

Poonam Dogra1, Tanvi1, Nilanjan Ghosh1, Suresh Janadri2, Darshan Raj C.G3, Rajendra S.V*4
1Department of Pharmacology, GRD(PG)IMT,214, Rajpur Road, Dehradun-248009, (Uttarakhand), India
2 Department of Pharmacology, Acharya & BM Reddy College of Pharmacy, Bengaluru-560090 (Karnataka,) India
3Department of Chemistry, PA College of Engineering, Nadupadavu, Near Mangalore University, Mangalore-574153 (Karnataka) India.
4Department of Pharmacology,Krupanidhi College of Pharmacy, Bengaluru-560035, (Karnataka), India.
*Address for Correspondence
Dr. Rajendra S.V. M. Pharm., Ph. D.
Professor & HOD
Department of Pharmacology
Krupanidhi College of Pharmacy
Bengaluru-560035, Karnataka, India
Abstract
Objective: To evaluate the antiproliferative and cell cycle analysis of aqueous extract of Emblica officinalis fruits on human neuroblastoma and colorectal cancer cell lines. Materials and methods: In the first phase of the study, E. officinalis aqueous extract was subjected to phytochemical investigation and following which in vitro antioxidant activities were performed. In the second phase, brine shrimp lethality assay was carried out. In the final phase of the investigation, MTT assay was performed on human neuroblastoma and colorectal cancer cell lines to assess the in vitro cytotoxicity activity of the E. officinalis aqueous extract. Subsequently, cell cycle arrest was assessed by flow cytometry. Results: Phytochemical screening revealed the presence of various vital phytoconstituents. There was significant DPPH free radical scavenging activity. Aqueous extract of the fruits of E. officinalis markedly increases the mortality in brine shrimp lethality assay. Further, the extract exhibited cytotoxicity on human colorectal and neuroblastoma cancer cell lines and cell cycle arrest at G0/G1 phase. Conclusion: It can be concluded that the aqueous extract of fruits of E. officinalis showed antioxidant and anticancer activities and even arrested the cell cycle at G0/G1 phase.
Keywords: Emblica officinalis, brine shrimp lethality assay, MTT assay, cell cycle, flow cytometry
Introduction
Medicinal herbal drug research has made tremendous growth in the area of anticancer treatment. India is popularly referred to as the medicinal garden of the world since we have been bestowed with a huge number of medicinal plants (Krishnaveni and Mirunalini, 2011). Many herbs have been used for alleviating the problems associated with cancer (Rao et al., 2008). E. officinalis is one such plant used by many local traditional practitioners and its detailed literature is quoted in Ayurveda. This medicine is also being used in China and in Thailand. The raw fruit of this plant is used for a wide variety of human diseases including cancer. Further the plant is deployed in pain, bacterial infection, inflammation and as antimutagenic, antioxidant, antitumor (Kin et al., 2005; Arora et al., 2003). Extract of E. officinalis reduced solid tumors in mice subcutaneously induced by Dalton’s lymphoma ascites cells via interference with the cell cycle regulation. Further studies demonstrated that fruits of E. officinalis extract exhibited cell toxicity and inhibit the in vitro proliferation of MK-1 (human gastric adenocarcinoma) and B16F10 (murine melanoma) (Rajeshkumar et al., 2003). The phenolic compounds isolated from the fruit extract of E. officinalis are responsible for the antiproliferative activity and upon investigation; the fruits reveal the presence of pyrogallol, an active compound present in both crude and n-butanol fractions that inhibits proliferation of human lung cancer cells by inducing G2/M phase cell cycle arrest and apoptosis (Ngamkitidechakul et al., 2010).
E. officinalis is a rich in vitamin C and the extract being water soluble may scavenge free radicals responsible for initiating lipid peroxidation. It contains high levels of total proteins, polysaccharides and calcium (Thakur et al., 1989). Moreover, the plant is rich in tannins (gallic acid, elagic acid, phyllemblic acid, emblicol etc.) (Ghosal et al., 1996). Aqueous extract of fruits of E. officinalis inhibited cell growth of different human tumor cell lines including A549 (lung), SW620 (colorectal), MDA-MB- 231(breast) HeLa (cervical) (Ngamkitidechakul et al., 2010). The phytochemical components including alkaloids, flavonoids, terpenoids and tannins have been shown to produce biological activities. The present study is proposed to investigate the antioxidant, anticancer and cell cycle analysis of aqueous extract of E. officinalis fruits on colorectal and neuroblastoma cell lines. Further, free radicals are implicated in pathophysiological conditions like neurodegenerative disorders, cancer, rheumatoid arthritis and organ damage. Reports indicate the use of this plant as a dietary source as well as scavenging free radicals too and the plant producing these activities find place in cancer treatment (Sumalatha, 2013). Fruits of E. officinalis contain many phytoconstituents like polyphenols including flavonoids, triterpenoids and ascorbic acid. The latter constituent present in fruits of E. officinalis has powerful antioxidant, immunomodulatory and anticancer activities (Kamboj, 2000; Xia, 1997). In the literature, it indicates that the E. officinalis exhibits induction of cancer cell apoptosis and inhibition of in vivo tumour promotion as well as in vitro invasion of Human cancer cell lines such as cervical cancer cell lines (HeLa) and breast cancer lines (MDAMB-231) (Narah et al., 2012). Folklore practitioners in many parts of India including Karnataka have employed this particular plant for the treatment of various cancers including colorectal, breast, cervix and lung. However, the details pertaining to the in vitro cytotoxicity against Human colorectal and neuroblastoma cancers is not yet investigated. Further, cell cycle inhibitory property of the plant is also unclear. Hence it is planned to evaluate these activities using aqueous extract of E. officinalis fruits.
