

Rajashekar S. Chavan1, Vinuth Chikkamath2, Shanmukha I.2*
1PG Dept of Pharmacology, K L E’s College of Pharmacy, Belgaum, Karnataka, India.
2PG Dept of Pharmacology, S.C.S College of Pharmacy, Harapanahalli, Davanagere 583131
Karnataka, India.
*Corresponding author
Shanmukha I.
PG Dept of Pharmacology, S.C.S College of Pharmacy, Harapanahalli, Davanagere 583131, Karnataka, India.
Abstract
Objective: This study was aimed to determine the quantity of total poly phenol, flavonoids, and tannic acid and, to assess the cerebroprotective potential of Sonchusasper (L) hill against monosodium glutamate (MSG) and aluminium fluoride (AlF3) induced cerebrotoxicity in wistar albino rats. Materials and methods: Quantitative determinations of phytochemicals of 95% ethanolic extract by estimation of total phenolic content and tannins. The albino rats received 95% ethanolic extract at a dose of 250 and 500 mg/kg and ascorbic acid 500 mg/kg (p.o) daily for 7 days. On all the 7 days, MSG (2g/kg, i.p.) was administered one hour before drug treatment and aluminium fluoride (AlF3) at 600 ppm for 7 days in drinking water. During the treatment the rats were observed for neurobehavioral performance for 50minutes daily. Oxidative damage and histopathological analysis of tissue were also assessed. Results: By preliminary phytochemical screening found that 95% alcoholic extract contains glycoside, flavonoid, tannin, alkaloid, saponins etc. It was qualitatively observed the extract contains higher concentration of polyphenol, flavonoid, and tannin components. Extract and ascorbic acid administration significantly improved body weight and attenuated locomotors activity, rota rod and balance beam test performance compared with diseased groups and reversed the enzyme levels to the normal. Conclusion: It was concluded that ethanolic extract of Sonchusasper (L) Hill (EESA) contain significant amount of phenols and tannins and these constituents play an essential role as an antioxidant and cerebroprotective effects in both diseased groups.
Keywords: Sonchusasper (L) Hill, cerebroprotective activity, monosodium glutamate, aluminium fluoride
Introduction
The brain is a highly sensitive organ, requiring a continuous supply of oxygen, glucose and other nutrients to maintain its function and viability. Several neurons die because of energy depletion, and increased oxidative stress (Moro, 2004). Many natural or manmade toxic substances also alter the normal activity of the nervous system leading to neurotoxicity, which is observed as various physiological and psychological disorders (Massaro, 2002).
Neurodegenerative diseases defined as progressive loss of the functional and structural integrity of central nervous system. The oxidative stress is a primary factor to the development of neurodegenerative diseases like Parkinson’s, Alzheimer’s diseases, neurological condition like seizures, epilepsy, stroke, CNS trauma and amyotrophic lateral sclerosis etc. Oxidative damage usually occurs in organs like brain, heart, skeletal muscles, since the organ are collected primarily post mitosis cell. All the cells and tissues of our body are also equipped with anti-oxidative enzymes like super oxide dismutase, Catalepsy, Lipid peroxidase and substances like reduced glutathione. Normally, the balance is maintained between the oxidative attack of the free radicals and, the antioxidative defence system in the cells and tissues of living body. They dispose the free radicals and when they are generated thereby protecting the cells and tissues from the oxidative attack (Mikhail and Richard, 1994).
Oxidative stress is an important risk factor in the pathogenesis of numerous chronic diseases. Reactive oxygen species are also said to be responsible for the human aging. An antioxidant can be broadly defined as any substance that delays or inhibits oxidative damage to a target molecule. The main characteristic of an antioxidant is its ability to trap free radicals. Antioxidant compounds like phenolic acids, polyphenol and flavonoids scavenge free radicals such as peroxide, hydro peroxide or lipid peroxyl and thus inhibit the oxidative mechanisms that lead to degenerative diseases. Herbal plants are considered as good antioxidant since ancient times (Mahdi et al., 2012).
