

Km. Saba Khan*, Shamim Ahmad, Anuj Kumar Sharma
Translam Institute of Pharmaceutical Education and Research, Meerut, Uttar Pradesh, India
*Address for corresponding author
Km. Saba Khan
Translam Institute of Pharmaceutical Education and Research, Meerut, Uttar Pradesh, India
Email: sabakhan102@gmail.com, Phone: +91 7417568853
Abstract
Objective: The present work was under taken to evaluate the anti-obesity effect of Rubia cordifolia leaves extract in Cafeteria diet induced obesity in rats. Materials and methods: Wistar rats weighing 150-180 g were randomly divided in to seven groups (n=6/group). Group І served as normal control & received vehicle; Group ІІ served as CD control & received vehicle; Group ІІІ received standard drug Orlistat (30 mg/kg); Groups ІV & V were administered with ethanolic extract of Rubia cordifolia leaves (250 & 500 mg/kg respectively) and Groups VІ & VІІ received aqueous extract of Rubia cordifolia leaves (250 & 500 mg/kg respectively). All treatments were given orally, started after 6 weeks feeding with CD (except normal control group) and continued for six weeks along with CD. Weight gain, feed intake, BMI, obesity index, lipid profile, blood glucose and body fat depots content were the parameters evaluated. Results: Ethanolic and aqueous extract of Rubia cordifolia leaves at 250 and 500 mg/kg showed reduction in weight gain, feed intake, BMI (Basal metaboilic rate, obesity index and significant decrease in serum glucose, triglyceride, and total cholesterol with increase in HDL (High density lipoprotein), level. Also these were significant decrease body fat depots when compared to CD control group. Conclusion: Present study revealed that aqueous and ethanolic extracts of Rubia cordifolia leaves extracts possess significant anti-obesity activity against Cafeteria diet induced obesity in rats and that could be due to presence of flavonoids, tannins, diosgenin and saponins. However, detailed phyto-pharmacological study of leaves extracts will be useful to pin point the activity.
Keywords: Rubia cordifolia, anti-obesity, Cafeteria, obesity index, lipid profile
Introduction
On a world scale, obesity has reached epidemic proportion and is a major contributor to the world burden of chronic disease and disability. Currently, more than one billion adults worldwide are overweight and at least 300 million of them are clinically obese. According to WHO, obesity is related to cardiovascular disease, hypertension, endocrine disorders, diabetes mellitus, gallbladder disease, cancer, metabolic disturbances, osteoarthritis, gout & pulmonary disease, as well as psychological issues, including social bias, prejudice, discrimination, and overeating. Economically, obesity and its health consequences place enormous costs now and for future health care. Being overweight is a cosmetic issue, a major health risk factor, and may decrease life expectancy.
Many anti-obesity drugs are available in the market. However, they exhibit side effects, such as gastrointestinal and kidney problem, only orlistat and sibutramine can be used long- term, in spite of issues regarding weight loss and tolerance (Kang et al., 2012). Basic health science offers the promise of improving our understanding of disease pathophysiology, identifying novel mechanism that can be targeted by more effective pharmacotherapies and screening of herbal sources of drugs (Katiyar et al., 2012).
These considerations implicate the search for new anti-obesity agents that have a fast onset of action, with less side effects and a wider safety margin. Natural ingredients and medicinal plant preparation may improve metabolism, and speed up weight loss. Including these foods in the diet may therefore assist slow, individual weight loss. Despite the global market for satiety, fat burning, dietary supplements and other weight management remedies, patient awareness of these products is insufficient (Kazemipoor et al., 2012).
Various plants are being used in complementary and alternative medicines for management of obesity. Attention is being focused on the investigation of the efficacy of plant-based drugs used in the traditional medicinal system because they are cheap, have little side effects and according to WHO, about 80% of the world population still rely mainly on herbal remedies. The present work was under taken to evaluate the anti-obesity effect of Rubia cordifolia leaves extract in Cafeteria diet induced obesity in rats.
Materials and methods
Chemicals and reagents
Ethanol (SD fine Chemicals Limited, India), Serum cholesterol (Choral Clinical System, Goa, India), Serum glucose kit (Vital Diagnostics, Mumbai, India), Sodium hydroxide (SD Fine Chemicals Limited, India), Phenolphthalein indicator (Qualigens Fine Chemicals, Pvt. Ltd.), Topfer’s Reagent (Qualigens Fine Chemicals, Pvt. Ltd.), Petroleum Ether (RFCL Limited, India), Di ethyl Ether (RFCL Pvt. Ltd.), Orlistat (Mankind Pvt. Ltd.), Glycerine (RFCL Limited, India), Serum triglyceride kit (Span Diagnostic Ltd.), Serum HDL kit (Span Diagnostic Ltd.).
Plant collection and authentication
The Rubia cordifolia (leaves) were collected from Sahibabad, Uttar Pradesh (Voucher No. BRD-SBD-01). The drugs was identified and authenticated by Dr. G.P Kimothi, The Principal Scientist, Bioresources Department, Dabur India Ltd, Ghaziabad, Uttar Pradesh, where a voucher specimen is preserved for further reference.
Phytochemical screening
Phytochemical are non-nutritive, chemically active substances present in plants that produce a definite physiological action on the human body and play a significant role in the treatment of serious diseases. Since ancient times, the plants have been used in prevention and treatment of various diseases. The leaf powder of plant under study were analysed for alkaloids, tannins, saponins, quinones, coumerins, sugar and gums as per standard protocols given below (Kokate, 1996; Khandelwal, 2008; Prabhu et al., 2011).
Preparation of ethanolic and aqueous extracts of Rubia cordifolia leaves
The dried and powdered leaves of Rubia cordifolia was placed in thimble of Soxhlet apparatus.100 g of powdered leaves was placed and extraction was carried out by using petroleum ether (50-55 0C) for defatted and residue is extracted with ethanol as solvents using ethanol as solvent for 72 hrs. The extracts were filtered; ethanol was distilled off using rotary evaporator to remove excess solvent. The 25g of air dried ethanol filtrate was soaked in 100 ml distilled water for 24 hrs. The extract was filtered by using muslin cloth. The aqueous & ethanolic extracts was then transferred separately into the empty beakers and evaporated to a thick paste on the water bath, maintained at 500C to get ethanol and aqueous extracts. Finally extracts was air dried thoroughly to remove all traces of the solvent. The dried extracts was then stored in an air tight container for anti-obesity activity.
Animals
Male wistar rats (150-200 g) were used for anti-obesity activity; the animals were kept in the animal house of Translam Institute of Pharmaceutical Education and Research Meerut for experimental purpose. All the animals will acclimatize for seven days under standard husbandry conditions, i.e. room temperature of 25 ± 10°C; relative humidity 45-55% and a 12:12h light/ dark cycle. The protocol was approved by the Institutional Animal Ethical Committee (IAEC) (Approval No. IAEC/PH-16/TIPER/075) of Translam Institute of Pharmaceutical Education and Research (TIPER), Mawana road, Meerut (U.P.). All procedures for animal handling were in accordance with the guidelines of the CPCSEA, Ministry of Forest and Environment, Government of India.
Acute oral toxicity study
Animals were deprived of food except water 16-18 hour prior to dosing on day 0 and the Organization of Economic Cooperation and Development (OECD) guideline for testing of chemicals 420 both the extract up to the dose of 5,000 mg/kg was given orally to test group of rats. Body weight, signs of toxicity (general behaviour, respiratory pattern, cardiovascular signs, motor activities, reflexes, and change in skin and fur) and mortality were observed after the administration at the first, second, fourth and sixth hour and once daily for next 14 days.
Test dose selection
From the result of acute oral toxicity it was clear that no sign of mortality was found at the dose of (5000 mg/kg; hence 1/10th & 1/20th of this dose i.e. 500 mg/kg and 250 mg/kg respectively were selected for study).
Preparation of cafeteria diet
Obesity was induced by Cafeteria diet, prepared by mixing powdered normal chow diet 300 g/kg, Nestle milk powder 250 g/kg, Lard 200 g/kg, Chocolate biscuits 55g/kg, Savory snacks 30 g/kg, Processed meat 60 g/kg, Sweetened cereal 100 g/kg, Sodium chloride 05 g/kg.
Grouping of animals
The animals were divided into seven groups (n=6) which were subjected to following treatment scheduled:
Group 1 (Normal Control): Maintained on standard chow diet and water ad libitum received vehicle (1% TWEEN 80, 5 ml/kg; p.o.) only.