Materials and methods
Chemicals
Dulbecco’s modified eagle medium (DMEM), Trypsin-EDTA, Hank’s Balanced Salt Solution (HBSS) and Foetal bovine serum (FBS) were procured from High media. Thiazolyl Blue Tetrazolium Bromide (MTT), Propidium iodide (PI), doxorubicin, DPPH, DMSO and Tris-HCl were purchased from Sigma Chemical Co. (St. Louis, MO, USA).
Cell Lines
HCT-116 (colorectal cancer cell lines) and SH-SY-5Y (Neuroblastoma cell lines) were procured from National Centre for Cell Science (NCCS) Pune, India.
Collection and authentication of plant material
E. officinalis fruits were procured from local markets of Dehradun in the month of October-November. The fruits was identified and authenticated by Prof. Lakshmaya, Director of Pharmacy, GRD(PG)IMT, Dehradun. The fruits were shade dried at room temperature and pulverized.
Preparation of aqueous extracts
Dried fruits were submitted to decoction (10% w/v, plant: water) for 15-20 min at a temperature of around 100°C to obtain the aqueous fruit extract of E. officinalis. The aqueous extract obtained was kept under refrigerated conditions until use.
Phytochemcial screening
Preliminary phytochemical screening was performed as per the available standard protocol to identify the various phytoconstituents present in aqueous fruit extract of E. officinalis (Kokate, 2005; Trease and Evans, 2002)
DPPH radical scavenging activity
Antioxidants react with DPPH, which is a stable free radical and is reduced to the DPPHH and, the absorbance decreased from DPPH radical to DPPH-H form. The amount of discoloration indicates the scavenging potential of the antioxidant compounds or extracts in terms of hydrogen donating ability. DPPH in methanol (0.1 Mm) was prepared and 1.0 ml of this solution was mixed with 3.0 ml of extract solution in methanol at different concentrations. After thirty minutes, the absorbance was measured at 517 nm. A blank solution was prepared without adding extract. If lower the absorbance the reaction mixture indicates the higher free radical scavenging activity. The capability of scavenging the DPPH radical was calculated using the following equation (Arulpriya et al., 2010; Oktay et al., 2003).

Brine shrimp lethality assay
The brine shrimp lethality assay was carried out to investigate the cytotoxicity of the drug. Freshly hatched brine shrimp larvae were used as test organisms. The different concentrations of the aqueous extract of E. officinalis (1 to 5000µg/ml) was prepared in distilled water and used against brine shrimp larva in different sets. Artificial sea water medium was used as control. Different concentrations of drug along with 10 shrimp larva was added to each well of the micro titer plate and incubated at room temperature (30-37oC) for 24 h. The numbers of dead larva in each well were counted under the light microscope. Then analyze the data by probit analysis and LC50 was calculated (Mosa and Lazarus, 2011; Sreeshmal and Binder, 2014; Firoz et al., 2012).
