

Raman Kumar Tripathia, Shamsher Singhb
aDepartment of Pharmacy Practice, I.S.F College of Pharmacy, Moga, Punjab, India
bNeuroscience Division, Department of Pharmacology, I.S.F College of Pharmacy, Moga, Punjab, India
*Corresponding author
Dr. Shamsher Singh (M. Pharm, PhD)
Neuroscience division (Department of Pharmacology)
I.S.F College of Pharmacy, Moga (142001), Punjab, India
Abstract
Aim: The aim of this article is use of Kymriah technique which uses CAR-T Therapy in the treatment of Leukemia along with the Actemera that is technique which treat cytokine release syndrome in the patients those are treated with the Kymriah. Discussion: Gene therapy is the technique that utilizes gene transfer into patient’s cell as a drug to treat the patient from the various diseases. Gene could be transferred either by germline and somatic techniques with the help of both viral and non-viral vectors. The main target of the gene therapy is to reduce or treat genetic disorders but recently it has been considered as approachable therapy for cancer also called leukemia. Presently the technique which is going to be used as the treatment measure for the leukemia is known as kymriah. It is considered as a first gene therapy for the leukemia patients. The approach is known as adoptive cell transfer that is utilization of chimeric antigen receptor therapy (CAR-T). It allows the direct targeting of anticancer agents and to kill malignant cells without harming the normal cell. CAR-T also diminishes long-term transgenic expression that’s why successful rate is high. In CAR-T tisagenlecleucel is used which directly target cluster of differentiation (CD-19) ALL. The modification of the CAR-T cells uses retrovirus, lentivirus and transposon which can insert the transgene into the host cell genome. The major outcome of CAR-T therapy is that there will be no or least toxic effect to the patient. Conclusion: Due to less side effect of kymriah as compared to the other medications it is using as lifesaving therapy because it kill only the leukemia cells without damaging to the normal cells functioning. Patient treated with the kymriah could develop CRS but Actemera is used to treat CRS.
Keywords: Gene therapy, Acute Lymphoblastic leukemia (ALL), Cytokine release syndrome (CRS), Kymriah, Actemra
Introduction
Gene therapy is the therapeutic transfer of gene in the patient as a medication in command to treat disease (Naldini, 2015). Number of trials has been conducted around in 1989 to analyze the effectiveness of gene therapy. Bone marrow and organ transplants are similar to the gene therapy because they also allow the transfer of the DNA in the cells. It is a powerful treatment for the disorders in which either first line drugs have failed or unsucceeded to treat (Ginn et al., 2013). This approach is useful for disorders initiated by single gene mutation such as cystic fibrosis, sickle cell anemia, thalassemia, hemophilia and muscular dystrophy. Instead of inserting gene into the patient, gene editing might also be done that maybe helpful in treating diseases like cancer and viral diseases (Prakash et al., 2016). Basic method for gene editing includes CRISPER and zinc finger nucleases that are the present and future target approaches. In this, gene is transferred to target cell with the help of carriers that is vector, particularly viral and non-viral vectors (Ramamoorth et al., 2015).
The transfer of DNA or genetic material into host cell helps to replicate for showing their expression (Crawfordet al., 2014). The DNA coding is done by following some instructions to produce specific new copies of viruses which can infect more cells. So with the idea of the virus to inert their gene in the host cell for the rescue of that cell, we can use these viruses to insert the therapeutic gene inside the patient cells (Finer et al., 2017). After transferring, the gene of virus is removed that can show therapeutic activity for a particular disease (Rabinovichet al., 2017). Non-viral vectors are those carriers which can help in transferring gene into patient cells without the help of viruses (Harde et al., 2017). These include biological vectors and engineered vectors. Injection of naked DNA, sonoporation, gene gun, magnetofection, electroporation the use of oligonucleotides, dendrimers, lipoplexes and inorganic nanoparticles. Gene gun and electroporation are the commonly used techniques. Lipoplexes and oligonucleotides are also used in treatment of some diseases (Slivac et al., 2017; Kaji EHet al., 2001; Figure 1).