Group 2 (Cafeteria Diet Control/Negative Control): Received vehicle (1% TWEEN 80, 5 ml/kg; p.o.) and Cafeteria diet for twelve weeks to induce obesity.
Group 3 (Standard/Positive Control): Received Orlistat (Standard drug) (30 mg/kg; p.o.) once daily for 6 weeks to rats on Cafeteria diet at the end of sixth week and continued up to the end of the twelve weeks.
Group 4 (Test Group I): Aqueous extract of Rubia cordifolia leaves (250 mg/kg; p.o.) was administered once daily for 6 weeks to rats along with Cafeteria diet at the end of sixth week and continued up to the end of the twelve weeks.
Group 5 (Test Group II): Aqueous extract of Rubia cordifolia leaves (500 mg/kg; p.o.) was administered once daily for 6 weeks to rats along with Cafeteria diet at the end of sixth week and continued up to the end of the twelve weeks
Group 6 (Test Group III): Ethanol extract of Rubia cordifolia leaves (250 mg/kg; p.o. 6 weeks) was administered to rats along with Cafeteria diet at the end of sixth week and continued up to the end of the twelve weeks.
Group 7 (Test Group IV): Ethanol extract of Rubia cordifolia leaves (500 mg/kg; p.o) was administered once daily for 6 weeks to rats along with Cafeteria diet at the end of sixth week and continued up to the end of the twelve weeks.
Assessment of physical parameters
The increase in the body weight was compared to age matched control group and regarded as obesity. BMI, obesity index and adiposity index was calculated and was considered as marker for obesity.
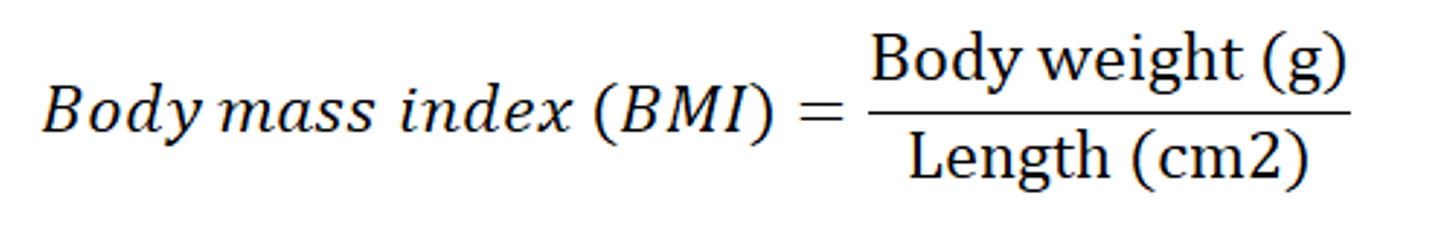
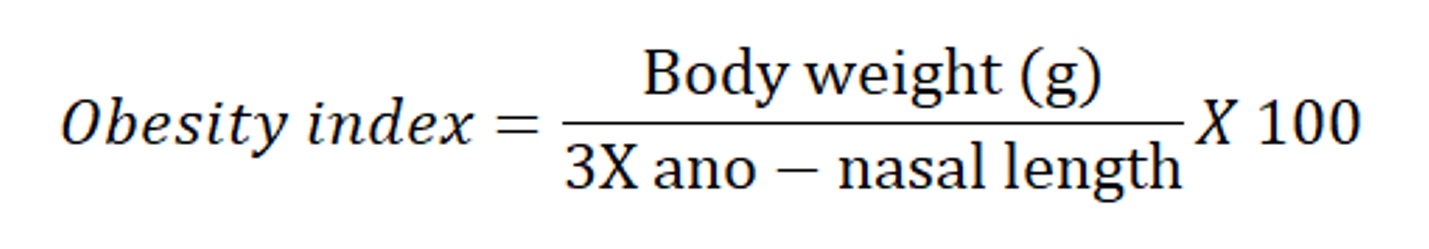
To evaluate the effect of Cafeteria diet and drug treatment, adipose tissues; epididymal, retroperitoneal and mesenteric fat depots were isolated, freed from surrounding tissues, weighed individually and after that Adiposity index was calculated using the following formula (Ainslie et al., 2000):
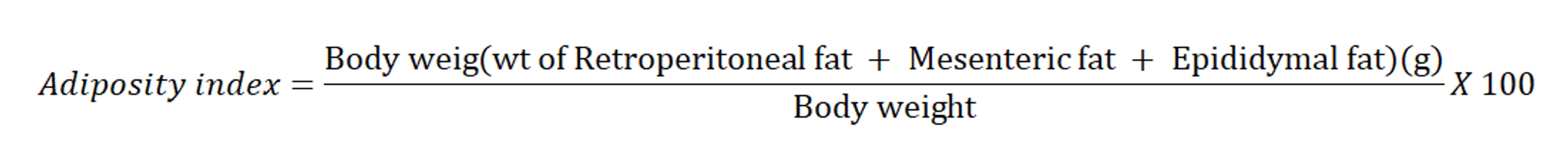
Assessment of biochemical parameters
The hyperlipidemia was assessed by estimating the levels of Total cholesterol, High Density Lipoprotein (HDL), and Triglycerides in blood serum using commercially available kits. Values were expressed in mg/dl. Additionally glucose level in serum was also estimated by using commercially available kits. Values were expressed in mg/dl.
Estimation of Serum Total Cholesterol
The total cholesterol was estimated by cholesterol oxide peroxidase CHODPOD method (Allain et al., 1974) using commercially available kit (Coral Clinical System, Goa, India). 1000 μl of cholesterol reagent was added to 10 μl of serum, 10 μl of standard cholesterol (200 mg / dl) and 10 μl of purified water to prepare test, standard and blank, respectively. All the test tubes were incubated at room temperature for 10 minutes. The absorbance of test and standard samples were noted against blank at 505 nm spectrophotometrically.
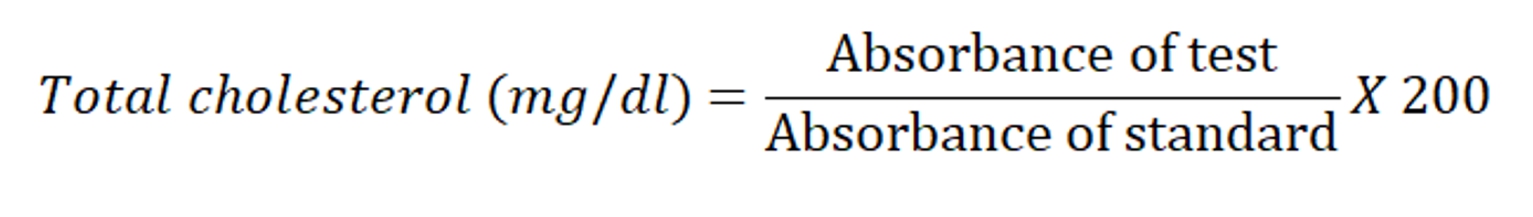
Estimation of Serum Triglycerides
The serum triglycerides were estimated by glycerol-phosphate oxidase peroxidase GPO-PAP method (Werner and Mittelbach, 1981) using commercially available kit (Coral Clinical System, Goa, India). 1000 μl of enzyme reagent was added to 10μl of serum, 10μl of standard (200mg/dl) and 10μl of purified water to prepare test, standard and blank, respectively. All the test tubes were incubated at room temperature for 10 minutes. The absorbance of test and standard samples were noted against blank at 505 nm spectrophotometrically.
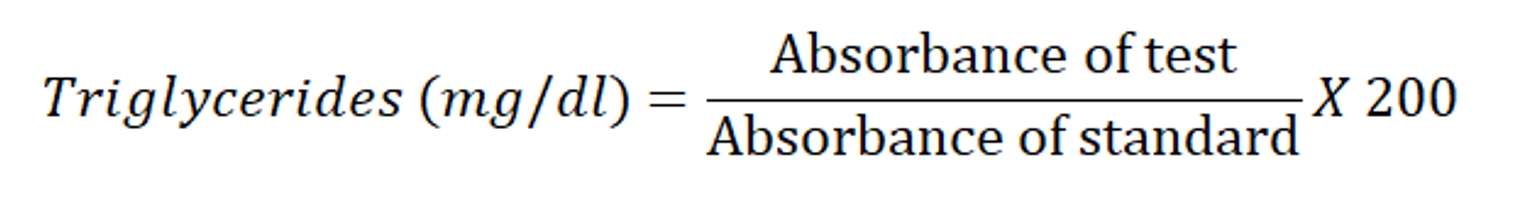
Estimation of High Density Lipoprotein (HDL)
The HDL was estimated by cholesterol oxide peroxidase CHOD-POD method (Allain et al., 1974) using commercially available kit (Coral Clinical Systems, Goa, India).