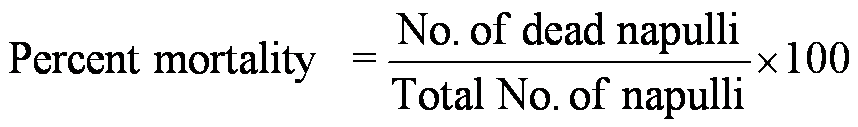
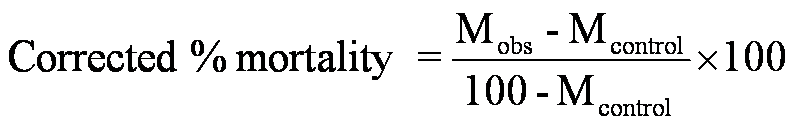
In vitro cytotoxicity
Maintenance of cell lines
Cell lines were procured from NCCS Pune and grown in 25 cm2 tissue culture flasks containing Dulbecco’s modified eagles medium (DMEM) supplemented with 10% FBS, 1% L- glutamine and 50μg/ml gentamycin sulphate at 37ºC in CO2 incubator in an atmosphere of humidified 5% CO2 and 95% air. The cells were maintained by routine sub culturing in 25 cm2 tissue culture flasks. Cell viability was checked with a small sample of the suspension by trypan blue dye exclusion test. Then they were further trypsinzed and tumor cells from the first and second passage of transplantation were stored in liquid nitrogen in cryo vials containing DMEM medium supplemented with 10% FBS and 10% DMSO as preservative at a concentration of 1x 106 cells /ml. This constituted the tumor bank. After every 10 crossings, that tumor cell line was discarded and new passage was started using the original tumor cells from the tumor bank (Bast, 2002; Gambridge, 1985).
Evaluation of cytotoxicity by MTT Assay
This is a colorimetric assay to measures the reduction of yellow 3-(4, 5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide by mitochondrial succinate dehydrogenase. The MTT enter into the cells and passes the mitochondria where it is reduced to an insoluble, dark purple colored formazan product. The cells were mixed with isopropanol and the released solubilized formazan reagent was measured spectrophotometrically. The reduction of MTT can occur only in the metabolically active cells. Trypsinized one T-25 flask and added 5 ml of complete media to trypsinized cells. Cells were counted and recorded as cells per ml. Cells were diluted to give 75,000 cells per ml. Added 100 μl of cells (7500 total cells) into each well and incubated overnight. Then the cells were treated after 24 h with standard or drug. Media was removed carefully. Final volume should be 100 μl per well and Incubated it for 24 h. After 24h, Added 20 μl of 5 mg/ml MTT to each well. One set of wells with MTT but no cells in the control. Incubated for 3.5 h at 37°C in culture hood and remove media carefully. Do not disturb cells and do not rinse with PBS. Added 80 μl DMSO/acidified isopropanol and covered with tin foil then agitate cells on orbital shaker for 15 min. The absorbance was read at 540 nm (Maliyakkal et al., 2013; Masrianif, 2013).
Calculation
Percentage cytotoxicity calculated from the formula: [(AC −AB) − (AT − AB)]/ (AC –AB) *100.
Where, AC, AT and AB are absorbance of control, test and blank respectively. IC50 values can be determined from the dose response curves, by linear regression method.
Cell cycle analysis using flow cytometry
1 x 106 cells were seeded in 25cm2 flasks and after overnight adherence, incubated with indicated test compounds for 24 h. Trypsinized adherent cells to detach, and wash once in 5-10ml 1X PBS to remove residual serum and trypsin. Cell number per sample should be 1-2x106 cells. Cells were spin 1400 rpm for 5 min at temperature 4ºC. Washed the cells once in 5-10ml 1X PBS and re-suspended each cell pellet in 0.5ml 1X PBS. Vortexed the tubes and gently add 4.5 ml ice cold 70% ethanol drop wise over 30 second to a minute. Incubated the cells at -20ºC overnight. Again cells were spin at 1400 rpm for 5 min, 4ºC. Supernatant was removed. Cells were washed twice with 5-10ml 1X PBS. Cells were pellet down at 1400 rpm for 5 min at 4ºC. Remove the supernatant and resuspended cells in 0.5 ml PI staining solution (or 1X PBS solution for –PI negative controls). Incubate the cells for 20 min at 37ºC. Filtered and stained cells were analyzed using Accuri C6 flow cytometer (BD Biosciences, San Jose, CA, USA) using excitation at 488nm and emission at 575/40nm and data analysis was done by using BD AccuriTM C6 software (Dongsool et al., 2005).
Results
Preliminary phytochemical screening
The preliminary phytochemical screening revealed the presence of various phytoconstituents like carbohydrates, flavonoids, alkaloids, tannins and vitamin C.
DPPH radical scavenging activity
It was observed that the extract have demonstrated dose dependent increase in the free radical scavenging activity. There was significant increase in the inhibition of DPPH radicals due to the scavenging activity of the extract. The IC50 value was found to be 151.93µg/ml. The results are graphically depicted in figure 1.
