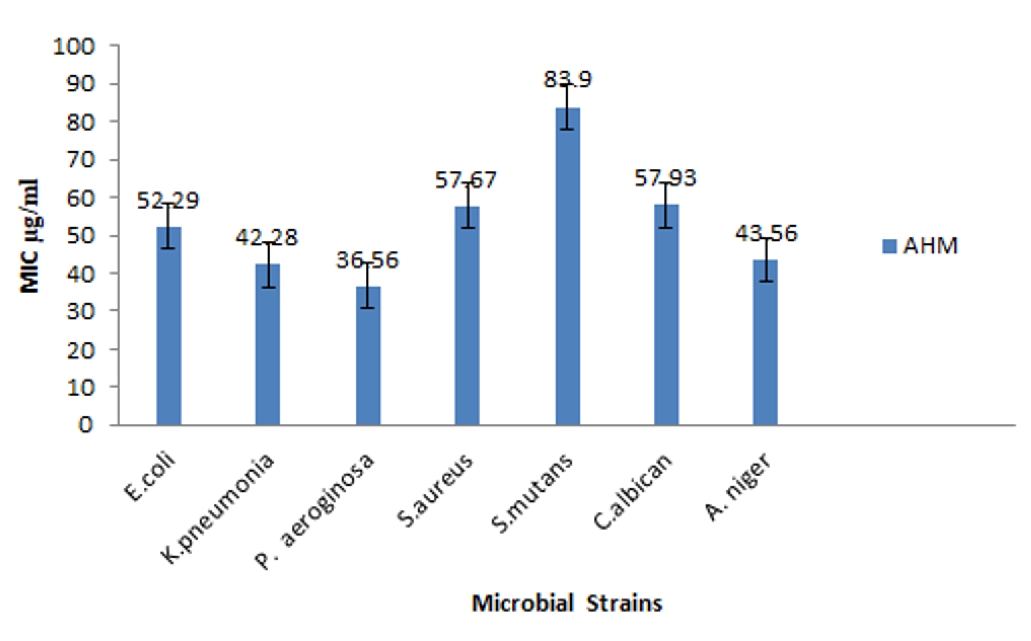
Figure 3. Minimum Inhibitory Concentration (MIC) of methanol extracts of Artocarpus heterophyllus (AHM) against microbial organisms
The MIC of the methanol extract against the microbial strains is shown in the figure 3. The MIC value of the extract of A. heterophyllus against P. aeroginosa was the lowest ie. 36.56µg/ml while the highest value for S. mutan (83.9 µg/ml). The MIC value against E. coli, K. pneumonia, and S. aureus were 52.29, 42.28 and 57.67µg/ml respectively. The MIC value against the fungal strains, C. albican and A. niger showed 57.93 and 43.56µg/ml.
Discussion
The present study evaluates the phytochemical composition of the different extracts of A. heterophyllus and their antimicrobial activity. Literature studies reveal that jackfruit contains useful compounds like flavonoids, sterols and prenylfavones which may have various pharmacological values and clinical studies have shown the hypoglycemic effect of the decoction of the leaves (Baliga et al., 2011). The preliminary phytochemical screening of the leaves of A. heterophyllus showed the presence of flavonoids and steroids
(Saxena et al., 2016).
In the present study, the highest concentration of phenols, flavonoids, alkaloids were seen in methanol extract of leaf petiole of A. heterophyllus followed by ethyl acetate and hexane extracts. Studies report that the presence of the major phytochemicals is present in the polar solvents (Kumoroa et al., 2009).
Of the twenty compounds identified, many of them possess various biological properties. Methyl N-acetylalanine, 3, 3-dithiobis (R/T 15.66) possesses anticancer, nitric oxide scavenging and neuroprotective properties. Hexadecanoic acid, octadecyl ester (R/T 20.82) possess antioxidant, hypocholesteolemia, nematicide, pesticide, antiandrogenic and haemolytic properties (Hussein et al., 2016). Hydroxamic acid and its derivatives act as inhibitors of melanocyte tyrosinase for tropical skin lighteners (Dooley et al., 2003).
Antimicrobial activity against the five bacterial strains showed that the methanol extract of the leaf petiole of A. heterophyllus possess better activity. Khan et al. (2003), reported a significant activity of the A. heterophyllus root bark and fruits n-butanol fractions against the bacterial strains. Loizzo et al. (2010) reported the antibacterial activity of total water extract, ethyl acetate and aqueous fractions from the leaves of A. heterophyllus. Antimicrobial activity of the plant extract was considered to be good if its MIC was less than 100µg/ml, moderate if MIC was from 100.0 to 500.0µg/ml and poor over 500.0µg/ml (Dabur et al., 2007). Since the MIC of the methanol extract of the plant against all the microbial strains were less than 100µg/ml, the methanol extract can be considered as a good quality. This antimicrobial activity may be due to some phenolic compounds in the extracts which were present in high concentration in methanol extract. Hence further elucidation of bioactive compounds is required.
Conclusion
The present study revealed significant antimicrobial activity of the hexane, ethyl acetate and methanol extract of the leaf petiole of Artocarpus heterophyllus when compared with standards. The best activity was observed in the methanol extract. However, further studies are required to evaluate the biological activity of the plant against various diseases.