Glutamate is an excitatory neurotransmitter in the brain. It interacts with two main sub types of membrane receptors like Ionotrophic, is coupled with a cation gated channel and metabotropic, is coupled with G-protein receptor, are further sub divided based on selective agonists into N-methyl-D-aspartate (NMDA), kainite and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA). Interact of glutamate with its inotropic mainly NMDA receptor can lead to neurotoxic changes in some experimental situation by allowing excessive amount of calcium to enter the neuron.
Sonchus asper (L.) Hill is an herbaceous annual plant it grow up to 2 meters in height, the family of Asteraceae (Compositae). The entire plant contains sticky, white latex. There were reported that chemical constituents such as polyphenolic compounds and sesquiterence, lactones glycosides such as sonchusides E, F, G, H and I, and three ion one’s of sonchuionosides A,B and C. It also contains α-amyrin, ß-amyrin, germanicol, taraxasterol, lupeol, epifriedelinol acetate, and stigmasterol while in sesquiterpene lactones glycosides have flavonoids, tannin, saponins, terpenoids, ascorbic acid and carotenoids possess antioxidant, anticancer, anti-inflammatory property. In Pakistan, it is used as traditional (folk) medicine for hormonal disorder and oxidative stress. In Spain, it is commonly used as emollient and it is also used in various disorders like wounds and burns, gastrointestinal infection, kidney and liver disorders like jaundice, cancer etc. Hence, this plant was selected for study of Cerebroprotective activity (Narasimharao et al., 2011).
Materials and methods
Preparation of plant extract and phytochemical screening
The whole plant of Sonchus asper (L.)Hill was collected surrounding Harapanahalli, Karnataka, India in the month of June-July 2013. The plant materials were identified and authenticated by Prof. K Prabhu, Department of Pharmacognosy, SCS College of Pharmacy, Harapanahalli, India. A herbarium specimen No.SCSCOP.Ph.Col Herb.No.004/2013-2014.The dried plant were extracted successively with petroleum ether, chloroform, 95% ethanol, and distilled water at room temperature. The extract was concentrated under reduced pressure (bath temperature 50°C) and stored in airtight container in refrigerator below 10°C. The preliminary phytochemical screening was carried out on different extracts, to identify the presence of qualitative identification type of phytoconstituents (Kokate, 1999; Khandelwal, 2000).
Estimation of total phenolic content (TPC) by Folin-Ciocalteau method
The total phenolic content of 95% extract of Soncusasper (L) Hill was determined. Aliquot of the extract was taken in a 10 ml glass tube, and the volume was made upto 3 ml with distilled water. Then 0.5 ml Folin-ciocalteau reagent (1:1 with water) and 2 ml Na2CO3 (20%) were added sequentially in each tube. A blue colour was developed in each tube and the intensity of the colour was directly proportional to the phenolic content. The blue coloration is obtained due to the formation of molybdenum blue as a result of complex redox reaction between phenols and Phosphomolybdic acid Folin-ciocalteau reagent in alkaline medium. The test solutions were warmed for 1minute, cooled and absorbance was measured at 650 nm. The calibration curve was prepared using catechol as standard. The phenolic content of the plant was expressed as a milligram equivalent of phenol per gram of extract. (Singh, 1980; Heljima et al., 2007)
Estimation of tannins by Prussain blue method
The quantitative estimation of the tannin content in the samples has done prior to the presence of tannins was identified by using the FeCl3 and Gelatin tests (Urve et al., 2010). The quantitative estimation of tannin content in samples was calculated by the method of Price and Butler with some modifications. In short, of a dry plant sample was transferred to 100 ml flask; 50 ml water was added and boiled for 30 min. After filtration with cotton filter, the solution was further transferred to a 500 ml flask and water was added up to500 ml mark. 0.5 ml aliquots were finally transferred to vials, 1 ml K3Fe (CN) 6 and 1 ml FeCl3 were added and water was added up to 10 ml volume. After five min time period, the solutions were measured spectrophotometrically at 720 nm. The actual tannin concentrations were calculated on the basis of the optical absorbance values obtained for the standard solutions in range 5 -25 μg /10 ml.