Step1: 200μl serum and 300μl of precipitating reagent were taken into the centrifuge tube, mixed well and were incubated at room temperature for 5 min and then centrifuged at 3000 rpm for 10 min to get clear supernatant.
Step 2: 1000μl of cholesterol reagent was added to 100μl of supernatant (from step 1), 100μl of HDL cholesterol standard (50 mg/dl) and 100μl of purified water to prepare test, standard and blank, respectively. All the tubes were incubated at room temperature for 10 min. The absorbance of test and standard samples were noted against blank at 505 nm spectrophotometrically. On addition of the precipitating reagent to the serum, followed by centrifugation, HDL fraction remains in the supernatant while the lipoprotein precipitate out.
Cholesterol esters are hydrolyzed by the enzyme cholesterol esterase (CHE) to give free cholesterol and free fatty molecules. This free cholesterol gets oxidized in the presence of cholesterol oxidase (CHOD) to liberate Cholest – 4ene 3 – one and H2O2.
The indicator quinoneimine is formed from hydrogen peroxide and 4 –Aminoantipyrine in the presence of phenol and peroxides (POD). The intensity of the color complex is directly proportional to the cholesterol concentration present in sample. The serum triglyceride was calculated using the following formula:
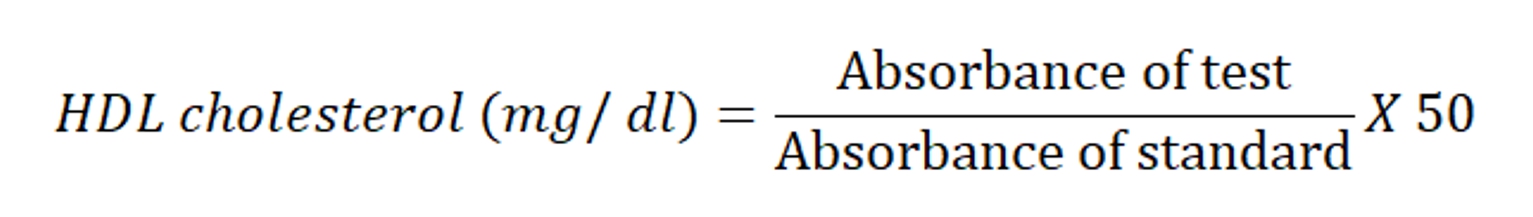
Determination of Low Density Lipoprotein Cholesterol
The LDL was determined by the Friedewald’s equation:
LDL Concentration (mg/dl) = TC– HDLC – (TG/5)
Estimation of Serum Glucose
At the end of the experimental protocol, the blood samples were collected and serum was separated. The serum samples were frozen until analyzing the biochemical parameters. The glucose concentration was estimated by glucose oxidase peroxidase GOD-POD method (Trinder, 1969) using commercially available kit (Vital diagnostic, Mumbai, India). 1000 μl of working glucose reagent was added to 10 μl of standard glucose (100mg/dl) and 10 μl of purified water to prepare test, standard and blank, respectively. All the test tubes were incubated at room temperature for 30 min. The absorbance of test and standard samples were noted against blank at 505 nm spectrophotometrically (Thermo Double Beam Spectrophotometer, Thermo Electron Corporation, United Kingdom).
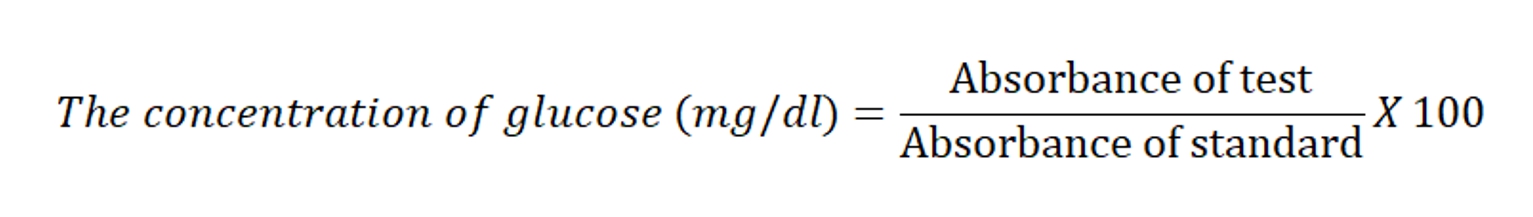
Histopathological study
The Cafeteria diet induced changes in liver and size of body fat depots were assessed for histology, as described by (Buettner et al., 2006; Mourya et al., 2017) and for fat depots as described by (Buettner et al., 2006). The liver and fat depots were excised and immediately immersed in 10% formalin. The liver and fat depots were dehydrated in grated concentration of alcohol, immersed in xylene and then embedded in paraffin. From the paraffin blocks, section of 5-μm thickness were made and stained with haemotoxylin and eosin to assess changes in liver and size of fat depots using light microscopy (40X).
Statistical analysis
Results are expressed as the Mean ± Standard Deviation (SD). The biochemical data for random glucose, lipid profile and fat pad weights were statistically analysed using one-way analysis of variance (ANOVA) followed by Bonferroni multiple comparison test. Confidence level of 95% (p<0.05) was considered to be significant. The effect of aqueous and ethanol extract of Rubia cordifolia leaves, on feed intake, body weight, BMI and Obesity index at different time points were statistically analysed using repeated measure two way ANOVA followed by Bonferroni multiple comparison test.
Results
Phytochemical screening
The present study revealed that aqueous and ethanolic extract of Rubia cordifolia leaves possessed presence of alkaloids, tannins, saponins, quinones, coumarin, sugar and gums (Table 1).
Table 1. Observation of phytochemical screening of extracts
|
Chemical Tests |
Observation |
Inference |
|
|
Aqueous extract |
Ethanolic extract |
||
|
Detection of alkaloids |
|||
|
Mayers test |
Yellow Cream precipitates produced |
+ |
+ |
|
Hagers test |
Yellow Cream precipitates produced |
+ |
+ |
|
Detection of Tannin |
|||
|
Gelatin test |
White precipitate produced |
- |
- |
|
Detection of Saponins |
|||
|
Foam test |
Foam produced |
+ |
+ |
|
Froth test |
Formation of layer of foam |
+ |
+ |
|
Detection of Quinones |
|||
|
Quinones test |
Change of color blue green to red |
+ |
+ |
|
Detection of Coumarin |
|||
|
Coumarin test |
Yellow color produced |
+ |
+ |
|
Detection of Sugar |
|||
|
Sugar test |
Change color yellow to red |
+ |
- |
|
Detection of Gum |
|||
|
Gum test |
Formation of swells |
+ |
+ |
Acute oral toxicity study
No death or perception of adverse reactions was observed on the animals within the fourteen days follow up period. This is an indication that the extracts, were not toxic. From the result of acute oral toxicity it was clear that no sign of mortality was found up to the dose of 5000 mg/kg. Hence the 1/10th & 1/20th of this dose 500 and 250 respectively were selected for study.
Physical assessment
Effect of Orlistat and Rubia cordifolia Extracts on Body Weight, Body Mass Index (BMI) and Feed Intake of Rats
Obesity was induced in normal rats by feeding a Cafeteria diet for 12 weeks. The mean body weights of the seven experimental groups were similar at the start of the experiment. A significant increase in body weight and body mass index (BMI) along with decrease in feed intake was observed in rats of CD control group after 12 weeks, when compared to that of experimental animals of normal control group. On the other hand, treatment with standard drug Orlistat (30 mg/kg, p.o.) once daily for six weeks, significantly (p<0.05) reduced the body weight, BMI and feed intake when compared to that of CD control group. Whereas, once daily treatment for 6 weeks with low and high doses of Rubia cordifolia aqueous & ethanolic extract (250 & 500 mg/kg; p.o.), exhibited significant dose-dependent decrease in body weight, BMI and feed intake, compared to that of CD control group (Figure 1, 2 and 3).