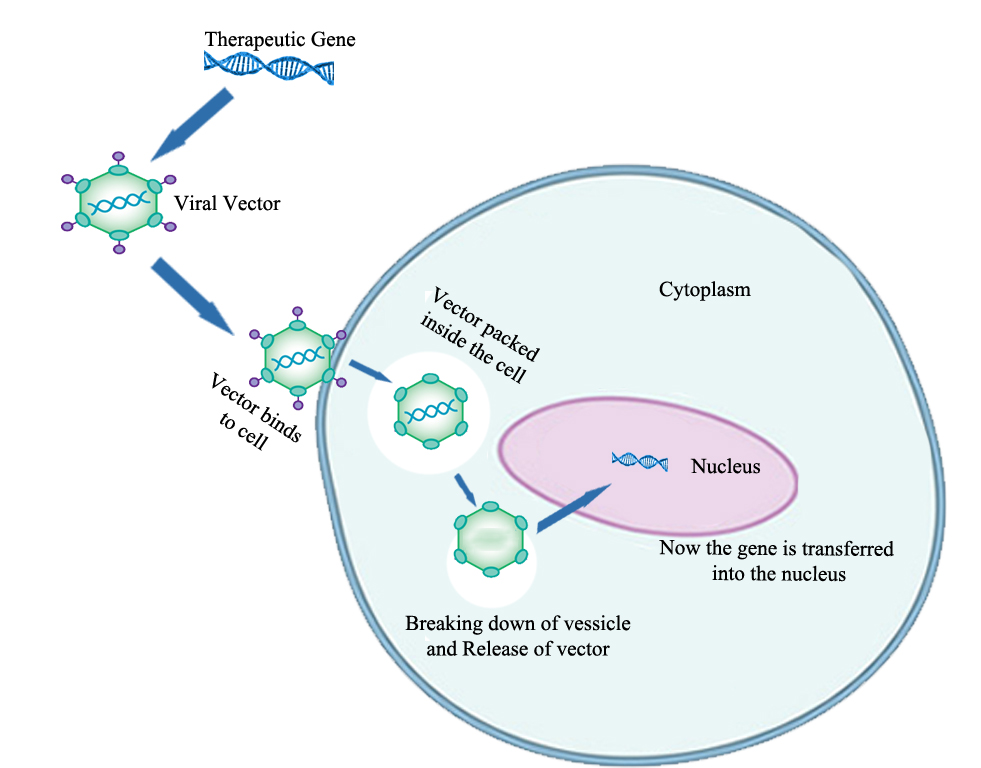
Figure 1. Steps of gene transfermation technique (Kaji et al., 2001)
Cancer is a disease occurs due to abnormal growth of cells as well as their ability to spread from one body parts to the other body parts (Swisher et al., 2017). Lump formation is the primary sign of cancer and other includes abnormal bleeding, unexpected weight loss, alteration in bowel movement and prolonged cough. There are about more than hundred kinds of cancer affecting the people worldwide (Stefani et al., 2017). At the early stage it is difficult to detect but there are some possible signs that the cells show when they are going become malignant. The avoidance of apoptosis (programmed cell death), promotion of blood vessel construction, uninterrupted growth and division even when it’s not needed and formation of metastases which spreads from the primary site to other secondary sites within the host’s body (Ibrahim et al., 2015). On an average about1 in 1,000 pregnant women is affected by cancer that also affect fetal. The breast cancer, cervical cancer, leukemia, lymphoma, melanoma, ovarian cancer and colorectal cancer are the most common cancers found during pregnancy (Salani et al., 2015).
Canceris categorized by the type of cells which the tumor cells look like with. These types include carcinoma, lymphoma, sarcoma, germ cell tumor and blastoma. Carcinoma: the cancer which is derived from the epithelial cells (Tran et al., 2014). The cancer of the breast, lung prostate, pancreas and colon are of the carcinoma type. Sarcomaarises from the connective tissueand it affects to bone, cartilage, fat, nerve cells. Normally each cell develops from cells initiating in mesenchymal cells (Kumar, 2017). Lymphoma and leukemia arise from the hematopoietic cell that matures in the blood and lymph nodes after leaving the bone marrow (Goetsch et al., 2017). Germ cell tumor is derived from pluripotent cells and they are not easy to predict at the early stage in both tests and ovary (Oosterhuis et al., 2017). Similarly, blastoma develops from the precursor cells or embryonic tissue. The carcinoma, sarcoma or blastoma words are used as a suffix and the Latin or Greek word for the organ or tissue of origin is used as the root (Taylor, 2017). For example, cancers of the liver parenchyma derived from the epithelial cells are called Hepatocarcinoma. However, for common type of cancers the English name of the organs are used like ductal carcinoma is most common reason of deaths in the breast cancer due to metastasized (Mileski et al., 2017; Grossman et al., 2008; Jeon et al., 2007) (Figure 2 and 3, Table 1).