Acknowledgement
The financial support from the University Grand Commission in the form of Teacher Fellowship under the Faculty Development Programme (No.FIP/12th Plan/KLKE013 TF- 08 dated 09/02/2016) to the first author is gratefully acknowledged.
Conflicts of Interests
None declared.
References
Baliga MS, Shivashankara AR, Haniadka R, D souza, J, Bhat HP. 2011. Phytochemistry, nutritional and pharmacological properties of Artocarpus heterophyllus Lam (jackfruit): A review. Food Research International, 44(7):1800-1811.
Dabur R, Gupta A, Mandal TK, Singh DD, Vivek B, Gurav AM. 2007. Antimicrobial activity of some Indian medicinal plants. African Journal of Traditional, Complementary and Alternative Medicines, 4:313–318.
De Fátima A, Modolo LV, Conegero LS, Pilli RA, Ferreira CV, Kohn LK, De Carvalho JE. 2006. Styryl lactones and their derivatives: biological activities, mechanisms of action and potential leads for drug design. Current Medicinal Chemistry, 13(28): 3371-3384.
Dooley T, Cheng L. 2003. Inventors; Integri Derm Inc, assignee. Hydroxamic acid and its derivatives as inhibitors of melanocyte tyrosinase for topical skin lighteners. United States patent application US 10/328:404.
El Far MM, Taie HA. 2009. Antioxidant activities, total anthocyanins, phenolics and flavonoids contents of some sweet potato genotypes under stress of different concentrations of sucrose and sorbitol. Australian Journal of Basic and Applied Sciences, 3(4): 3609-3616.
Fang SC, Hsu CL, Yen GC. 2008. Anti-inflammatory effects of phenolic compounds isolated from the fruits of Artocarpus heterophyllus. Journal of Agricultural and Food Chemistry, 56(12):4463-4468.
Fernando MR, Wickrama singhe SN, Thabrew MI, Ariyananda PL, Karunanayake EH. 1991. Effect of Artocarpus heterophyllus and Asteracanthus longifolia on glucose tolerance in normal human subjects and in maturity-onset diabetic patients. Journal of Ethnopharmacology, 31(3): 277-282.
Harborne JB. 1973. Phytochemical methods. Chapman and Hall, Ltd., London: New York. 49-188.
Hasan P, Yasa N, Ghanbari SV, Mohammadirad AG, Dehghan Abdollahi M. 2007 . In vitro antioxidant potential of Teucrium rolium, as compared to a-tocopherol.Acta Pharmaceutica, 57:123–129.
Hedge JE, Hofreiter BT, Whistler RL. 1962. Carbohydrate chemistry. Academic Press, New York. 17.
Hussein HJ, Hadi MY, Hameed IH. 2016. Study of chemical composition of Foeniculum vulgare using Fourier transform infrared spectrophotometer and gas chromatography-mass spectrometry. Journal of Pharmacognosy and Phytotherapy, 31, 8(3):60-89.
Kabir S. 1998. Jacalin: a jackfruit (Artocarpus heterophyllus) seed-derived lectin of versatile applications in immunobiological research1. Journal of Immunological Methods, 212(2): 193-211.
Khan MR, Omoloso AD, Kihara M. 2003. Antibacterial activity of Artocarpus heterophyllus. Fitoterapia, 74(5): 501-505.
Ko FN, Cheng ZJ, Lin CN, Teng CM. 1998. Scavenger and antioxidant properties of prenylflavones isolated from Artocarpus heterophyllus. Free Radical Biology and Medicine, 25(2):160-168.
Kumoroa AC, Hasana M, Singh H. 2009. Effect of solvent properties on the soxhlet extraction of diterpenoid lactones from Andrographis paniculata leaves. Science Asia, 35:306-309.
Loizzo MR, Tundis R, ChandrikaUG, Abeysekera AM, Menichini F, Frega NG. 2010. Antioxidant and antibacterial activities on foodborne pathogens of Artocarpus heterophyllus Lam. (Moraceae) leaves extracts. Journal of Food Science, 75(5).
National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Disk Susceptibility Tests-Fifth Edition: Approved Standard M2-A5. NCCLS, Villanova, Pa: 1993a.
National Committee for Clinical Laboratory Standards. Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved standard. Document M27-A. National Committee for Clinical Laboratory Standards, Wayne, Pa; 1997.
Saxena K, Irchhaiya R, Chagti KK. 2016. Antihepatotoxic effect of Artocarpus heterophyllus Leaves against paracetamol induced hepatic damage in albino rats. International Journal of Pharmacy & Life Sciences, 7(2): 4895-4899.
SingletonVL, Orthofer R. Lamuela-Raventós RM. 1999. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in enzymology, Academic press, 99:152-178.
Sreevidya N, Mehrotra, S. 2003. Spectrophotometric method for estimation of alkaloids precipitable with Dragendorff's reagent in plant materials. Journal of AOAC international, 86(6): 1124-1127.
Trindade MB, Lopes JL.2006. AS Costa; AC Moreira, RA Moreira, ML Oliva; LM Beltramini.Biochimica et Biophysica Acta. 1:146-152.
Vital PG, Rivera WL. 2009. Antimicrobial activity and cytotoxicity of Chromolaena odorata (L.f.) King and Robinson and Uncaria perrottetii (A. Rich) Merr extracts. Journal of Medicinal Plants Research, 3(7): 511-518.