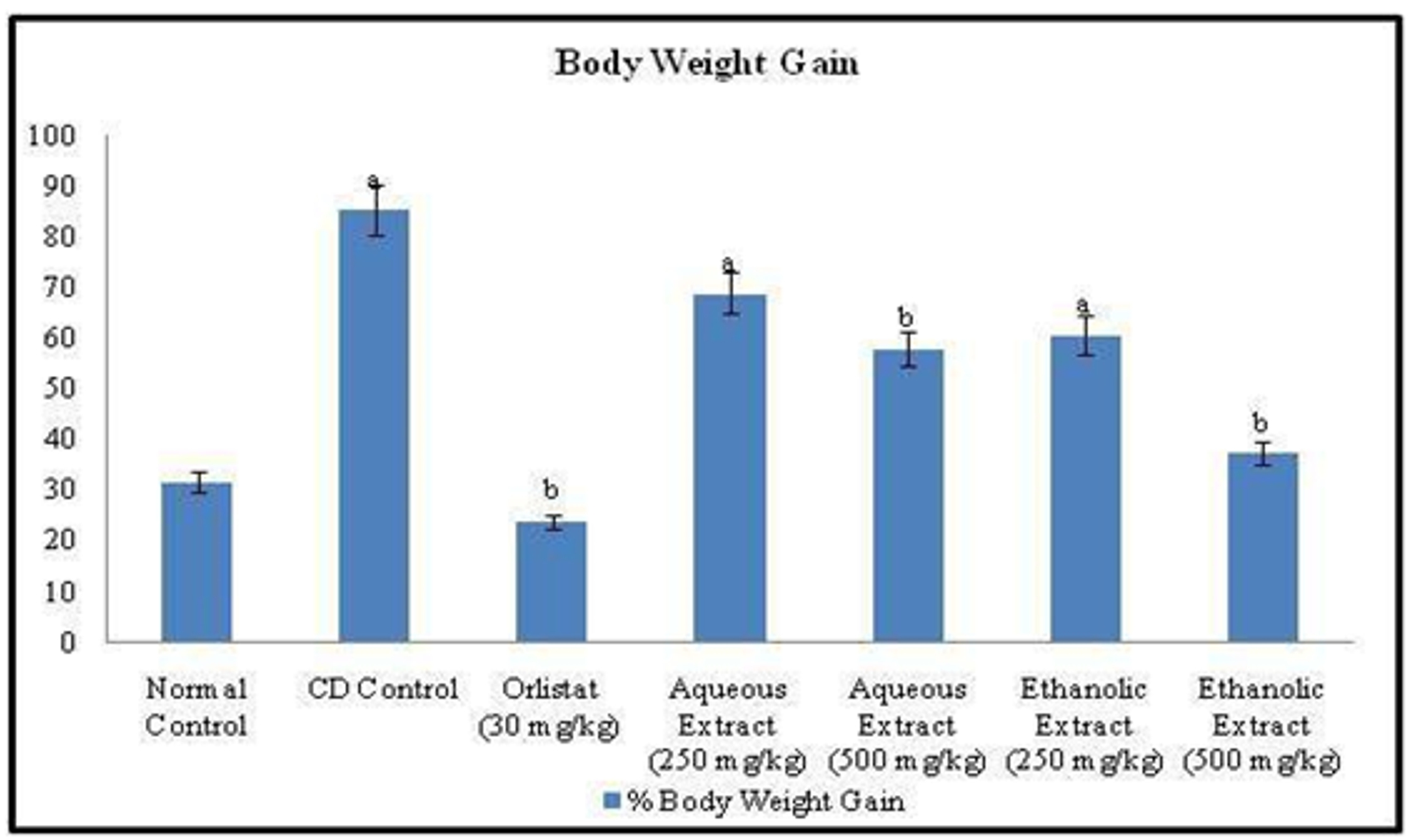 Figure 1. Effect of Orlistat and Rubia cordifolia leaves extracts on body weight of experimental rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Figure 1. Effect of Orlistat and Rubia cordifolia leaves extracts on body weight of experimental rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
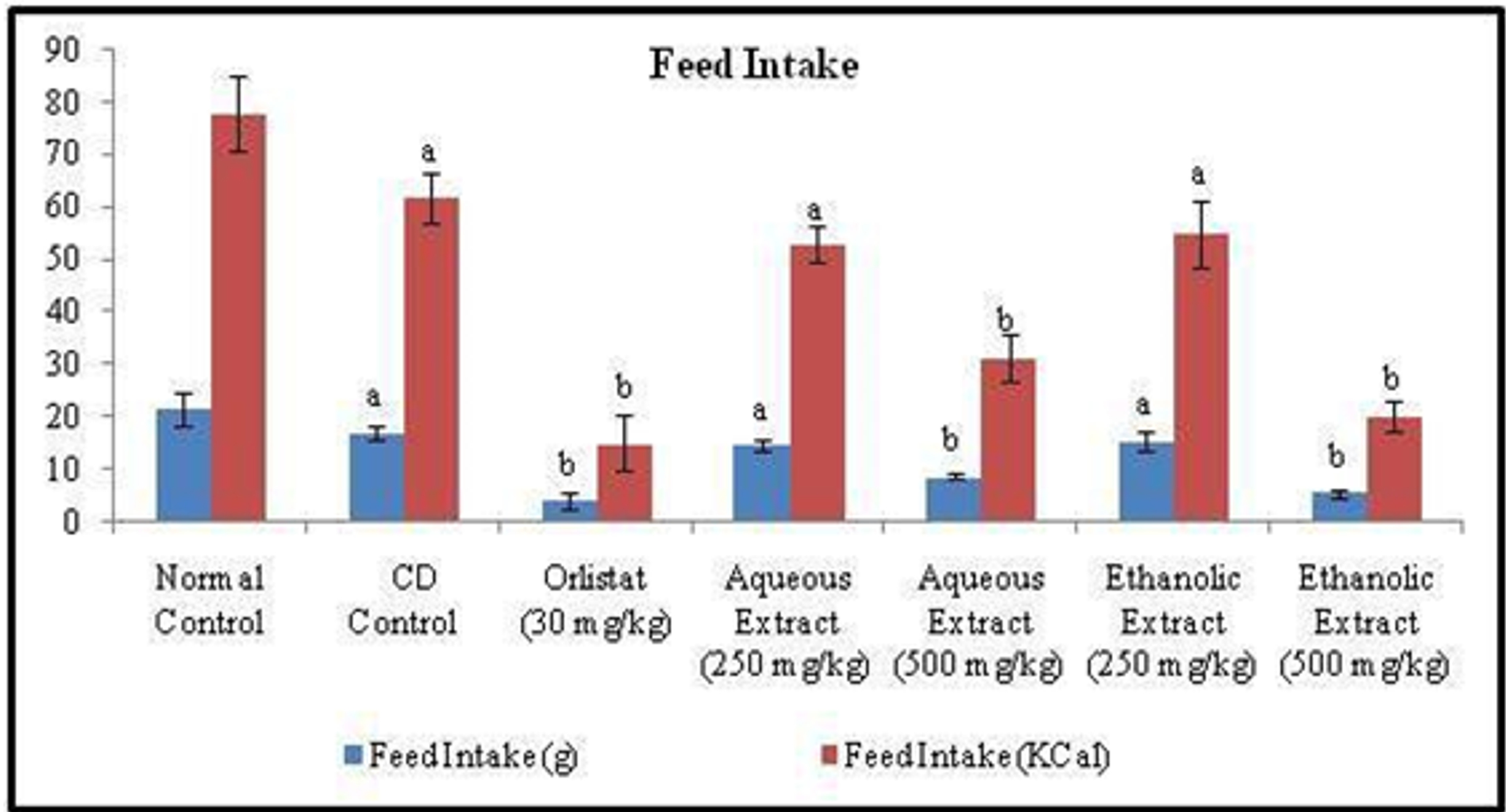
Figure 2. Effect of Orlistat and Rubia cordifolia leaves extracts on feed intake of rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
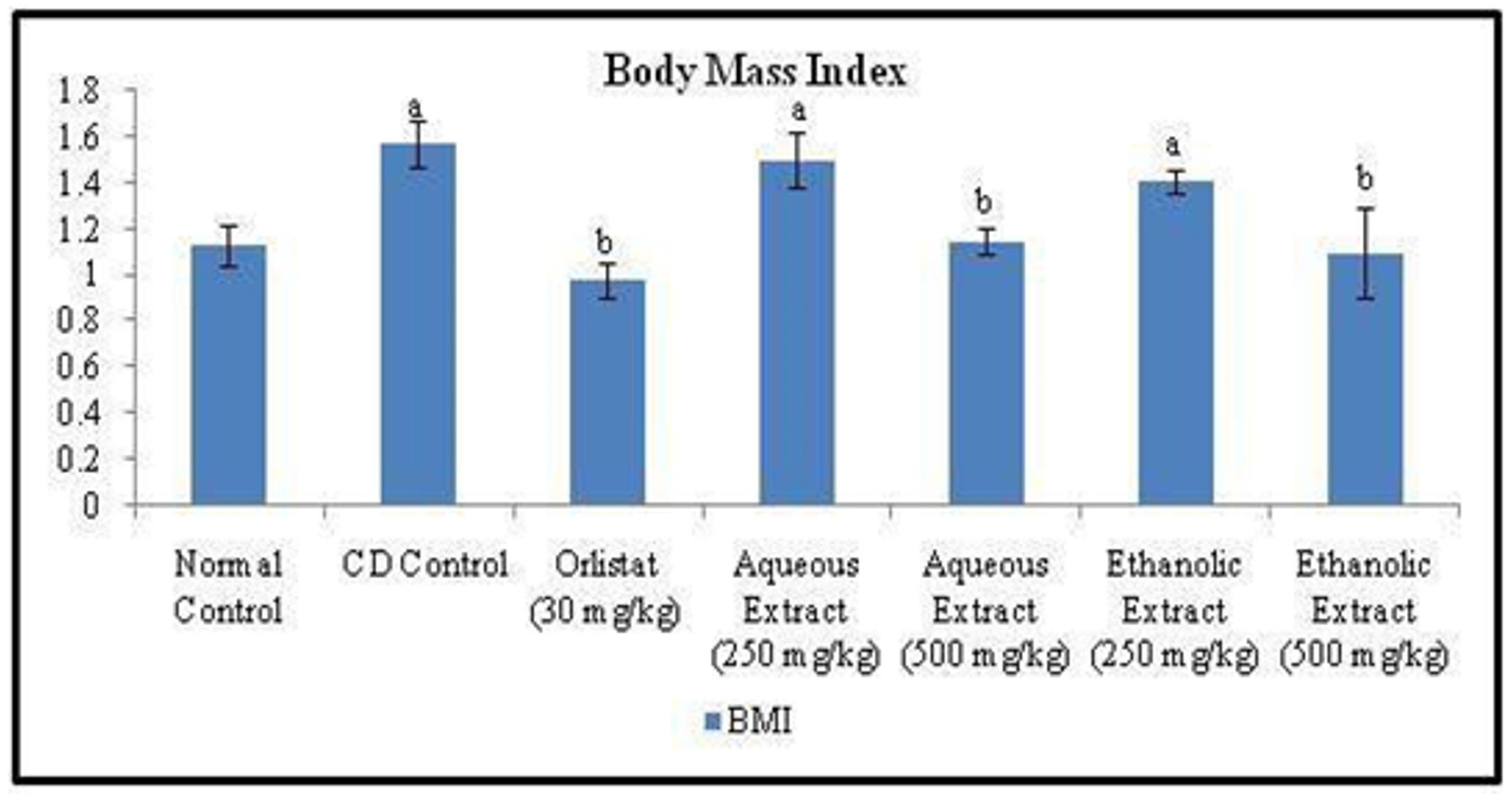
Figue 3. Effect of Orlistat and Rubia cordifolia leaves extracts on BMI of rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Effect of Orlistat and Rubia cordifolia Leaves Extracts on Fat Pad Weights, Total Fat, Obesity Index and Adiposity Index of Rats
The fat pad weights (Epididymal, Mesenteric, Retroperitoneal and Total fat) significantly increased in CD control rats, as compared to those of normal control rats. (Figure 4, 5 and 6). The once daily oral treatment of animals either with standard drug (Orlistat) or aqueous or ethanolic extract of Rubia cordifolia leaves for six weeks significantly decreased the fat pad weights, total fat, obesity index and adiposity index as compared to that of CD control group (Table 2).
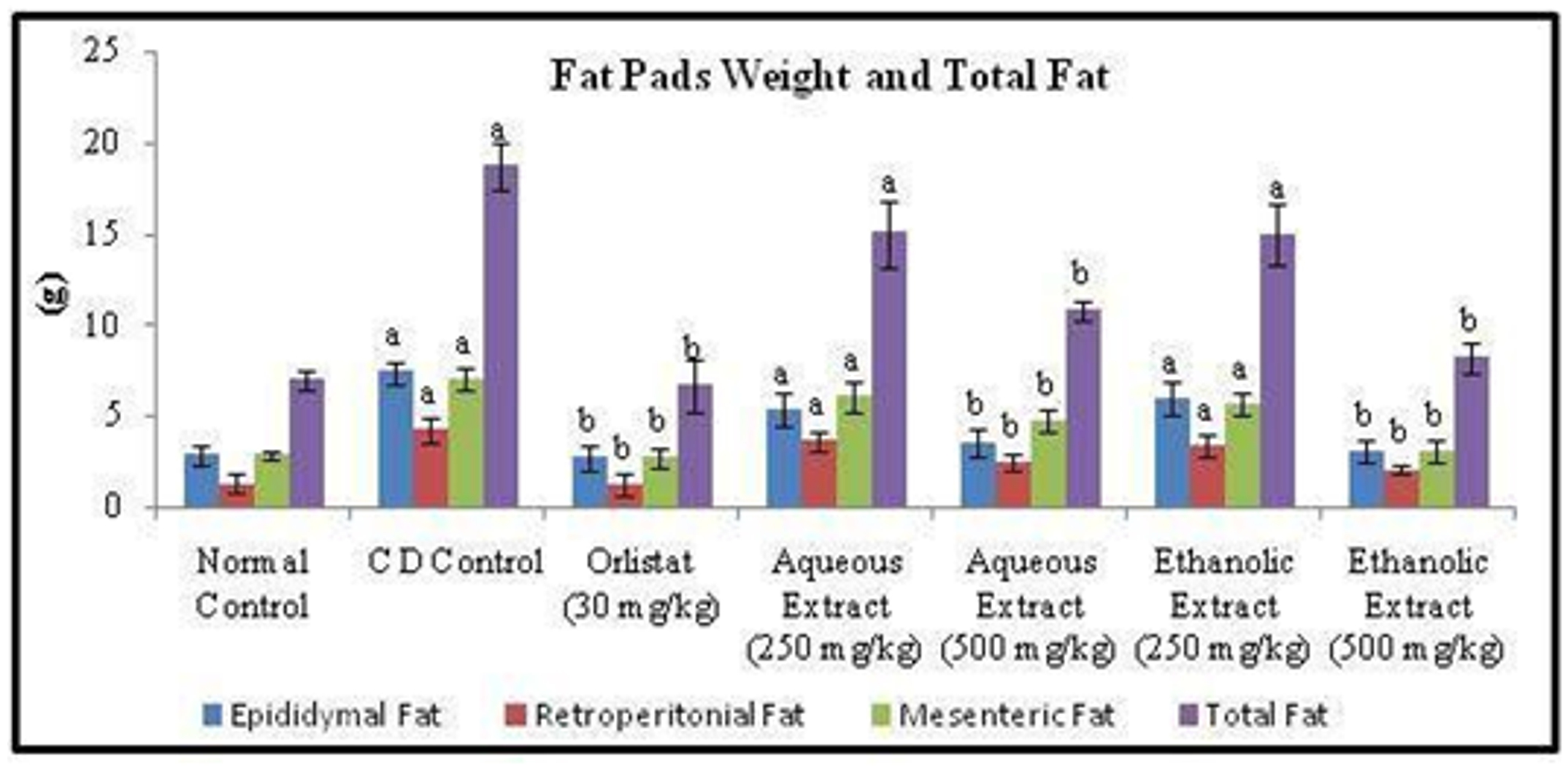
Figure 4. Effect of Orlistat and Rubia cordifolia leaves extracts on Fat Pads Weight and Total Fat of rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
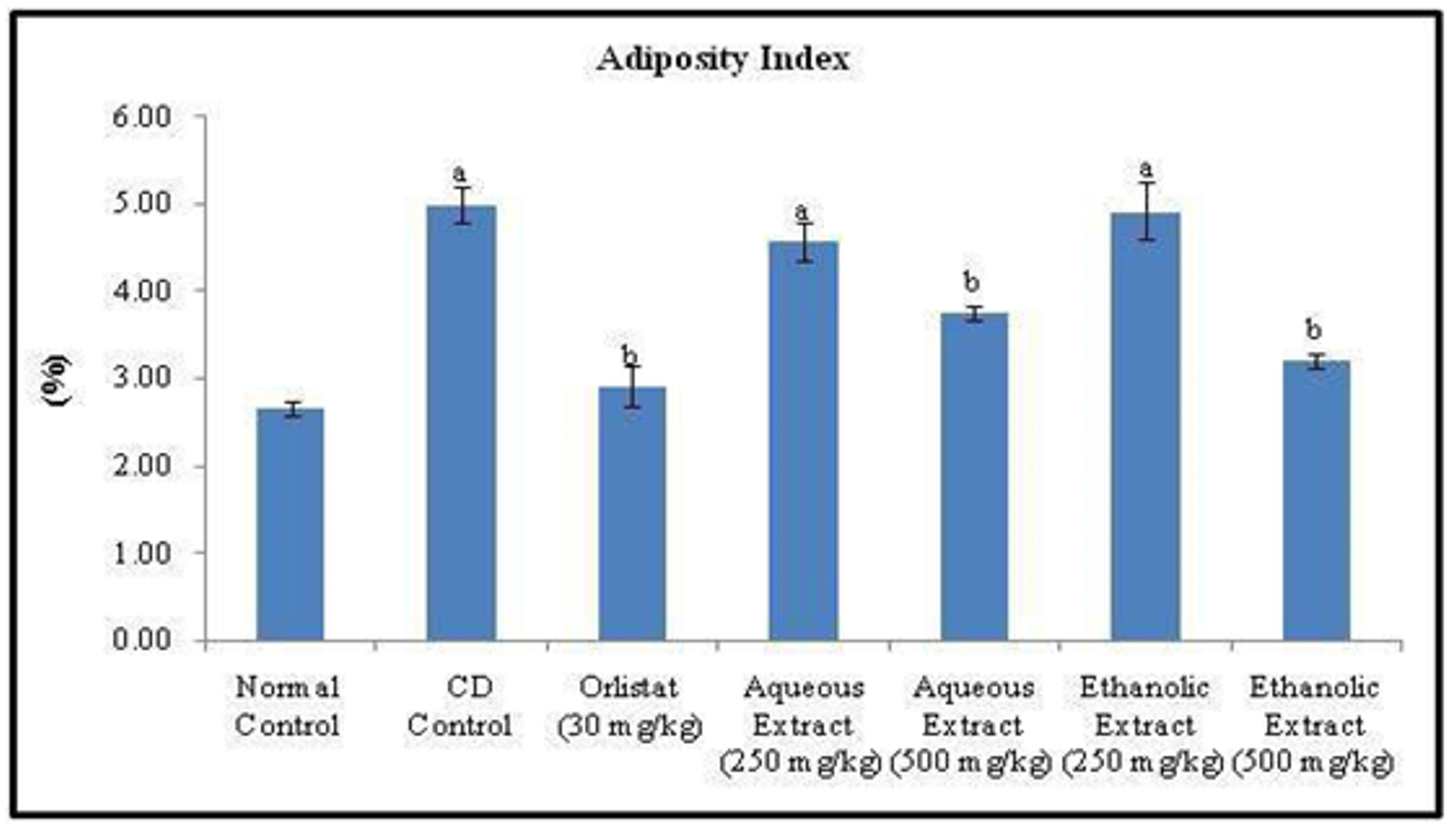