Table 1. Cause and death percentages by cancer
|
Sr. No. |
Causes |
Death Percentage |
|
1. |
Tobacco |
22% |
|
2. |
Obesity, lack of physical activity and drinking of alcohol |
10% |
|
3. |
Infections such as hepatitis B, hepatitis C and human papilloma virus infection |
20% |
|
4. |
Inherited genetic defects |
2-10% |
Figure 2. Squamous Cell Carcinoma (Grossman et al., 2008)
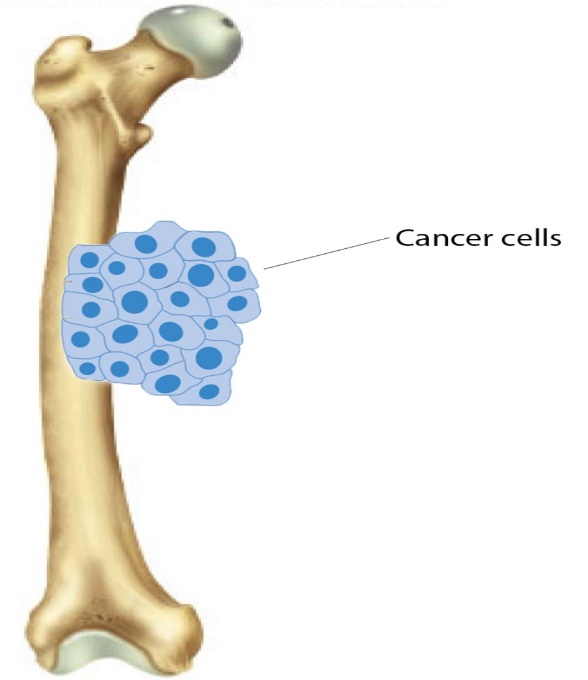 Figure 3. Femoral osteosarcoma (Jeon et al., 2007)
Figure 3. Femoral osteosarcoma (Jeon et al., 2007)Cancer is multifactorial disorder but the major causative factors areisogenetics and epigenetics type (Vineis et al., 2014). In genetics, a cell with normal function and growth has to be altered in order to cause cancer. Oncogenes and the tumor suppressor genes are the infected genes (Liuet al., 2014). Oncogenes promote cell growth, reproduction and tumor suppressor genes suppress the cell division and survival of the cell. Due to the disabling of the tumor suppressing genes, there will be the over expression of the oncogenes that leads to convert the normal cell into tumor cell (Bradneret al., 2017). Mutation, translocation of chromosome and mitosis errors is strongly involved in the alteration in DNA integrity and its function. Due to replication of the altered DNA infected genes extensively damage the cell (Bartek et al., 2017). Sometimes cell can undergo self-disruption that is apoptotic process may trigger the transfer of infected mutated cell from parents to their offspring which further starts to divide and become cancerous (Davidson et al., 2017; Merlo LM et al., 2006) (Figure 4).
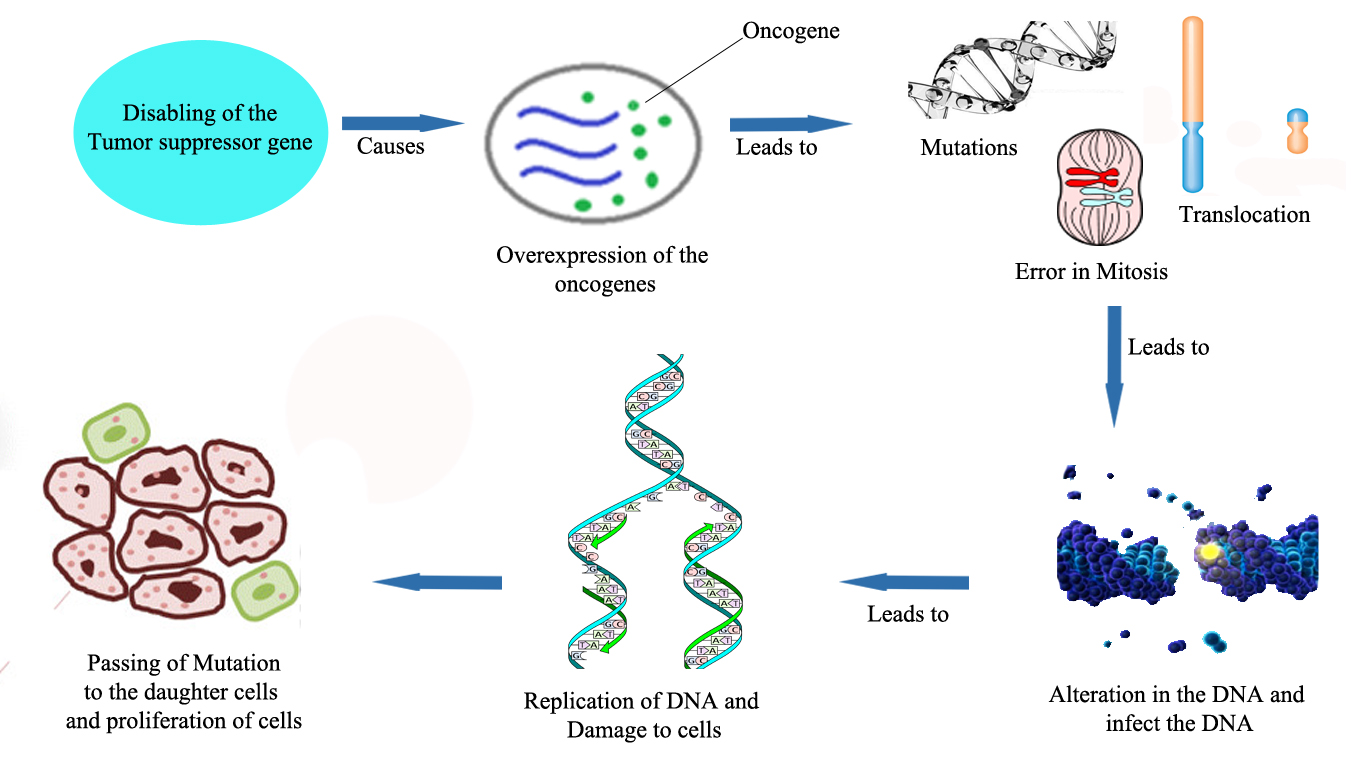 Figure 4. Genetic pathology of cancer (Merlo et al., 2006)
Figure 4. Genetic pathology of cancer (Merlo et al., 2006)