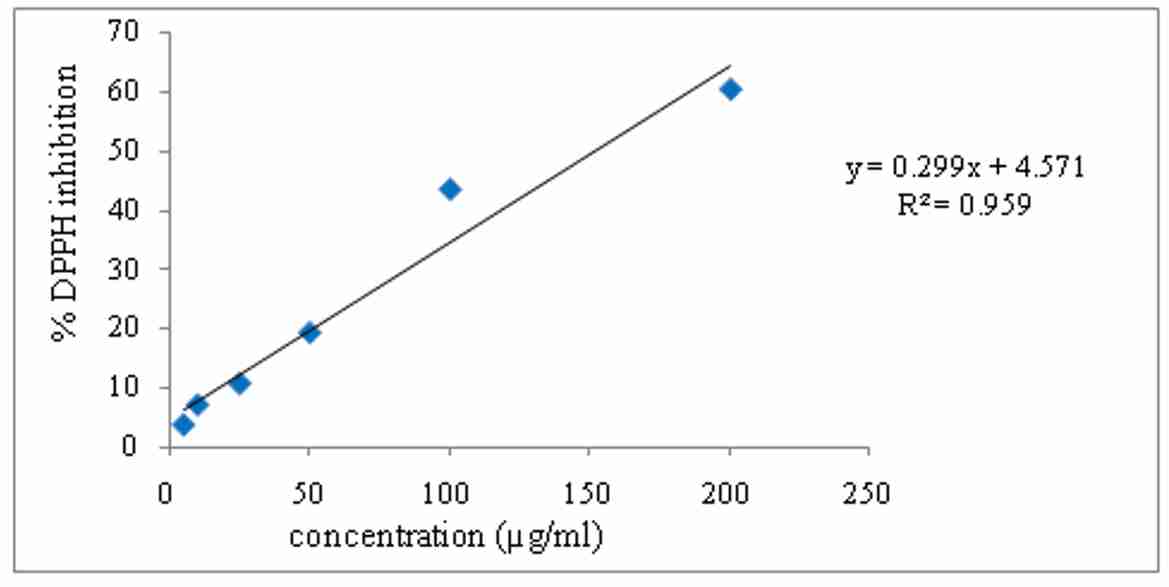 Figure 1. DPPH radical scavenging activity of aqueous extract of E. officinalis
Figure 1. DPPH radical scavenging activity of aqueous extract of E. officinalis
Brine shrimp lethality assay
Aqueous extract of E. officinalis have demonstrated dose dependent increase in the death %age of napulli. The study reveals maximum mortality at a concentration of 2500µg/ml. The LC50 value of the aqueous extract of E. officinalis was found to be 78.69µg/ml. The results graphically depicted in figure 2.
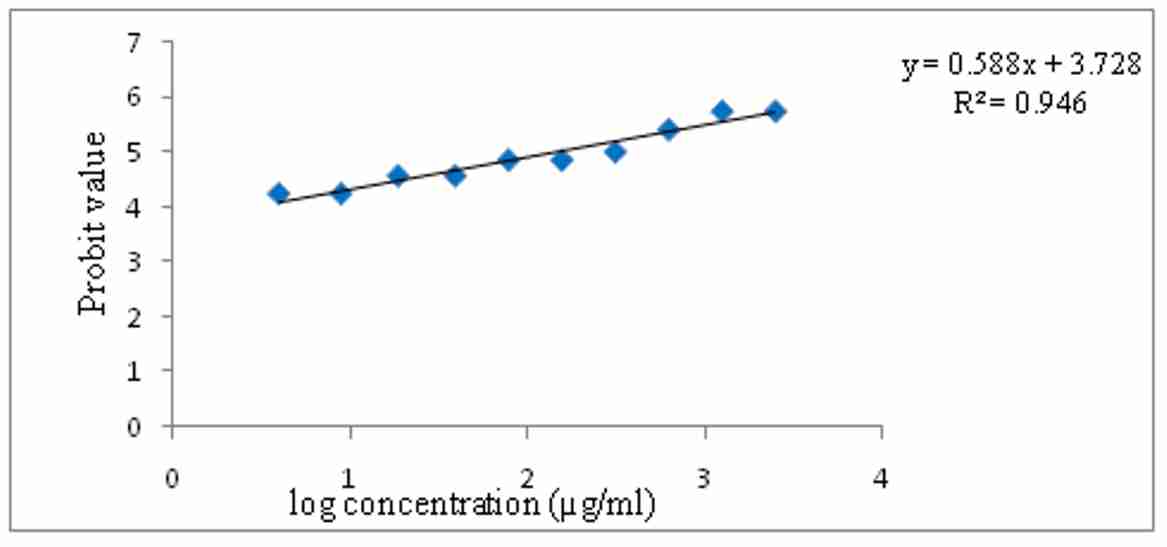 Figure 2. Brine shrimp lethality assay of aqueous extract of E. officinalis
Figure 2. Brine shrimp lethality assay of aqueous extract of E. officinalis
In vitro cytotoxicity
MTT assay on human colorectal cancer cell lines (HCT-116)
It was observed that aqueous extract of E. officinalis has demonstrated the dose dependent increase in the cell death and % cell viability decreases significantly in colorectal cells. The study reveals that the maximum cell death takes place at concentration 200 µg/ml. The IC50 value of the extract was found to be 200µg/ml and IC50 value of Doxorubicin (std.) was found to be 4.5µg/ml. The results were graphically depicted in figure 3 and 4.