Figure 5. Effect of Orlistat and Rubia cordifolia leaves extracts on adiposity index of rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Figure 6. Effect of Orlistat and Rubia cordifolia leaves extracts on Obesity index of rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Table 2. Effect of Orlistat and Rubia cordifolia leaves extracts treatment on fat pad weights, total fat, obesity index and adiposity index of experimental rats
|
Parameter→ Groups↓ |
Epididymal Fat (g) |
Retro- peritonial Fat (g) |
Mesenteric Fat (g) |
Total Fat (g) |
Adiposity Index (%) |
Obesity Index |
|
Normal Control |
2.83±0.49 |
1.26±0.54 |
2.86±0.25 |
6.95±0.49 |
2.67±0.08 |
345.9±8.07 |
|
CD Control |
7.4±0.62a |
4.26±0.65a |
7.06±0.55a |
18.72±1.34a |
4.98±0.20a |
368.5±6.06a |
|
Orlistat (30 mg/kg) |
2.75±0.69b |
1.22±0.65b |
2.67±0.52b |
6.64±1.44b |
2.91±0.23b |
333.7±3.17b |
|
Aqueous Extract (250 mg/kg) |
5.38±0.93a |
3.6±0.55a |
6.06±0.81a |
15.04±1.83a |
4.57±0.22a |
364.6±9.37a |
|
Aqueous Extract (500 mg/kg) |
3.58±0.73b |
2.45±0.48b |
4.73±0.63b |
10.76±0.56b |
3.75±0.09b |
337.6±7.75b |
|
Ethanolic Extract (250 mg/kg) |
5.98±0.98a |
3.38±0.55a |
5.63±0.63a |
14.99±1.63a |
4.92±0.32a |
336.4±8.34a |
|
Ethanolic Extract (500 mg/kg) |
3.08±0.60b |
2.05±0.24b |
3.08±0.65b |
8.21±0.89b |
3.20±0.09b |
320.8±11.23b |
All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Biochemical assessment
Effect of Orlistat and Rubia cordifolia leaves extracts on Cafeteria Diet Induced
Changes in Blood Glucose Level of Rats Random blood glucose levels were measured at the end of study. Feeding with Cafeteria diet for 12 weeks significantly increased the blood glucose level in CD control group when compared to that of normal control group. Further, the once daily per oral treatment with Orlistat (standard drug) 30 mg/kg for six weeks