In epigenetics conditions the action of both endogenous (diet related macrophage and neutrophil increase ROS, RNS) and exogenous agents (smoke, radiations, metals, viruses and other genotoxins) further leads to DNA damage (Ow et al., 2017). The damage to DNA alters MiRNA. The various DNA repair genes help to repair the damage but in case of DNA repair deficiency the chances of somatic mutations and epigenetic alterations are too high (Crean et al., 2017). Now the multiplication of altered field defect with driver mutations will progress the cancer (Neureiter et al., 2014; Kanwal et al., 2012) (Figure 5).
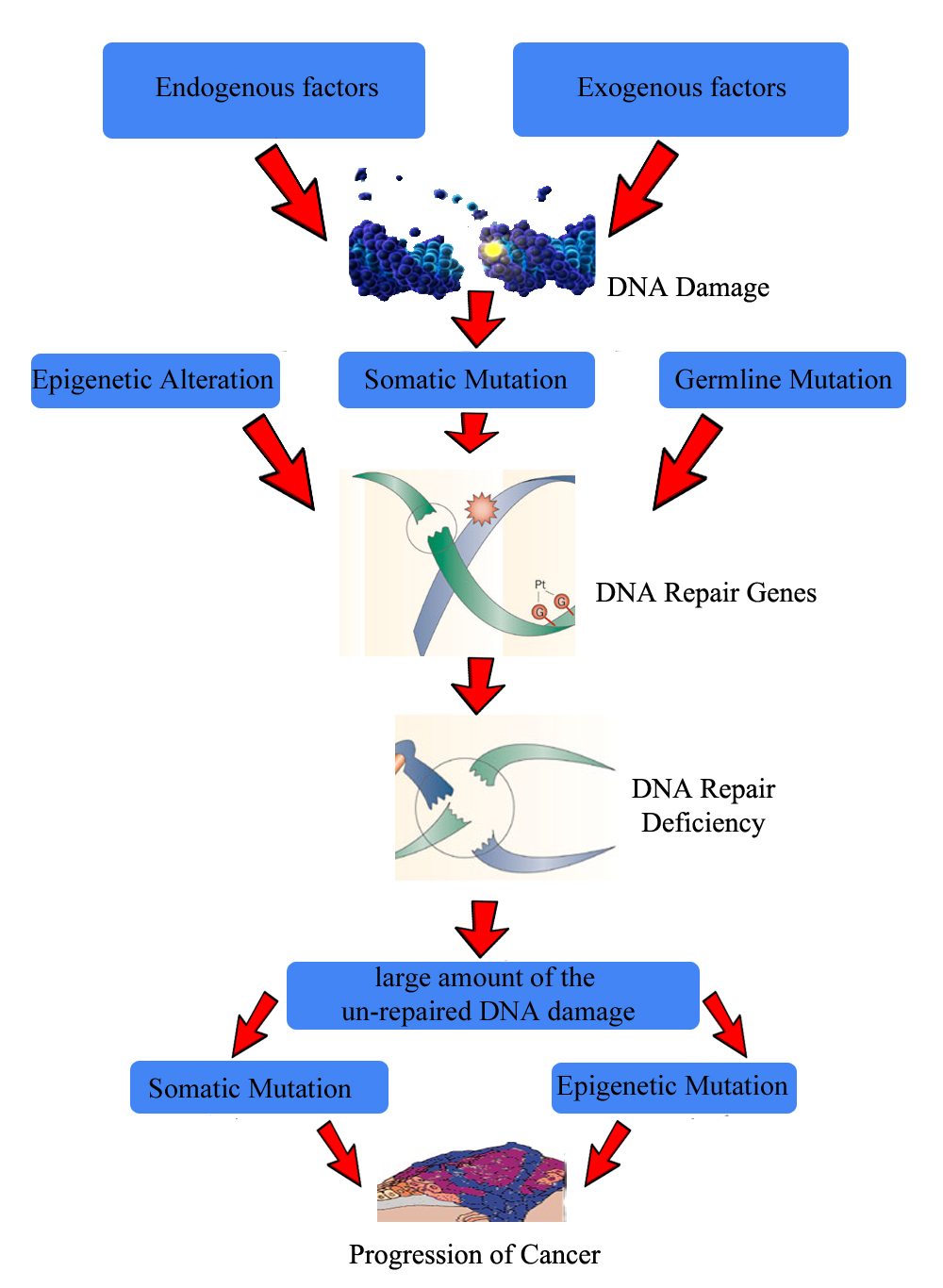
Figure 5. Epigenetic pathology of cancer (Kanwal et al., 2012)
In the last few decodes number of techniques has been used to prevent somatic and genetic mutation, as well as targeting the hyper immune cells but all have failed to suppress the cancer growth. Recently, some of the antibodies are targeted to specific cells, technique is CAR-T therapy. CARs are the receptors which help in the binding of the specific antibody to a T cell with the help of coding sequence placed inside the retrovirus (Poirot et al., 2015). Adoptive cell transfer is also successfully treated the cancer using CARs. The T-cells from the patient are collected and then they are modified in such way so that they can recognize the cancer cells and kill them. The CARs is having three domains i.e. ectodomain, transmembrane domain and endo domain (Kenderian et al., 2017). The ectodomain is present outside the cell, exposed to the extra cellular space and extra cytoplasmic. The ectodermal domain is exposed to the extracellular space as like cell surface receptor. In the endoplasmic reticulum the protein is leaded by signal peptide. The scFv is usually an antigen recognition region although many other regions also exist (Kershaw et al., 2013). Anything having high affinity