

Suchit Jain1, Sharad Bhandarkar2
1School of Pharmaceutical Sciences, SOA University, Bhubaneswar, Odisha, India
2Institute of Pharmacy, Abdul Majeed Central Education Society, Nagpur, Maharashtra, India
*Corresponding address
Sharad Bhandarkar
Institute of Pharmacy, Abdul Majeed Central Education Society,
Nagpur, Maharashtra, India.
Abstract
Objective: Aim of present study was study gastroprotective activity of ethanol extract of Martynia annua leaves in rats. Materials and methods: Ethanol extract of M. annua was selected for study with 200 mg/kg and 300 mg/kg b.w. doses of extract on ethanol induced gastric ulcer. Effect of extract was observed by calculating ulcer index based on lesion index and pH. Results: Phytochemical screening confirmed the presence of flavonoids, glycosides and terpenoids in leaves of M. annua. Ethanol extract with both doses (200 mg/kg and 300 mg/kg) produced significant inhibition on the ulcer lesion index in rats. Effect of ethanol extract with 300 mg/kg dose significantly (p<0.05) change the gastric volume, ulcer index and pH. Conclusion: The results of the study indicated that significant inhibition of gastric lesions was found. The results of macroscopic observation also supported the gastroprotective effect and may be associated with its antioxidant property of flavonoids.
Keywords: Martynia annua, gastroprotective, flavonoids, antioxidant, omeprazole
Introduction
Peptic ulcer is considered a modern age epidemics, produced due to an imbalance between acid and pepsin along with the limitations of the mucosal barrier (Shah et al., 2006). It is commonly connected with injure of the stomach's mucosal layer, that is simply caused via excess production of exogenous and endogenous oxygen free radicals. Some of the main causes of gastric ulcers include continual use of alcoholic beverages and anti-inflammatory drugs, long time stress and Helicobacter pylori infection (Barocelli et al., 1997).
Martynia annua L. (Martyniaccae), is commonly known as Bichchhu. It is commonly used in epilepsy and applied locally to tuberculosis glands of camel’s neck. The juice of leaves used as a gargle for sore throat and fruits were used in inflammation. The leaves paste has beneficial effect topically on wounds of domestic animal (Manandhar and Manandhar, 2002). Phytochemical examination of M. annua plant revealed the presence of glycosides, tannins, carbohydrates, flavonoids and glycosides. The leaves reported to has chlorogenic acid, and seeds contain 10.35% of semidrying oil and fatty acids (palmitic acid, stearic acid, oleic acid and arachidic acid) (Kiritikar and Basu, 1999). Flowers are reported cyanidin-3-galactoside and p-hydroxy benzoic acid, luteolin and gentisic acid are present in leaves (Khare, 2007). Methanolic extract of M. annua has wound healing (Lodhi and Singhai, 2013) and antioxidant activity. The anti-inflammatory activity and antifertility activity of ethanol extract of Martynia diandra was reported in both acute and subacute inflammatory process (Ashwani et al., 2013; Mali et al., 2002).
Materials and Methods
Plant material and phytochemical studies
M. annua leaves were collected from the campus of Institute of Pharmacy, Abdul Majeed Central Education Society, Nagpur, India. The plant material was identified and a herbarium was submitted to Department of Botany, JNKV, Jabalpur (M.P., India). The plant material was dried in shade, powdered and stored for extract with ethyl alcohol. The powdered leaves were subjected to extraction with petroleum ether for defatting and defatted materials were extracted with ethyl alcohol in a soxhlet apparatus at 70-80 0C up to complete extraction (Lodhi et al., 2010). The ethanol extract was concentrated and dried under reduced pressure and yield was calculated. Extract was investigated for qualitative chemical test for different chemical constituents.
Ethanol induced ulcer
Albino rats (150-160g) of either sex were selected for antiulcer activity. They were housed and divided into six groups containing each group of six animals in polypropylene cages, under standard laboratory conditions of temperature (25 ± 2oC). The animals had free access to standard food (Brooke Bond-Lipton, India) and water. The animals were acclimatized for minimum 7 days before experiment. All experimental protocol was approved by the Institutional Animals Ethics Committee and care of animals was taken as per CPCSEA guidelines. The control group was given only vehicle (sodium carboxymethyl cellulose, 0.5 % p.o.), test group II and II were given ethanol extract (200 and 300 mg/kg, p.o.), respectively. Standard group IV was given Omeprazole (20 mg/kg, p.o.).
Rats were fasted for 48 hrs prior receiving different doses of treatment with ethanol extract (200 mg/kg and 300 mg/kg) and Omeprazole (20 mg/kg) in respective group of animals. The ulcer was induced using 1 ml of 80 % ethyl alcohol by administered orally to each animal. After 1 hr the rats were anesthetized using diethyl ether for anesthesia and die by cervical dislocation. The macroscopic observation of stomach was recorded for ulcer scoring after removal of stomach. Whole stomachs were gently rinsed with water to remove the gastric contents and blood clots and examined for lesions in the four stomach portion for indexed according to severity (Jhariya et al., 2015). Mean ulcer score for each animal was expressed as ulcer index.