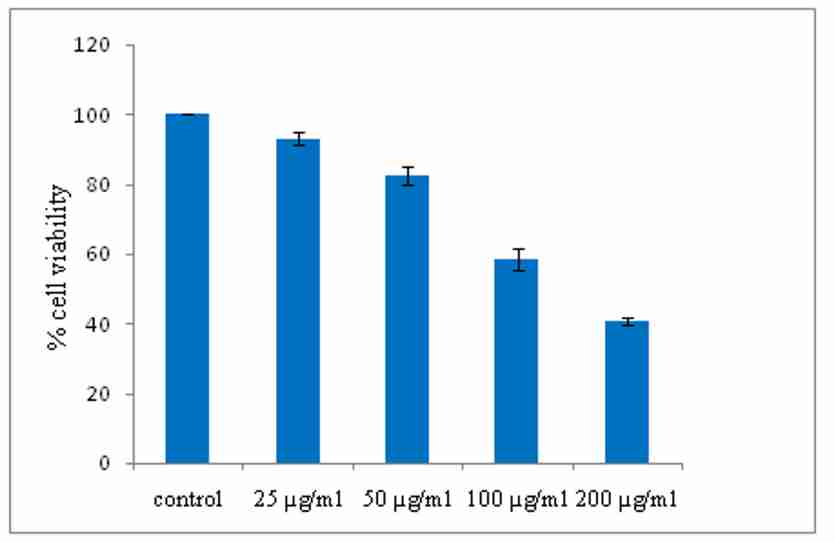
Figure 3. Effect of aqueous extract of E. officinalis on cell viability of colorectal cancer cell lines (HCT-116) after 48 hours
MTT assay on human neuroblastoma cell lines (SH-SY-5Y)
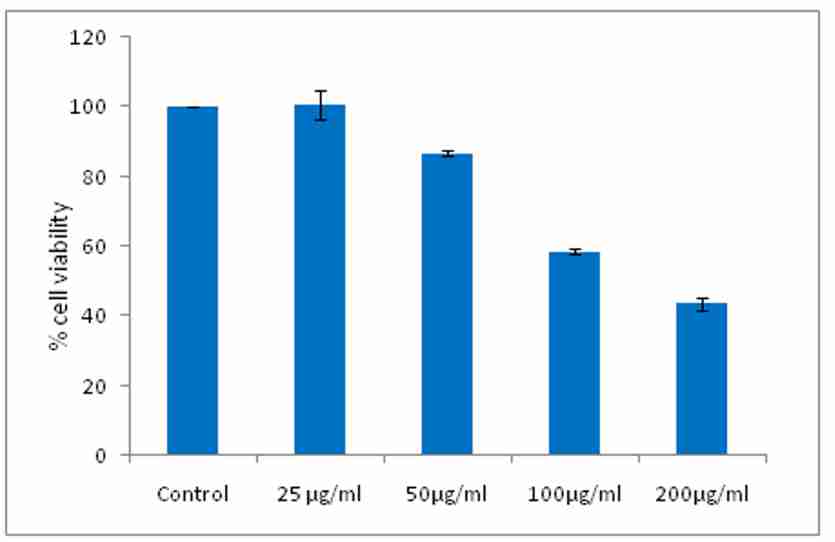
E. officinalis extract demonstrated the dose dependent increase in the cell death and % cell viability decreases significantly in neuroblastoma cells. The study reveals that the maximum cell death takes place at concentration 200 µg/ml. The IC50 value of the extract was found to be 185.4µg/ml and IC50 value of Doxorubicin was found to be 1.007µg/ml. The results are graphically depicted in figure 5 and 6.
Cell cycle analysis using flowcytometry
Cell growth inhibition is often accompanied by cell cycle arrest. It was observed that the normal control have demonstrated the maximum percentage of cells in G0/G1 phase and it was found to be 70.3% and the aqueous extract of E. officinalis fruits (100µM) exhibited accumulation of cells about 79.9% at G0/G1 phase. The results were graphically represented in figure 7 and 8.