to the target site can be used as an antigen recognition region. The link between the antigen binding domain and transmembrane domain is spacer region (Richter et al., 2017).So the spacer regions of antigen binding domain help to recognize and make link with antigen recognition for e.g. IgG1. The transmembrane domain extents the membrane. The functional ends of the receptor behave as endodomain (Zani et al., 2015; Zhang et al., 2017) (Figure 6).
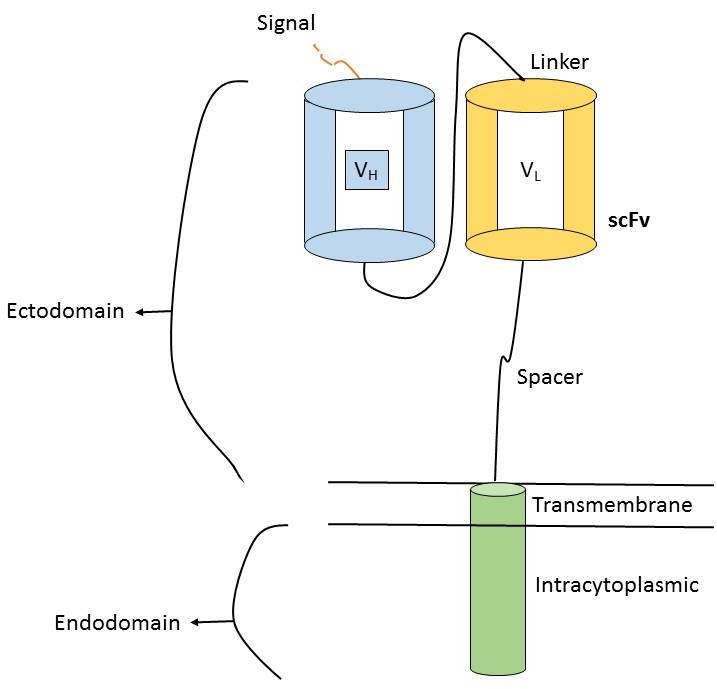 Figure 6. Chimeric Antigen Receptor (Zhang et al., 2017)
Figure 6. Chimeric Antigen Receptor (Zhang et al., 2017)
Gene therapy for leukemia treatment
Leukemia is group of cancers which starts in the bone marrow and results in proliferation of white blood cells that are known as leukemia cells (Petrella et al., 2015). The common symptoms are bleeding and bruising problems with slight fever and tiredness. There is also increased risk of infection after the minor sickness of exposure toward non-healthy environment. The causes of the leukemia are ionization radiation, smoking, Down syndrome and prior chemotherapy. Leukemia have four types like acute lymphoblastic leukemia (ALL), chronic lymphocytic leukemia (CLL), acute myeloid leukemia (AML) and chronic myeloid leukemia (CML)(Modak et al., 2014). The diagnosis of the leukemia includes complete blood counts and bone marrow examination as per the symptoms. It is not necessary that the blood tests confirm that the person is having leukemia and it’s most common in the early stage. In some situations a lymph node biopsy is too necessary to diagnose certain types of leukemia (Soni et al., 2015). MRI, X-ray or ultrasound also use to check the hurdles which are arise due to leukemia. The lymph nodes present in the chest will be checked by non-inventive the CT scanning (Latifi et al., 2015).