Animals
Wistar albino rats (weighing 150-250g) of either sex were procured from Sri Venkateshwara Enterprises, Bangalore. They were acclimatized for one week under laboratory conditions. They were housed in polypropylene cages and maintained at 27°C ± 2°C under 12 hrs dark / light cycle and fed with standard rat feed (Gold Mohur Lipton India Ltd.) and water ad labium was provided. Ethical clearance for handling the animals was obtained from the Institutional animal ethical committee prior to the beginning of the project work, and the registration no. is SCSCP/626/6/2012-13 CPCSEA and date is 09.01.2013.
Experimental design
Monosodium glutamate- induced cerebrotoxicity
Overnight fasted rats were randomly divided into five groups of six rats (n=6) each as treated for 7 days follows:
Group 1: Received normal saline (i.p.) + vehicle (p.o.).
Group 2: Received MSG 2 g/kg (i.p.) + normal saline (p.o.).
Group 3: Received MSG 2 g/kg (i.p.) +EESA250mg/kg (p.o)
Group 4: Received MSG 2 g/kg (i.p.) + EESA 500mg/kg (p.o)
Group5: Received MSG 2 g/kg (i.p.) + ascorbic acid500 mg/kg, (p.o) (reference standard)
The gap between 95% alcoholic extract and MSG is 1 hour. The dose of MSG was selected based on previous literature. During the drug treatment, rats were observed for the behavioural changes for 50 minutes daily. On 8th day the rats were evaluated for Balance beam test, digital rota rod test and locomotors. On 9th day rats were sacrificed and brains were isolated for estimation of glutathione, SOD, CAT, lipidperoxidation and protein (Ramanathan et al., 2007)
Aluminium fluoride-induced Cerebrotoxicity
The animals were divided into five groups of six rats each as follows;
Group 1: Received normal saline (i.p.) + vehicle (p.o.).
Group 2: Received (AlF3) 600ppm + normal saline (p.o.).
Group 3: Received (AlF3) 600 ppm +95%EESA250mg/kg (p.o)
Group 4: Received (AlF3) 600 ppm +95%EESA 500mg/kg (p.o)
Group 5: Received (AlF3) 600ppm + ascorbic acid 500 mg/kg, (i.p.) (reference standard)
Animals from each group were fed with drug or vehicle for 10 days prior to aluminium fluoride treatment through their drinking water (7 days at a dose of 600 ppm). During the drug treatment, rats were observed for the behavioural changes for 50 minutes daily (Chandrashekhar, 2010).
Statistical analysis
To check the significance of data, following statistical test were performed: ANOVA: to see the variability within all the groups.
Results
It is observed from the preliminary phytochemical screening that glycoside, flavonoid, tannin, alkaloid, saponins, protein and carbohydrate were found in 95% alcoholic extract. It was qualitatively observed that 95% ethanolic extract contain higher concentration of polyphenol, flavonoid, and tannin components and hence, selected this extract for further study.
Spectroscopy determination of total phenol and yotal tannin
The total phenolic content of 95% ethanolic extract was 18.18 mg/g expressed as equivalent to catechol and the tannin content was found to be 27.82mg/g, as shown in figure 1& 2.
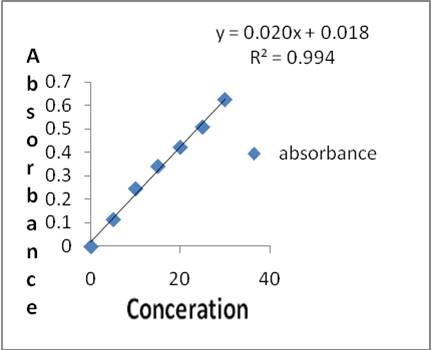 Figure 1. Standard graph of catechol
Figure 1. Standard graph of catechol
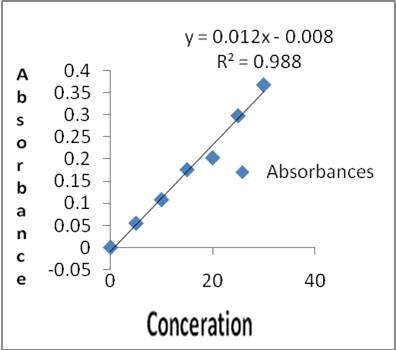 Figure 2. Standard graph of Tannin
Figure 2. Standard graph of Tannin
Effect of 95%EESA on Behavioural characters in MSG treated rats
Motor coordination, Body balance and Locomotor activity were significantly (P<0.001) affected in MSG administered rats, when compared to normal control rats (Table 1). Normal control animal showed the fall of time in 199.5±9.59s, 76.33 ± 6.307s & 98.33 ± 2.3 respectively, whereas MSG alone treated rats showed the fall of time in 117.8±4.9, 24.67 ± 1.7 &50.67± 2.3s. Pre-treatment with 95%EESAandAscorbic acid at 500 mg/kg significantly improved the motor coordination and body balance in MSG treated rats and traversed the beam in168.3±1.72, 53.17± 1.37, 78.83 ± 3.005 and 147.0±2.037, 44.67± 2.525, 66.00 ± 2.017 respectively. There was a dose dependent inhibition of in-vivo LPO and increase in GSH, CAT and SOD in both the models. This was statically significant when compared to the positive control group. Histopathological observations of different groups in MSG are shown in figure 3.