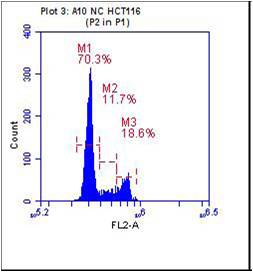
Figure 7. Histogram of propidium iodide v/s cell count of normal control (NC) M1=G0/G1phase, M2= S phase, M3= G2 phase
Discussion
Plants are the largest reservoir of secondary metabolites which are used for the treatment of different disease from ancient time. From ancient times, humans always depend on nature for survival which has been their main source of food, protection, clothing and remedies (Farnsworth et al., 1985). Herbal products have been used as important source that could produce potential chemotherapeutic agents (Kim and Park E, 2002). Plants used in traditional medicine in all over the world. Experimental results suggest that free radicals and reactive oxygen species are linked with the cause and atrophy of disease. The free radicals are generated during the regular physiological functioning of cells/tissues and organs. These free radicals may be very useful because they may promote beneficial oxidative processes. Reactive oxygen species are formed constantly in living organisms as metabolic byproducts (Majumdar and Bhattacharya, 2010). DNA damage may be formed through a direct incorporation of damaged nucleoside triphosphate during replication, which leads to the faulty translation of faulty genetic code, which is a critical event in carcinogenic transformation. Antioxidants are, used for inhibiting the tissue damage by countering the free radicals; most of the antioxidants available in the market are from natural origin eg. Vitamin E, vitamin C, tocopherol, quercetine, etc. in addition there are reports that polyphenolic compounds like flavonoids, tannins are useful as antioxidants. Therefore many researchers are attempting to screen the herbs and herbal preparations containing polyphenolic compounds for cancer protective properties (Ngamkitidechakul, 2010). Hence, in this study a widely grown plant E. officinalis reported to possess flavonoids, tannins, vitamin C has been selected for colorectal cancer and neuroblastoma cancer.
In the preliminary phytochemical studies, the results indicated that the fruits possess flavonoids, tannins and phenolic compounds, carbohydrates, alkaloids and vitamin C. The aqueous extract of fruits was selected for screening in vitro antioxidant properties. Aqueous extract of fruits was subjected to antioxidant activity using in vitro models like reducing power activity, total antioxidant capacity, and DPPH radical scavenging activity. The DPPH assay has been largely used as a reliable parameter to search for the in-vitro antioxidant activity of plant extract. The DPPH antioxidant activity of the extract was estimated from the absorbance of the free radical. The radical scavenging activity of the extract was observed from the decrease in absorbance of the DPPH with the extract with increase in concentration. The results showed a concentration dependent percent scavenging antioxidant activity. The extract showed maximum %DPPH inhibition of 60.3% at concentration 100µg/ml. In the present study, the brine shrimp lethality assay of aqueous extract of E. officinalis was determined. The study reveals maximum mortality took place at a concentration of 1250µg/ml and 2500µg/ml. The degree of lethality was found to be directly proportional to the concentration of the extracts. Brine shrimp lethality assay determines the cytotoxicity of the drug. In this assay degree of lethality were observed with exposure to different dose of the test samples. The aqueous extract of E. officinalis fruits appear to be effective at higher concentration as it showed an LC50 value 78.69µg/ml.
Plants are the promising source of anticancer chemotherapeutic agents. In this study, the cytotoxic effect of aqueous extract of E. officinalis fruits against colorectal cancer cell lines (HCT-116) and neuroblastoma cell lines (SH-SY-5Y) was investigated. The extract significantly inhibited the growth of colorectal cells and neuroblastoma cells in a dose dependent manner. The % cell viability decreases with increase in concentration of the extract. From the results, it was obvious that the aqueous extract of E. officinalis shown IC50 at 200µg/ml using colorectal cancer cell lines and on neuroblastoma cell lines, the IC50 value was found to be 185.4µg/ml. In all our cytotoxic screening, Doxorubicin was used as a positive control and commonly used drug in the treatment of wide range of cancers. The standard anticancer drug doxorubicin exhibited a stronger cytotoxic effect both on colorectal cancer cell lines and neuroblastoma cell lines. Its IC50 value was respectively found to be 4.5µg/ml and 1.007µg/ml. There are many reasons supporting the different toxicity effect of the aqueous extract of E. officinalis towards the colorectal cancer. One of them is due to differ in the levels of polyphenolic compound that have been proven to possess anticarcinogenic effect. Polyphenols, especially ellagic acid and tannic acid found in the plant of the present study have been known to possess cytotoxic/antitumor activity. Our findings through phytochemical investigation indicate the presence of rich concentrations of polyphenols in the aqueous extract of E. officinalis fruits. Further, it was reported in the earlier literature that the polyphenols exhibits potent antioxidant properties28, 29. Hence the anticancer properties of the E. officinalis fruits will be attributed to the presence of these phytoconstituents.