The gene therapy for the leukemia treatment is considered as the first gene therapy. The patients with refractory B-cell precursor acute lymphoblastic leukemia (ALL) are treated with tisagenlecleucel (Wells et al., 2017). Tisagenlecleucel is a technique which includes a chimeric antigen receptor(CAR) T-cell therapy in which the patient specific immune cells is used to treat the leukemia. The CAR-T cells are the recombinant cells which are having tumor antigen specific antibodies. The approval for the kymriah was firstly given by FDA in July 2017 (Bach et al., 2017). CAR-T cells have three parts: a transmembrane domain, extracellular antigen recognition domain of the (single-chain variable fragment) scFV and an intracellular T cell activation domain of CD3ζ. The CAR-T therapy targets specifically CD19-positive ALL. In CAR-T therapy the patient’s T cells are collected and genetically modify them with a receptor which will specifically target CD19 that are present on the surface of the cancer cells (Olweus, 2017). After modification these cells are transferred to the patient to kill the malignant cells in a specific area without any toxicity. For the CAR-T therapy the patient should have the age of 25 or above. The trails of 63 pediatrics and adult patients with ALL leads to its approval. The overall remission rate was about 83%- 84% after 3 months of treatment with Kymriah (Hallek, 2014). The Tisagenlecleucel is having a boxed warning for CRS that is a response which can activate the proliferation of CAR-T cells leading to high fever and flu like symptoms. To treat the severe CRS, FDA has approved Tocilizumab, which is commonly known as Actemra in patients aged 2 year or above (Valecha et al., 2017). The approval of tocilizumab was based on the treatment of the 69% patients from CRS within 2 weeks by giving 1 or 2 doses of drug. Kenneth C Anderson, MD, American Society of Hematology president, of the Dana-Farber Cancer Institute in Boston said “The approval of CAR T-cell therapy for pediatric leukemia marks an important shift in the blood cancer treatment paradigm” (Ding et al., 2017). CAR-T therapy has been approved after evaluating the risk and mitigation strategy that requires hospitals and clinics for safe and effective delivery of drug. The health professional staff should be trained with the proper knowledge of recognizing the CRS and neurological event. Recently, a drug is developed by Novartis for the treatment or malignancy but its treatment cost for a Patient is $475,000 (Brower et al., 2017; Zhao L et al., 2018) (Figure 7).
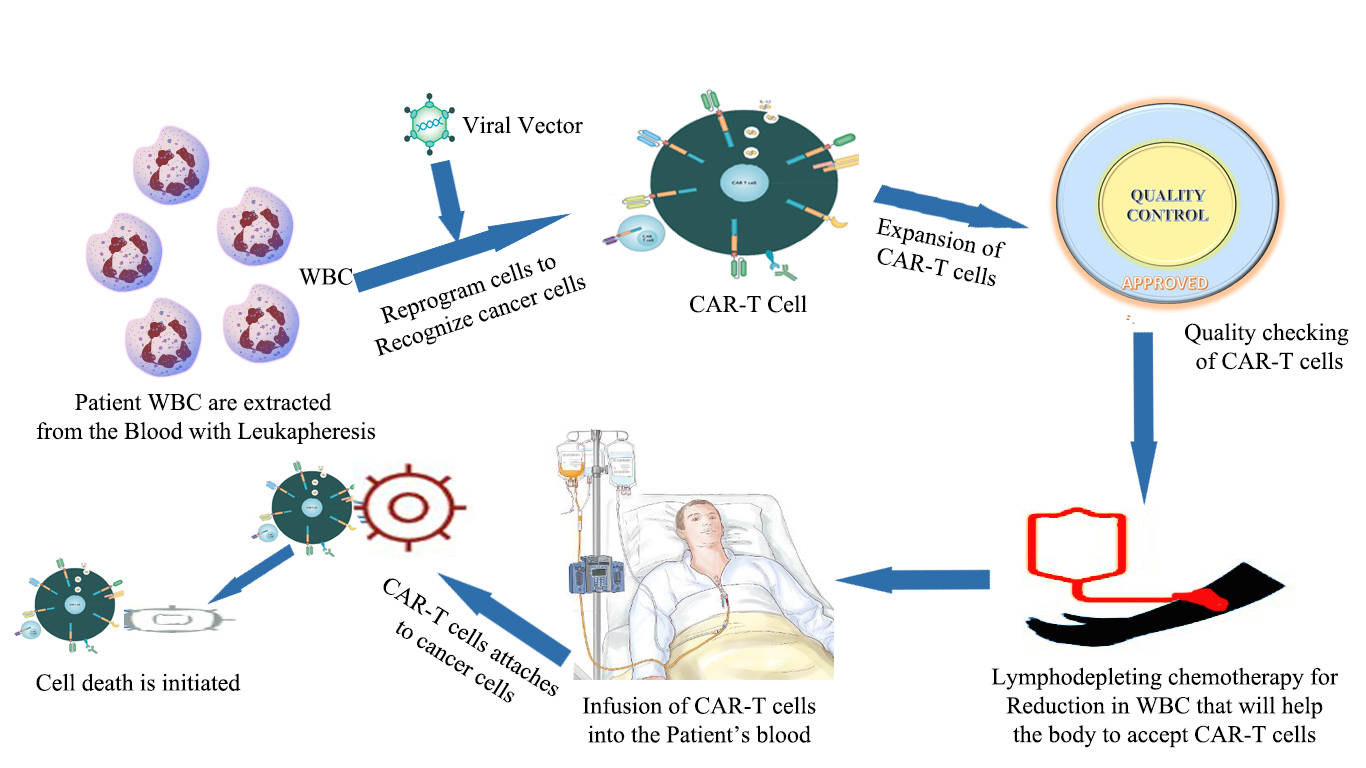 Figure 7. CAR-T Therapy (Zhao et al., 2018)
Figure 7. CAR-T Therapy (Zhao et al., 2018)
Conclusion