Ulcer Index (UI) = 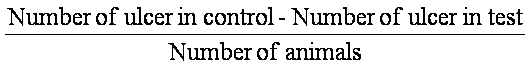
Percent Inhibition (% I) = 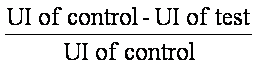 X100
X100
Gasric juice from each stomach were collected and measured. The pH of the gastric secretion was recorded by a pH meter and determines total acidity of the gastric juice by titration with 0.01 N NaOH and phenolphthalein as indicator. The total acidity is expressed as mEq/l using the following formula:
Total/free acidity = n×0.01×40×1000
Pharmacological data were represented as the mean ±S.D. for six rats. All data were statistically evaluated using the Tukey test. Values of P <0.05 were considered to be statistically significant.
Results and discussion
Practical yield of ethanol extract from leaves of M. annua was found 8.58 w/w%. Phytochemical screening results the presence of flavonoids, terpenoids, tannins and glycosides in ethanol extract. The ethanol extract was reported safe dose up to a dose of 1000 mg/kg body weight (Lodhi et al., 2013). The extract dose of 200 and 300 mg/kg p.o. was selected for the gastroprotective study.
Ethanol induced ulcer
Ethanol administration resulted in the production of gastric mucosal damage. The ulcer index in control animals was found 19.54±1.84. Ethanol extract (300 mg/kg) was showed 57.83± 4.25 inhibition of the ulcer index and significantly reduced ulcer index. The reduction in ulcer index by alcoholic extracts (200 mg/kg) is 44.01%. Omeprazole, a standard anti-ulcer drug was showed 62.74 % ulcer index inhibition (Table 1).
Table 1. Effect of ethanol extract of M. annua leaves on ulcer index in ethanol induced model
|
Treatment groups |
Doses |
Ulcer Index |
% Inhibition |
|
Control |
5 ml/kg |
19.54±1.84 |
- |
|
Omeprazole |
20mg/kg |
7.28±0.65* |
62.74± 5.28 |
|
EEMA (200 mg/kg) |
200 mg/kg |
10.94± 0.93 |
44.01± 3.84 |
|
EEMA (300 mg/kg) |
300 mg/kg |
8.24± 0.82* |
57.83± 4.25 |
EEMA: Ethanol extract of M. annua; Values are mean ± SD; N = 6 in each group *P < 0.05, when experimental groups compared with control
Table 2. Effect of ethanol extract of M. annua leaves on ulcer parameters in ethanol induced model in rats
|
Treatment groups |
Dose |
Gastric volume (ml) |
pH |
Total acidity (mEq/l) |
|
Control |
5 ml / kg |
5.30±0.57 |
1.5±0.24 |
752.6±71.65 |
|
Omeprazole |
20 mg/kg |
2.58±0.27* |
4.2±0.62* |
326.5±28.36* |
|
EEMA (200 mg/kg) |
200 mg/kg |
3.25±0.56 |
3.2±0.12 |
416.4±50.24 |
|
EEMA (300 mg/kg) |
300mg/kg |
4.62±0.65* |
4.5±0.25* |
384.2±54.27* |
EEMA: Ethanol extract of M. annua; Values are mean ± SD; N = 6 in each group *P < 0.05, when experimental groups compared with control
Gastric total acidity in control group of animals was found 752.6±71.65. Ethanol extract (300 mg/kg) was showed significant decrease in total acidity 384.2±54.27 mEq/l, compared to control group (Table 2).
The control group of animals showed necrosis and hemorrhage in gastric lesions were predominant over vast surface area, perforations with complete mucosal destruction. The animal treated with 300 mg/kg extract and standard drug were observed that the stomach section not found any necrosis and hemorrhage that reflects the protective effect of extract and drug (Figure 1).
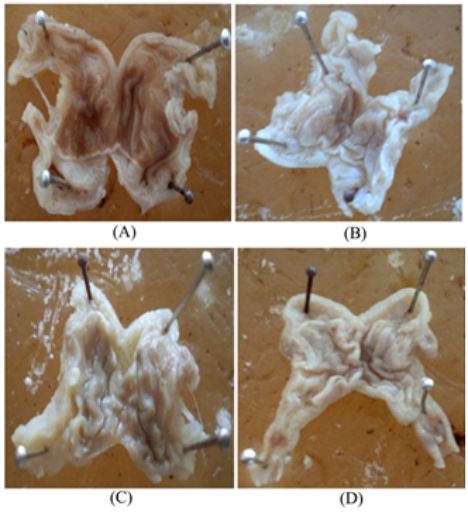
Figure 1. Observations of ulcers in ethanol induced model
(A) Control group; (B) Omeprazole Standard group; (C) Ethanol extract of M. annua (200 mg/kg) treated group; (D) Ethanol extract of M. annua (300 mg/kg) treated group
The anti-ulcer activity was tested against gastric lesions induced by ethanol, the experimental models related to lesion pathogenesis with production of reactive species. The plant extract was also found to increase the pH and decrease the acid volume and total acidity of gastric fluid. Alcohol rapidly penetrates the gastric mucosa which can cause cell membrane damage and leading to increased intra cellular membrane permeability to sodium and water. The enormous intracellular gathering of calcium represents a major step in the induction of gastric mucosal injury. This may support to cell death and exfoliation of epithelium surface (Raju, 2009). The antioxidant effects of M. annua extract was already reported that can support for gastroprotective effect of M annua. Some oxygen free radicals and causing agents have been implicated in the pathogenesis of ethanol-induced gastric ulcers (Shardul and Gangadhar, 2010).