significantly decreased blood glucose level when compared to CD control group. Also, the treatment of animals either with aqueous or ethanolic extracts (250 & 500 mg/kg) of Rubia cordifolia leaves show significant difference in blood glucose level, as compared to CD control rats (Figure 7).
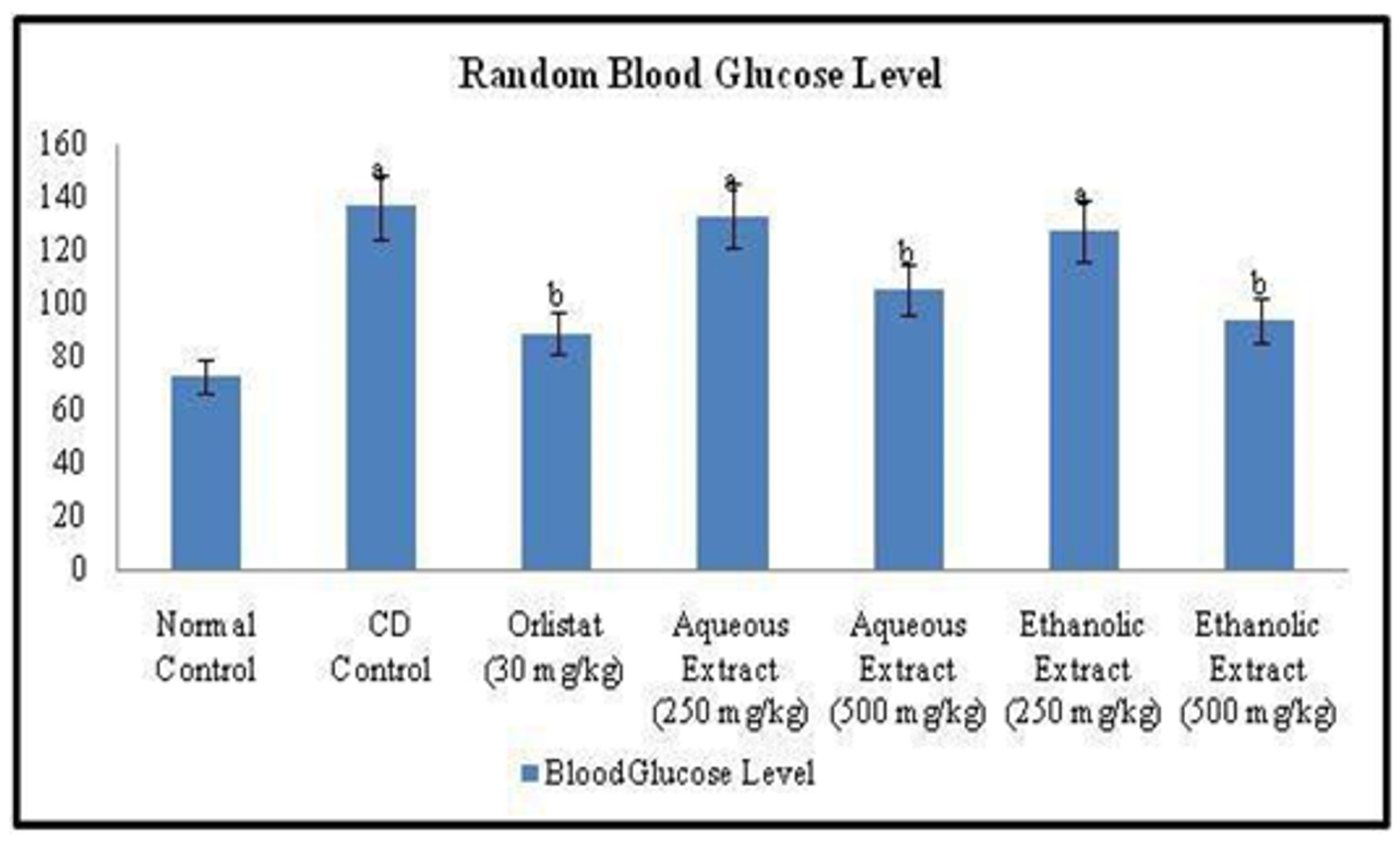 Figure 7. Effect of Orlistat and Rubia cordifolia leaves extracts on random blood glucose level of experimental rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Figure 7. Effect of Orlistat and Rubia cordifolia leaves extracts on random blood glucose level of experimental rats. All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.Effect of Orlistat and Rubia cordifolia leaves extracts treatment on Cafeteria Diet Induced changes in lipid profile of rats
The evaluation of serum lipid profile of experimental animals was carried out for all groups. There was statistically significant (p<0.05) increase in total cholesterol (TC), triglycerides (TG), low density lipoprotein (LDL) and very high density lipoprotein (VLDL) and along with decreased high density lipoprotein (HDL) in CD control group, when compared to normal control group. The once daily oral administration of Orlistat for six weeks along with CD significantly decreased the levels of TC, TG, LDL and VLDL with increase in HDL when compared to that of CD control group. Also, the once daily treatment either with aqueous extract (250 & 500 mg/kg; p.o.) or ethanolic extract (250 & 500 mg/kg; p.o.) significantly decreased the levels of TC, TG, LDL and VLDL with increase in HDL when compared to that of CD control group and comparable to standard drug (Orlistat) treatment (Table 3).