Cancers and many human diseases occur due to disturbances in the various phases of a cell cycle. This is accomplished by many regulatory factors of checkpoints that are either lost or arrested during the process of carcinogenesis. Cell cycle checkpoints are playing an important role in controlling the various mechanisms in cell cycle events. However, there are several chemo preventive phytochemicals could restore and alter regulatory checkpoints through inducing cell cycle arrest. Recent reports also suggest that anticancer agents arrest the cell cycle at the G0/G1, S or G2/M phase and induce cell apoptosis (Liu et al., 2012).
The flow cytometric study on normal control colorectal cancer cell lines (HCT-116) indicate that 70.3, 11.7 and 18.6% cells were arrested at G0/G1, S and G2/M phases respectively. However, the aqueous extract of E. officinalis fruits at 100µM exhibited accumulation of cells about 79.9%, 8.1% and 14.5% in G0/G1, S and G2/M phase respectively. Since the maximum amount of cell population was found to be at G0/G1 phase, this clearly indicates the G0/G1 phase cell cycle arrest. G1 phase arrest of cell cycle progression provides an opportunity for cells to either undergo repair mechanisms or follow the apoptotic pathway. It may imply that aqueous extract may interfere with protein synthesis of colorectal cells thus slow their progression from G1 to S phase during their cell cycle and subsequently initiating apoptosis. The cell cycle arrest observed could be due to inhibitory effect of aqueous extract of E. officinalis fruits on cyclin dependent kinases and activation of cell cycle check points. In general, CDK’s and cyclins are considered cell cycle regulators and initiate their function through the formation of cyclin-CDK complexes (Priyadarsini et al., 2010). However, Cancer cells, exhibit the deregulation of these regulatory proteins, and thus lead abnormal cell proliferation. Since the plant of the present study shown cell cycle arrest at the G0/G1 phase, this could be due to the up-regulation of CDK inhibitors such as p21 and down-regulation of CDK4, CDK6 and cyclin D1 (Bast, 2002). But further studies are required to understand the exact mode of action of E. officinalis extract.
Conclusion
From the results and discussion, it shows that the E. officinalis fruit extract exhibits free radical scavenging, in vitro cytotoxicty against human colorectal cancer cell lines (HCT-116) and neuroblastoma cell lines (SH-SY-5Y). Flowcytometric results exhibited that the extract could possibly arrest the cell cycle at G0/G1 phase. However further studies are required to investigate the effects of particular phytoconstituent responsible for anticancer activity. Proving anticancer potential of herbal extract may reduce the dose and side effects of conventional anticancer drug when used concomitantly.
Acknowledgements
The authors were thankful to the Director and the Management of GRD(PG)IMT, Dehradun (Uttarakhand) for providing all the facilities to carry out this work. Thanks are also due to MCOPS, Manipal, Karnataka for conducting MTT assay and Flow cytometric anlaysis.
References
Arora S, Kaur K, Kaur S. 2003. Indian medicinal plants as a reservoir of protective phytochemicals. Teratogenesis Carcinogenesis and Mutagenesis, (suppl 1):295-300.
Arulpriya P, Lalitha P, Hemalatha S. 2010. In vitro antioxidant testing of the extracts of Samanea saman (Jacq.) Merr. Der Chemica Sinica, 1(2):73-79.
Bast JM. 2002. Cell Preservation Technology, 1:63-80.
Dongsool Y, Rana PS, Chapla A, Sookyeon L, Hyungjoon C, Rajesh A. 2005. A Novel anticancer agent, decursin, induces G1 arrest and apoptosis in human prostate carcinoma cells. Cancer Research, 65:1035-1044.
Farnsworth NR, Akerele O, Bingel AS, Soejarto DD, Guo Z. 1985. Medicinal plants in therapy. Bull World Health Organ, 63:965-81.
Firoz A, Fatema I, Nusratun N, Mithil A, Aynal HR, Ahman KM, Hasanur Rahman, Mumit H, Mohammed R. 2012. Assessment of bioactivity of Bangladeshi medicinal plants using brine shrimp lethality assay. Advances in Natural and Applied Sciences, 6(8):1325-1329.
Gambridge MG. 1985. Vessels for cell and tissue culture. In: setting up and maintenance of tissue and cell cultures. Elsevier scientific Publishers, Shannon, pp1-19.
Ghosal S, Tripathi VK, Chauhan S. 1996. Active constituent of Emblica Officinalis: part 1st the chemistry and antioxidant effects of two new hydrolysable tannins, Emblicanin A and B. Indian Journal of Chemistry, 35b:941-948.
Kamboj VP. 2000. Herbal medicine. Current science, 78:35-38.
Kim J, Park E. Cytotoxic anticancer candidates from natural resources. Current Medicinal Chemistry Anticancer Agent, 2:485-87.