Table 1. shows the effect of 95% EESA change on Behavioural characters and biochemical parameters in MSG treated rats
|
Treatment |
Rota Rod Test (sec) |
Balance Beam Test (sec) |
Locomotor Activity (Count/5 min) |
GSH |
LPO |
SOD |
CAT |
TP |
|||||
|
Mean± SEM |
Mean± SEM |
Mean± SEM |
Mean± SEM |
% increase |
Mean± SEM |
% Inhibition |
Mean± SEM |
% increase |
Mean± SEM |
% increase |
Mean± SEM |
% increase |
|
|
Negative control |
199± 9.59 |
76.33± 6.30 |
98.33 ± 2.30 |
0.289± 0.020 |
…… |
0.308±0.018 |
……. |
0.139±0.007 |
…. |
0.455±0.016 |
….. |
8.112±0.255 |
…… |
|
Positive control |
117± 4.90 |
24.67 ± 1.74 |
50.67± 2.39 |
0.160± 0.003 |
….... |
0.595± 0.023 |
…… |
0.083± 0.003 |
…… |
0.268± 0.010 |
…… |
4.155± 0.348 |
….. |
|
Standard |
168± 1.72 |
53.17± 1.37*** |
78.83± 3.00*** |
0.249± 0.070*** |
55.03 |
0.411± 0.011 |
30.86 |
0.134± 0.003 |
83.96 |
0.448± 0.017 |
67.09 |
7.560± 0.370*** |
81.94 |
|
250mg/kg 95%EESA |
141± 2.27 |
43.33 ± 2.09** |
64.00± 4.71* |
0.219± 0.016* |
19.65 |
0.504± 0.013** |
15.20 |
0.112± 0.004** |
86.51 |
0.344± 0.008** |
28.21 |
5.362± 0.223* |
26.23 |
|
500mg/kg 95%EESA |
147± 2.03 |
44.67 ± 2.52*** |
66.00 ± 2.01** |
0.215± 0.015* |
33.89 |
0.479± 0.012*** |
19.52 |
0.109± 0.005** |
86.87 |
0.360± 0.022** |
34.25 |
5.245± 0.231* |
29.67 |
Each value is expressed as mean ± SEM (n = 6), where, NS represents non significant; ***P<0.001 – highly significant; **P<0.01- very significant; *P<0.05-significant, when compared to ALF3alone treated rats. One-way ANOVA followed by Dunnett's comparison test.