Table 3. Effect of Rubia cordifolia leaves extracts on lipid profile of rats
|
Parameter→ Groups↓ |
Total Cholesterol (mg/dl) |
Triglycerides (mg/dl) |
HDL (mg/dl) |
LDL (mg/dl) |
VLDL (mg/dl) |
|
Normal Control |
61.6± 9.01 |
81.73±10.84 |
35.17±6.48 |
10.08±2.13 |
16.34±2.56 |
|
CD Control |
137.58± 19.48a |
265.59±32.89a |
21.67±4.04a |
62.79±6.16a |
53.11±7.82a |
|
Orlistat (30 mg/kg) |
77.61±11.82b |
106.43± 17.54b |
32.61±3.83b |
23.71±3.45b |
21.28±3.58b |
|
Aqueous Extract (250 mg/kg) |
121.54±18.0a |
203.09±26.94a |
24.54±5.66a |
56.38±6.79a |
40.61±4.91a |
|
Aqueous Extract (500 mg/kg) |
89.18±11.57b |
144.3±23.65b |
30.3±6.17b |
30.02±3.69b |
28.86±3.22b |
|
Ethanolic Extract (250 mg/kg) |
134.94±14.96a |
220.32±28.56a |
25.61±5.57a |
65.26±7.93a |
44.06±5.61a |
|
Ethanolic Extract (500 mg/kg) |
76.21± 13.88b |
118.83±18.61b |
31.09±3.91b |
17.35±2.44b |
23.76±3.54b |
All values are represented as mean ± S.D of n=6/group. Where, a = p<0.05 vs Normal Control; b = p<0.05 vs CD Control.
Effect of various pharmacological interventions on histology of liver and adipose tissue of CD rats
The Cafeteria diet after 12th week produced microvascular and macrovascular steatosis in liver, as compared to rats fed on normal control. However, treatment with Orlistat (30mg/kg, p.o. from the end of 6th week upto 12th week) decreased (p<0.05) microvascular and macrovascular steatosis in liver. Moreover, treatment with Rubia cordifolia ethanolic & aqueous extract (500mg/kg, p.o. from the end of 6th week upto 12th week) produced significant decrease in microvascular and macrovascular steatosis in liver as compared to Rubia cordifolia ethanolic & aqueous extract (250mg/kg from the end of 6th week upto 12th week) and CD control group.
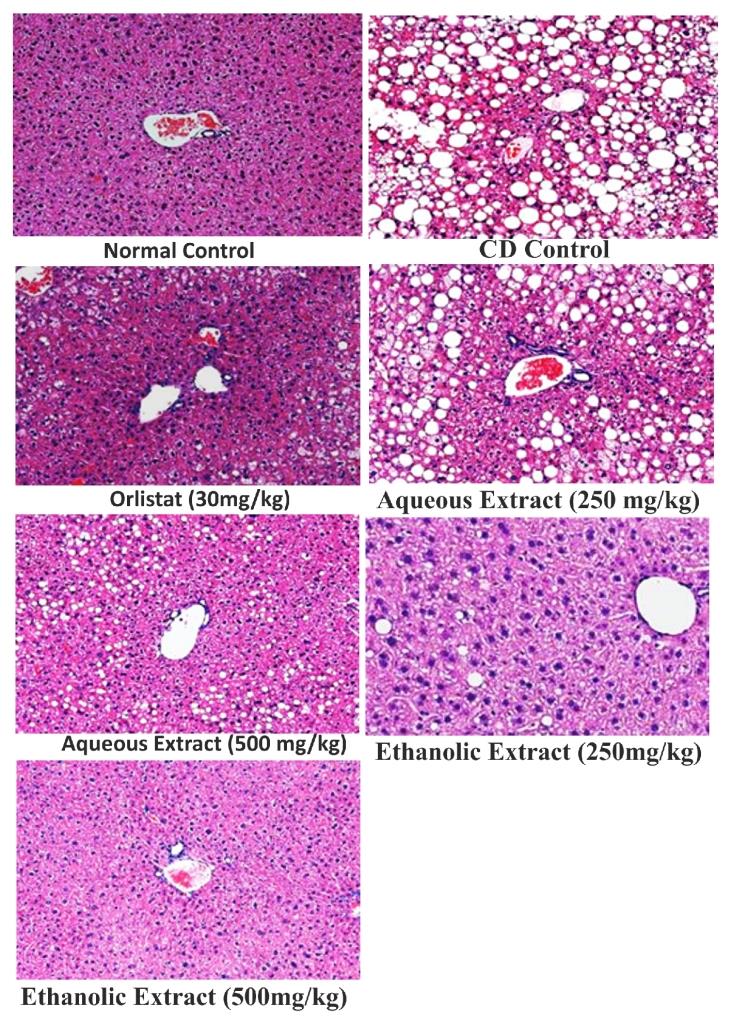 Figure 8. Effect of Orlistat and Rubia cordifolia extracts treatments on liver histology of rats
Figure 8. Effect of Orlistat and Rubia cordifolia extracts treatments on liver histology of rats
Discussion
Obesity has become a major worldwide health problem, not least because it is associated with many diseases, particularly diabetes, hypertension, osteoarthritis, and heart disease. As a result, obesity has been found to reduce life expectancy, and cause huge economic and social problems. Thus, the quest for possible natural products that aid in weight loss has been intensified. Intrigued by recent reports indicating that Rubia cordifolia possesses various biological functions including anti-inflammatory, antitumor, antiatheroscloresis, and antidiabetic activity, we sought to investigate whether Rubia cordifolia has anti-obesity effect. Fat-enriched diet has been used to produce obesity, dyslipidemia and insulin intolerance in rodents. Thus in the present study Cafeteria diet (CD) is used for 12th week to produce obesity and dyslipidemia. It is documented that Cafeteria diet has produced rapid weight gain in rats (Storlien et al., 1986; Singh et al., 1994). The CD rats weighed more than normal controls. The consumption of Cafeteria diet is thought to be one of the main factors. Dietary fat is calorically dense and extremely palatable. It is easily over consumed because it causes less satiety than carbohydrate and protein. The food intake (g & Kcal), body weight, body mass index, obesity index were used as parameters in present study to assess obesity occurred due to CD. The increase in body weight may be due to overconsumption of diet rich in energy in the form of saturated fats (lard) and its deposition into various body fat pads (Srinivasan et al., 2004). The lipogenesis was up regulated by CD in rats lead to elevation of plasma lipid levels (Storlien et al., 1991), which is characterized by elevated TG levels, LDL-C levels and decrease in serum HDL-C (Gluck et al., 1980) in obese rats. Further, feeding of Cafeteria diet produced hyperglycemia in rats (Ikemoto et al., 1995). Therefore serum lipid levels (total cholesterol, LDL, HDL and triglycerides) and glucose levels were estimated in present study as the marker of hyperlipidemia and hyperglycemia. The weight of each of the three adipose tissues (epididymal, retroperitoneal, mesenteric fat depots) increased progressively due to the ad libitum CD feeding (Digirolamo et al., 1998). Therefore three adipose tissues (epididymal, retroperitoneal, mesenteric fat depots) were weighed as an index of adiposity. The weights of adipose tissue (epididymal, perirenal, mesenteric fat depots) were increased in CD fed rats as compared to control rats. Liver Steatosis and size of adipose tissue is increased by CD administration (Adams et al., 2005). Therefore, in the present study extent of liver steatosis and size of adipose tissue was examined histologically using light microscope. In the present study we examined the anti-obesity effect of Rubia cordifolia aqueous and ethanol extracts in Cafeteria diet-fed rats, since this metabolic model of obesity reproduces human obesity better than the genetic obese models. RcAe & RcEe from the end of 6th week upto 12th week significantly and dose dependently decrease body weight as compare to CD control rats and this weight loss was accompanied by depletion of body fat, since treatment with RcAe & RcEe was also shown to decrease the weight of the epididymal, retroperitoneal and mesenteric fat pads. Pharmacological inhibition by RcAe & RcEe from the end of 6th week upto 12th week decrease CD induced obesity by significantly decreasing food intake. The administration of RcAe & RcEe (500 mg/kg/day; p.o.) to rats with Cafeteria diet-induced obesity, significantly lowered plasma TC, TG levels, increased HDL-C levels and improved glucose tolerance. The hypertriglyceridemia observed in CD-fed rats may be due to increased absorption and formation of triglycerides in the form of chylomicrons following exogenous consumption of diet rich in fat or through increased endogenous production of triglyceride-enriched hepatic very low density lipoprotein (VLDL) and decreased triglyceride uptake in peripheral tissues (Srinivasan et al., 2004).