Now a day’s gene therapy is an efficient technique used to treat various types of disorders like monogenic, polygenic and infectious diseases. However, from the last few years the cancer is the major illness infecting the mankind. The leukemia commonly known as blood cancer is a silent and running with a big mortality rate. Basically the over proliferation of hematopoietic cells give birth to leukemia. Presently, gene therapy for leukemia includes the kymriah treatment which was recently approved by the FDA in July 2017. Its foremost target is CD19 ALL in a specific way with negligible chances of organ toxicity. In this therapy the WBC of the patients are collected and modified and further use to kill cancerous cell in his own body. So due to less side effect of kymriah as compared to the other medications it is using as lifesaving therapy because it kill only the leukemia cells without damaging to the normal cells functioning.
Clinical Implications
The CAR-T Therapy is based on the adoptive cell transfer technique. In starting, CAR-T therapy was restricted to some of the small trails but now days it is being used for the treatment of acute lymphoblastic Leukemia and lymphomas.
Acknowledgement
This work was supported by Dr. Shamsher Singh, ISF College of Pharmacy, Moga, Punjab and author is very thankful to Dr. Shamsher Singh.
Conflict of interest
All the authors are not having any conflicts of interest in this article.
References
Bach PB, Giralt SA, Saltz LB. 2017. FDA Approval of Tisagenlecleucel: Promise and Complexities of a $475 000 Cancer Drug. JAMA, 318(19):1861-1862.
Bartek J, Fornara O, Merchut‐Maya JM, Maya‐Mendoza A, Rahbar A, Stragliotto G,Broholm H, Svensson M, Sehested A, Naucler CS, Bartkova J. 2017. Replication stress, DNA damage signalling, and cytomegalovirus infection in human medulloblastomas. Molecular Oncology, 35-48.
Bradner JE, Hnisz D,Young RA. 2017. Transcriptional addiction in cancer. Cell 168(4):629-643.
Brower V. 2017. First Chimeric Antigen Receptor T-Cell Therapy Approved. Journal of Clinical Oncology, 35(7509):1200-1213.
Crawford C, Beason-Abmayr B, Eich E, Scott J, Nichol C. 2014. Going Viral. The Science Teacher, 81(6):51.
Crean CM, Mills KI, Savage KI. 2017. The Potential of Targeting DNA Repair Deficiency in Acute Myeloid Leukemia. Journal of Cancer Therapy, 8(08):691-704.
Davidson SM, Vander Heiden MG. 2017. Critical Functions of the Lysosome in Cancer Biology. Annual Review of Pharmacology and Toxicology, 57:481-507.
Ding J, Fishel ML, Reed AM, McAdams E, Czader M, Cardoso AA, Kelley MR. 2017. Ref-1/APE1 as Transcriptional Regulator and Novel Therapeutic Target in Pediatric T-cell Leukemia. Molecular Cancer Therapeutics, molcanther-0099.