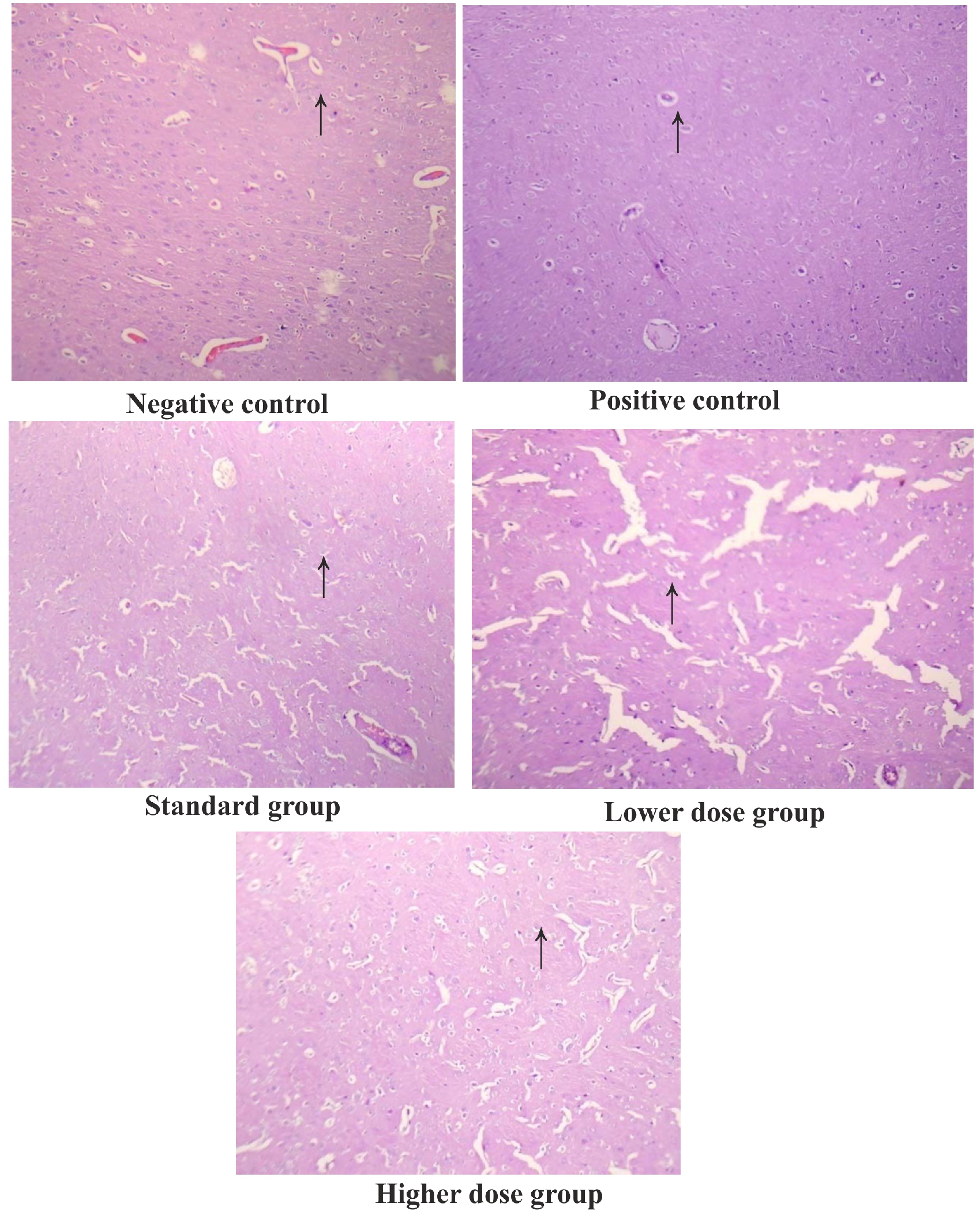 Figure 3. Histopathological observations of different groups in ALF3
Figure 3. Histopathological observations of different groups in ALF3
Effect of 95% EESA on behavioural characters in AlF3 treated rats
Motor coordination, Body balance and Locomotor activity were significantly (P<0.001) affected inAlF3administered rat, when compared to normal control rats (Table 2). Normal control animals showed the fall of timein177.0±3.48; 111.0±6.66, 74.±2.05 respectively. Whereas, AlF3 alone treated rats showed the fall of time in 64.67±4.12, 32.33±2.99 &30.17± 2.92s respectively. Pre-treatment with Ascorbic acid and95%EESA (250, and 500 mg/kg) significantly (P<0.001, P<0.01 and P<0.05) improved the motor coordination and body balance in AlF3 treated rats 115.3±4.745, 83.67±3.480, 89.17±3.683 respectively. There was a dose dependent inhibition of in-vivo LPO and increase in GSH, CAT and SOD in both the models. This was statically significant when compared to the positive control group. Histopathological observations of different groups in AlF3 are shown in figure 4.