Reactive oxygen species (ROS) are generated through numerous normal metabolic processes and also required for normal functioning of living organism. Different antioxidant enzymes like superoxide dismutase (SOD), gluthathione peroxidase (GPX) and catalase (CAT) are present in different tissues may control their accumulation (Fridovich, 1986). Any imbalance in the activity of these enzymes normally leads to faulty disposal of free radicals. These ROS are responsible for oxidation of tissues leading to lipid peroxidation and tissue damage. So, the antioxidants seemed to have protective role in gastric ulcers (Ito et al., 1996) The possible mechanism involves the antioxidant properties of flavonoids present in ethanol extract, which at a dose of 300 mg/kg has a protective effect against lesions induced by 50 % ethanol, probably by reducing the levels of lipoperoxides and increasing the activity of the antioxidant enzyme glutathione peroxidase. In conclusion the ethanol extract of M. annua protect the mucosa layer of stomach may be due to its flavonoid constituents. Thus, this finding confirms the traditional use of M. annua in the treatment of gastric ulcer.
Acknowledgements
The authors would like to acknowledge to Department of Botany, JNKV, Jabalpur (M.P., India) for identification of plant material.
Conflict of interest
The authors report no conflicts of interest.
References
Ashwani KD, Bhawna C, Sanjeev KM. 2013. Martynia annua L. ; A Review on its Ethnobotany, Phytochemical and Pharmacological Profile. Journal of Pharmacognosy and Phytochemistry, 1(6):135-140.
Barocelli E, Chiavarini M, Ballabeni V, Barlocco D, Vianello P, Dal Piaz V, Impicciatore M. 1997. Study of the antisecretory and antiulcer mechanism of new indenopyridazinone derivative in rats. Pharmacological Research, 35: 487-492.
Fridovich I. 1986. Biological effects of superoxide radical. Archives of Biochemistry and Biophysics, 247: 1–11.
Ito N, Hirose M, Imaida K. 1996. Antioxidants: Carcinogenic and hemopreventive properties. In: Bertino JR, editor. Encyclopedia of Cancer, Vol. 1, California, Academic Press, pp. 51–63.
Jhariya S, Rai G, Yadav AK, Jain AP, Lodhi S. 2015. Protective effects of Tridax procumbens Linn. leaves on experimentally induced gastric ulcers in rats. Journal of Herbs, Spices & Medicinal Plants, 21:308–320.
Khare CP. 2007. Indian medicinal plants: an illustrated dictionary. Springer-Verlag Heidelberg, pp 399-400.
Kiritikar KR, Basu BD. 1999. Indian Medicinal Plants; International book distributors booksellers and Publisher, Vol-3, Dehradun, pp 749.
Lodhi S, Pawar RS, Jain AP, Jain A, Singhai AK. 2010. Effect of Tephrosia purpurea (L) Pers. on partial thickness and full thickness burn wounds in rats. Journal of Complementary and Integrative Medicine, 7(1): 13.
Lodhi S, Singhai AK. 2013. Wound healing effect of flavonoid rich fraction and luteolin isolated from Martynia annua Linn. on streptozotocin induced diabetic rats. Asian Pacific Journal of Tropical Medicine, 6 (4): 253-259.
Lodhi S, Singhai AK. 2013. Wound healing effect of flavonoid rich fraction and Luteolin isolated from Martynia annua Linn on Streptozotocin induced diabetic rats. Asian Pacific Journal of Tropical Medicine. 6(4):253-9.
Mali PC, Ansari AS, Chaturvedi M. 2002. Antifertility effect of chronically administered Martynia annua root extract on male rats. Journal of Ethnopharmacology, 82(2-3): 61-67.
Manandhar NP, Manandhar S. 2002. Plants and people of Nepal. Portland: Timber Press, Inc; pp 311.
Raju D. 2009. Evaluation of Anti-ulcer activity of methanolic extract of Terminalia chebula fruits in experimental rats. Journal of Pharmaceutical Sciences and Research, 3:101‐107.
Shah DI, Santani DD, Goswami SS. 2006. A novel use of methylene blue as a pharmacological tool. Journal of Pharmacological and Toxicological Methods, 54(3): 273–277.
Shardul SW, Gangadhar BS. 2010. Antioxidant and hepatoprotective activity of Tridax procumbens Linn, against paracetamol induced hepatotoxicity in male albino rats. Advanced Studies in Biology, 2(3): 105–112.