Hypercholesterolemia may be attributed to increased dietary cholesterol absorption from the small intestine following the intake of CD. Indeed, obesity is also associated with an unfavorable lipid profile or dyslipidemia (Kelley et al., 2002). Reduction in body weight gain of CD-fed rats was accompanied by a depletion of body fat stores, since treatment with RcAe & RcEe (500mg/kg/day; p.o.) also significantly reduced the weight of the visceral fat stores compared with that of CD-fed rats. The antiobesity activity of RcAe & RcEe appears partly to be mediated by decreasing dietary fat absorption from the intestine via inhibition of pancreatic lipase activity (Malnick and Knobler, 2006).
Moreover, histological examinations revealed that the sizes of the adipocytes were significantly reduced in RcAe & RcEe -treated rats. The histological examination also showed macrovesicular steatosis in liver tissues of the CD group. However, RcAe & RcEe supplementation noticeably attenuated the extent of steatosis, suggesting that RcAe & RcEe may regulate lipid storage and mobilization in adipocytes. Clinical and experimental studies on obese patients have reported positive effects of Orlistat on serum lipid, cholesterol and glucose levels in addition to its promoting effect on weight loss. Cholesterol and other lipid levels were lower in Orlistat treated group as compared to CD control group. Some authors reported that glucose levels decrease during Orlistat treatment. Orlistat produced a significant decrease in the parameters of obesity (i.e. increase in body weight, body mass index, adiposity index and obesity index) as well as glucose and lipid levels in CD animals. A significant decrease in body fat pad weight was also observed due to treatment of Orlistat in CD fed animals. From the above discussion it can be concluded that Orlistat significantly attenuated the parameters of obesity in CD induced obesity. Conclusively, observed reduction in body weight gain, feed intake, BMI, obesity index, serum lipids, glucose and decreased body fat pad weight suggests that RcAe & RcEe possesses significant anti-obesity potential. During the present investigation both the aqueous and ethanol extracts of Rubia cordifolia were subjected for anti-obesity activity in rats where obesity is induced by giving CD for a period of 12 weeks.
Conclusion
This study investigated the anti-obesity potential of Rubia cordifolia leaves. For the detailed mechanism of these activity by Rubia cordifolia, intensive research is needed which can provide the knowledge about the principle constituents and mechanism of action of Rubia cordifolia leaves in management of obesity.
Acknowledgement
Authors would like acknowledge the management of Department of Pharmacology, Translam Institute of Pharmaceutical Education & Research, Meerut, for the meticulous support.
Conflicts of interest
The author declared no conflict of interest
References
Adams S, Matthews C, Ebbeling C, Moore, C, Cunningham J, Fulton J. Herbert J. 2005. The effect of social desirability and social approval on self‑reports of physical activity. American Journal of Epidemiology 161(1):389‑398.
Ainslie SJ, Fox DG, Perry TC. 2000. Predicting amino acid adequacy of diets fed to Holstein steers. Journal of Animal Science 71:1312-1319.
Allain CC, Poon L, Chan SG, Richmond W, Fu P. 1974. Enzymatic Determination of Total Serum Cholesterol, Clinical. Chemistry 20:470.
Buettner C, Kroenke CH, Phillips RS, Davis RB, Eisenberg DM, Holmes MD. 2006. Correlates of use of different types of complementary and alternative medicine by breast cancer survivors in the nurses’ health study. Breast Cancer Research and Treatment, 100(2):219-27.
Chemicals, D.O.F.O., 2005. OECD Guideline for testing of chemicals.
Digirolamo L, Varnai T, Davies R. 1998. Apparent breakdown of reciprocity in reflected solar radiances. Journal of Geophys Research, 103 (8):8795-8803.
Gluck FW, Kaufman SP, Walleck AS. 1980 Strategic management for competitive advantage. Harvard Busines School Publishing.
Ikemoto K, Satoh K, Maeda T, Fibiger HC. 1995. Neurochemical heterogeneity of the primate nucleus accumbens. Experimental Brain Research, 104(2):177–190.
Kandelwal KR, Pawar AP, Kokate CK, Gokhale SB. 2008. Practical pharmacognosy techniques and experiments, 3rd Edition, Nirali Prakashan, New Delhi, pp-9.
Kang JG, Park CY. 2012. Anti-obesity drugs: a review about their effects and safety. Diabetes & Metabolism Journal, 36(1):13-25.
Katiyar C, Gupta A, Kanjilal S, Katiyar S. 2012. Drug discovery from plant sources: An integrated approach. Ayu 33(1):10.
Kazemipoor M, Radzi CW, Cordell GA, Yaze I. 2012. Potential of traditional medicinal plants for treating obesity: a review. arXiv preprint arXiv:1208.1923.
Kelley WM, Macrae CN, Wyland CL, Caglar S, Inati S, Heatherton TF. 2002. Finding the self? An event-related fMRI study. Journal of Cognitive Neuroscience, 14(5):785-94.
Kokate CK. 1996. Practical Pharmacognosy, pp.115, 4th Edition, Vallabh Prakasan, New Delhi.
Malnick SD, Knobler H. 2006. The medical complications of obesity. Journal of the Association of Physicians, 99(9):565-79.
Mourya P, Shukla A, Rai G, Lodhi S. 2017. Hypoglycemic and hypolipidemic effects of ethanolic and aqueous extracts from Ziziphus oenoplia (L) Mill on alloxan-induced diabetic rats. Beni-Suef University Journal of Basic and Applied Sciences, 6:1-9.
Singh RB, Niaz MA, Ghosh S. 1994. Hypolipidemic and antioxidant effects of Commiphora mukul as an adjunct to dietary therapy in patients with hypercholesterolemia. Cardiovascular Drugs and Therapy, 8(4):659-64.
Srinivasan MV, Zhang SW, Lehrer M, Collett TS. 1996. Honeybee navigation en route to the goal: visual flight control and odometry. Journal of Experimental Biology, 199 (7):237-244.
Storlien LH, Jenkins AB, Chisholm DJ, Pascoe WS, Khouri S, Kraegen EW.1991. Influence of dietary fat composition on development of insulin resistance in rats: relationship to muscle triglyceride and ω-3 fatty acids in muscle phospholipid. Diabetes, 40(2):280-9.
Trinder P. 1969. Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Annals Clinical Chemistry, 15:1039.
World Health Organization. Obesity: preventing and managing the global epidemic. World Health Organization; 2000.