Table 2. shows the effect of 95% EESA change on Behavioural characters and biochemical parameters in ALF3 treated Rats.
|
Treatment |
Rota Rod Test (sec) |
BalanceBeam Test (sec) |
Locomotor Activity (Count/5 min) |
GSH |
LPO |
SOD |
CAT |
TP |
|||||
|
Mean± SEM |
Mean± SEM |
Mean± SEM |
Mean± SEM |
% increase |
Mean± SEM |
% Inhibition |
Mean± SEM |
% increase |
Mean± SEM |
% increase |
Mean± SEM |
% increase |
|
|
Negative control |
177.0± 3.48 |
111.0± 6.66 |
74.17± 2.05 |
0.199± 0.006 |
….. |
0.307± 0.032 |
….. |
0.067± 0.001 |
…… |
0.217± 0.003 |
….. |
7.24±0. 14 |
…… |
|
Positive control |
64.67± 4.12 |
32.33± 2.99 |
30.17± 2.92 |
0.076± 0.013 |
…... |
0.430± 0.010 |
….. |
0.028± 0.001 |
…… |
0.130± 0.007 |
….. |
3.91± 0.18 |
…… |
|
Standard |
115.3± 4.74*** |
62.33± 2.60*** |
50.67± 2.33*** |
0.177± 0.007*** |
133.00 |
0.252± 0.010*** |
41.39 |
0.059± 0.0009*** |
106.38 |
0.204± 0.003*** |
57.21 |
5.64± 0.20*** |
43.30 |
|
250mg/kg 95%EESA |
83.67± 3.48** |
51.33± 1.99** |
45.50± 2.43** |
0.113± 0.003** |
49.25 |
0.149± 0.009*** |
25.90 |
0.041± 0.003** |
45.34 |
0.150± 0.002 * |
15.36
|
4.84± 0.06** |
23.70 |
|
500mg/kg 95%EESA |
89.17± 3.68*** |
54.17± 1.74** |
52.83± 3.04*** |
0.146± 0.004*** |
96.91 |
0.319± 0.009*** |
25.96 |
0.053± 0.002*** |
87.75 |
0.167± 0.002** |
28.49 |
5.01± 0.20*** |
27.9 |
Each value is expressed as mean ± SEM (n = 6), where, NS represents non significant; ***P<0.001 – highly significant; **P<0.01- very significant; *P<0.05-significant, when compared to ALF3alone treated rats. One-way ANOVA followed by Dunnett's comparison test. *GSH- Glutathione, LPO- Lipid peroxides, SOD- Superoxide dismutase, CAT- Catalase, TP- Total Protein.
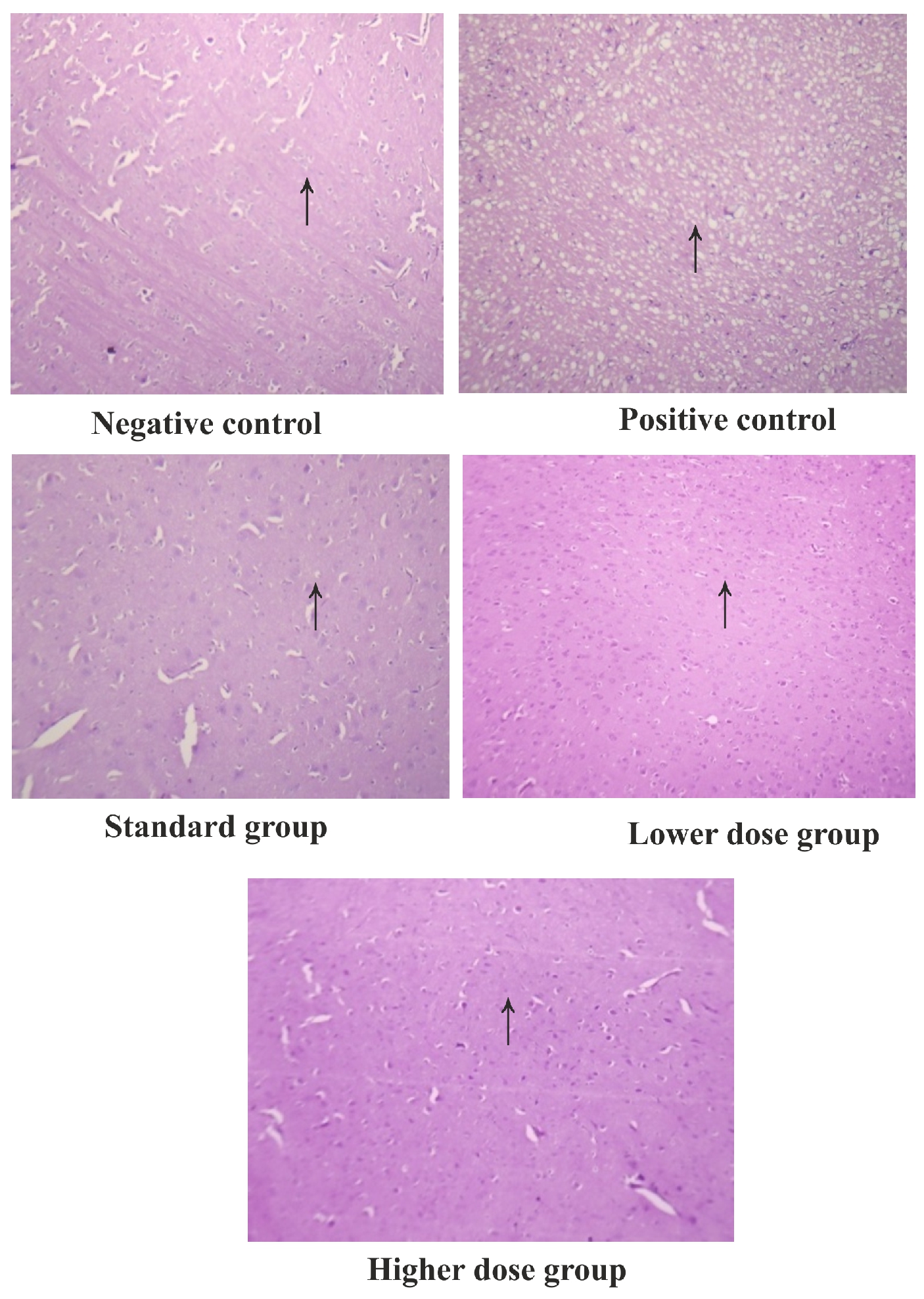 Figure 4. Histopathological observations of different groups in MSG
Figure 4. Histopathological observations of different groups in MSG
Discussion