Kin HJ, Yokozawat, Kimhy, Tohda C, Rao TP, Juneja LR. 2005. Influence of Amla (Emblica Officinalis Gaertnl) on hypercholesterolemia and lipid peroxidation in choleaterol fed rats. Journal of Nutrition Science and Vitaminology, 51:413-418.
Kokate CK. 2005. Practical Pharmacognosy, 15th Ed, New Delhi, Vallabh Prakashan.
Krishnaveni M, Mirunalini S. 2011. Amla-The role of ayurveda therapeutic herb in cancer. Asian journal of pharmaceutical and clinical research, 4(3):13-17.
Kuroda H, Nakazawa S, Katagiri K, Shiratori O, Kozuka M, Fujitani K, Masao T. 1976. Antitumor effect of bisbenzylisoquinoline alkaloids. Chemical and Pharmaceutical Bulletin, 24(10):2413-30.
Likhitwitayawuid K, Angerhofer CK, Cordell GA, Pezuto GM, Ruangrungsi D. 1993. Cytotoxic and antimalarial bisbenzylisoquinoline alkaloids from Stephania erecta. Journal of Natural Product, 56(1):30-8.
Liu R, M, LiY B, Zhong JJ. 2012. Cytotoxic and pro-apoptotic effects of novel ganoderic acid derivatives on human cervical cancer cells in vitro. European Journal of Pharmacology. 681:23-33.
Majumdar S, Bhattacharya S. 2010. Comparative in vitro free radical scavenging activity of some indigenous plants. International Journal of Pharm Tech Research. (2):1046-1049.
Maliyakkal N, Udupa N, Pai KSR, Rangarajan A. 2013. Cytotoxic and apoptotic activities of extracts of Withania somnifera and Tinospora cordifolia in human breast cancer. International Jounal of Applied Research in Natural Products, 6(4):1-10.
Masrianif, Mustofa, Jumina, Sunarti. 2013. Pycnarrhena cauliflora ethanolic extract induces apoptosis and cell cycle arrest in hela human cervical cancer cells. International Journal of Research in Pharmaceutical and Biomedical Sciences, 4(4):1060-1068.
Mosa A, Lazarus GG. 2011. In vitro anti-platelet aggregation, antioxidant and cytotoxic activity of extracts of some Zulu Medicinal plants. Journal of Natural Products, 4:136-146.
Narah M, Kalita JC,Kotoky J. Medicinal plants with potential anticancer activities: A review. 2012. International Research Journal of Pharmacy, 3(6):26-30.
Ngamkitidechakul C, Jaijoy K, Hansakul P, Soonthornchareonnon N, Sireeratawong S. 2010. Antitumour effects of Phyllanthus emblica L: induction of cancer cell apoptosis and inhibition of in vivo tumour promotion and in vitro invasion of human cancer cells. Phytotherapy Research, 24:1405-1413.
Oktay M, Gulein I, Kufreviolglu I, Labenson- Wiss U. 2003. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. Cell Preservation Technology, 36:263-71.
Priyadarsini RV, Murugan RS, Sripriya P, karunagaran D, Nagini S. 2010. The neem limonoids azadirachtin and nimbolide induce cell cycle arrest and mitochondria mediated apoptosis in human cervical cancer (Hela) cells. Free Radical Research, 44(6): 624-34.
Rajeshkumar NV, Pillai MR, Kuttan R. 2003. Induction of apoptosis in mouse and human carcinoma cell lines by Emblica officinalis polyphenols and its effect on chemical carcinogenesis. Journal of Experimental and Clinical Cancer Research, 22:201-212.
Rao GV, Sateesh Kumar, Mujahidul Islam, Mansour1Rao SE. 2008. Folk medicines for anticancer therapy-a current status. Cancer therapy, 6: 913-921.
Sreeshmal S, Binder R N. 2014. Brine shrimp lethality assay in two species of Biophytum DC. (oxalidaceae). International Journal of Pharmaceutical Sciences, 6(4):582-586.
Sumalatha D. 2013. Antioxidant and Antitumor activity of Phyllanthus emblica in colon cancer cell lines. International Journal of Current Microbiology and Applied Sciences, 2(5):189-195.
Thakur RS, Puri HS, Hussain Akhtar. Major Medicinal Plants of India. 1989. Central Institute of Medicinal and Aromatic Plants, Lucknow, India.
Trease GE, Evans WC. 2002. Pharmacognosy, 15th Ed, Philadelphia, Saunders.
Xia Q, Xiao P, Wan L, Kong J. 1997. Ethno pharmacology of Phyllanthus emblica L. Zhongguo Zhong Yao Za Zhi, 22:515-518.