The human body cells are involved in the metabolic process to generation of energy with release of free radicals scavengers. These free radicals are useful for many oxidative processes of the cells. Reactive oxygen species (ROS) includes free radicals, as well as other oxygen related reactive compounds. The most important ROS like superoxide anion (O2), Hydrogen peroxide radical ( H2O2), Nitric oxide (NO), Hydroxyl radical (OH) are closely involved in various Human diseases such as Alzheimer’s disease, Aging, Cancer, Inflammation, Rheumatoid arthritis and Atherosclerosis125. Plants are the source presence of poly phenols, flavonoids and tannins which possess wide range of biological activities. Hence in this study, the plant Sonchus asper (L) hill reported to possess polyphenolic compounds and tannins, which are reported to possess antioxidant property. These antioxidant property of may be assessed by several screening for assessing the Cerebroprotective activity of MSG and AlF3 induced cerebrotoxicity models were selected. The extracts were prepared separately. Thus prepared extracts were subjected to preliminary phytochemical studies, the results indicated that plant possess poly phenols, tannins, flavonoids, proteins, cardiac glycosides and saponins in 95% ethanolic extract of plant. The spectroscopic determination was observed that, total content of poly phenol and tannins contents in the plant, it was found the 95% EESA possess 18.18mg/gm and 27.82mg/gm expressed to catechol and tannic acid respectively.
The amount of total phenolic and tannin content was found higher amount in 95% ethanolic extract as compare to that of aqueous extract, and also we found that the amount of tannin was higher in 95% of ethanolic extract as compare to that of polyphenolic. In the present study the treatment with glutamate as monosodium glutamate (MSG) induced severe neurochemical damage and neurotoxic effect on some brain region. Various investigators have previously demonstrated some of the neurotoxicological signs induced by monosodium glutamate intake (2mg/kg i.p) in rat. Excessive accumulating of glutamate in the synaptic cleft has been associated with excitotoxicity is implicated in number of neurological disorder.
However there is accumulating evidence suggesting that glutamate-induced toxicity can be mediated through necrosis and apoptosis. The principle of modulating toxic effect of MSG by interfering with neurotransmitter role may have significant impact on understanding neurodegenerative disorder. Aluminium has the potential to be Neuro toxic in human and animals, and is present in many manufactured foods and medicines and also added to drinking water for purification purposes. The present study it is clear that the chronic administration of monosodium glutamate and its subsequent withdrawal caused a significant decrease in monoamine content in most of the tested brain areas at different time intervals. In AlF3 (600ppm p.o for 7 days) in drinking water revealed altered behavioural changes and causes oxidative damage to the brain was estimated by measuring GSH, LPO, SOD, CAT and total protein levels. The histopathological studies were performed to striatal region of the brain. A significant decline in the enzymatic activity of antioxidant enzymes like GSH, SOD, CAT and TP levels monosodium glutamate and aluminium treatment where as LPO levels showed a significant increase as compared to normal control. Synchronous administration of EESA during monosodium glutamate and aluminium treatment reversed the enzyme levels to the normal.
Conclusion
The present Study was carried out to evaluate Cerebroprotective effects of 95% ethanolic extract Sonchus asper (L) hill contain significant amount of phenols and tannins. These constituents may play an essential role as antioxidant and organ protective. Thus our studies support the folklore use of the title plant in cerebroprotective disorders.
Acknowledgments
The authors express deep sense of gratitude to the Management and Principal TMAE’s SCS College of Pharmacy, Harapanahalli.
Conflict of interest
The author declares there is no conflict of interest.
References
Chandrashekhar VM, Ranipariya VI, Ganapaty S, Parashal A, Muchandi AA. 2010. Neuroprotective activity of matricaria reticulates Linn against global model of ischemia in rats. Journal of Ethnopharmacology, 127:645-65.
Heljima K, Vaher M, Gorbatsova J, Kaljurand M. characterization of bioactive compounds contained invegetables of Thesolanaceas family by capillary electrophoresis. Proceedings of the Estonian Academy of Sciences Chemistry, 56(4):172-186.
Khandelwal KR. 2000. Practical Pharmacognosy techniques and experiments, 2nd Ed. Pune Nirali Prakashan, 150-156.
Kokate CK. 1999. Practical Pharmacognosy 4th ed. New Delhi, Vallabha Prakashan, 149- 156.
Mahdi Pour B, Jothy SL, Latha LY, Chen Y, Sasidharan S. 2012. Antioxidant activity of methanol extracts of different parts of Lantana camara. Asian Pacific Journal of Tropical Biomedicine, 2(12):960-965.
Malik CP, Singh EP. 1980. Plant Enzymology and Histoenzymology. M.B kalyani publishers. New Delhi;286.
Massaro EJ. 2002. Handbook of Neurotoxicology, New Jersy: Humana Press journals 2(4):532-3.
Bogdanov MB, Wurtman RJ. 1994. Effects of systemic or oral ad libitum
monosodium glutamate administration on striatal glutamate release as measured using micro dialysis in freely moving rats. Brain Research, 660:337-340.
Moro MA, Cardenas A, Hurtado O, Leza JC, Lizasoain I. 2004. Role of nitric oxide after brainischaemia. Cell Calcium, 36:265–75.
Narasimharao R. 2011. The scientific research on the ulcer protective acting of the plant Sonchusasper. Journal of Pharmacy Research, 4(11):4041-4046.
Ramanathan M, Sivakumar S, Anandvijaykumar PR, Saravanababu C, RathinavelPandian P. 2007. Neuroprotective evaluation of standardized extract of Centella Asciatica in monosodium glutamate treated rats. Indian Journal of Experimental Biology, 45:425-431.
Urve P, Vallo M, Ain R. 2010. Total tannin content in distinct Quercusrobur L. Galls. Journal of Medicinal Plants Research, 4(8):702-5.