Finer M, Glorioso J. 2017. A brief account of viral vectors and their promise for gene therapy. Gene therapy, 24(1):1-2.
Ginn SL, Alexander IE, Edelstein ML, Abedi MR, Wixon J. 2013. Gene therapy clinical trials worldwide to 2012–an update. Journal of Gene Medicine, 15(2):65-77.
Goetsch AL, Kimelman D, Woodruff TK. 2017. Malignant Hematologic Diseases.InFertility Preservation and Restoration for Patients with Complex Medical Conditions. Springer International Publishing, pp 39-60.
Grossman D, Leffell DJ. Wolff K. 2008. Squamous cell carcinoma. Fitzpatrick’s Dermatology in General Medicine, 7th edition. McGraw Hill Medical USA, pp 1028-36.
Hallek M. 2017. Chronic lymphocytic leukemia: 2017 update on diagnosis, risk stratification, and treatment. American Journal of Hematology, 92(9):946-965.
Hardee CL, Arévalo-Soliz LM, Hornstein BD, Zechiedrich L. 2017. Advances in non-viral DNA vectors for gene therapy. Genes, 8(2):1-13.
Ibrahim E, Elkhodary A, Elgazzar AH. 2015. Nuclear Oncology 1: Principles of Tumor Pathology and Biology. In The Pathophysiologic Basis of Nuclear Medicine Springer International Publishing, 347-367.
Jeon DG, Kim MS, Cho WH, Song WS, Lee SY. 2007. Pasteurized autograft–prosthesis composite for distal femoral osteosarcoma. Journal of Orthopaedic Science. 12(6):542-9.
Kaji EH, Leiden JM. 2001. Gene and stem cell therapies. JAMA, 285(5):545-50.
Kanwal R, Gupta S. 2012. Epigenetic modifications in cancer. Clinical Genetics, 81(4):303-11.
Kenderian SS, Porter DL, Gill S. 2017. Chimeric antigen receptor T cells and hematopoietic cell transplantation: how not to put the CART before the horse. Biology of Blood and Marrow Transplantation, 23(2):235-246.
Kershaw MH, Westwood JA, Darcy PK. 2013. Gene-engineered T cells for cancer therapy. Nature Reviews Cancer, 13(8):525-541.
Kumar D. 2017. Pathology of Bone and Soft Tissue Sarcomas. Springer International Publishing, pp 23-41.
Latifi F, Hosseini R and Mazinai M. 2015. A fuzzy expert system for diagnosis of acute lymphocytic leukemia in children. International Journal of Information, Security and Systems Management, 4(2):424-429.
Liu F, Gong J, Huang W, Wang Z, Wang M, Yang J, Wu C, Wu Z, Han B. 2014. MicroRNA-106b-5p boosts gliomatumorigensis by targeting multiple tumor suppressor genes. Oncogene, 33(40):4813-4822.
Merlo LM, Pepper JW, Reid BJ, Maley CC. 2012. Cancer as an evolutionary and ecological process. Nature Reviews Cancer, 6(12):924.
Mileski M, Ayala L, Campuzano E, Joy A, Ornelas S, Ortiz M, Saenz, J.2017. Quality of LifeConsiderations During Cancer Treatment in Invasive Ductal Carcinoma Patients: A Systemic Review. ABNF Journal, 28(1):1-9.
Modak H, Kulkarni S, Kulkarni SS, Gai P, Hallikeri U, Gai PB. 2014. Microarray based comparative genome-wide expression profiling of major subtypes of leukemia. African Journal of Biotechnology, 13(10):1-11.
Naldini L. 2015. Gene therapy returns to centre stage. Nature, 526(7573): 351-360.
Neureiter D, Jäger T, Ocker M, Kiesslich T. 2014. Epigenetics and pancreatic cancer: pathophysiology and novel treatment aspects. World Journal of Gastroenterology, 20(24):7830-7843.
Olweus J. 2017. Manufacture of CAR-T cells in the body. Nature Biotechnology, 35(6):520-521.
Oosterhuis J, Wolter, Leendert HJ Looijenga. 2017. Germ Cell Tumors from a Developmental Perspective: Cells of Origin, Pathogenesis, and Molecular Biology (Emerging Patterns). Pathology and Biology of Human Germ Cell Tumors. Springer Berlin Heidelberg, pp 23-129.
Ow SH, Chua PJ, Bay BH. 2017. Epigenetic regulation of peroxiredoxins: Implications in the pathogenesis of cancer. Experimental Biology and Medicine 242(2):140-147.
Petrella T, Bagot M, Willemze R, Beylot-Barry M, Vergier B, Delaunay M, Meijer CJ,Courville P, Joly P, Grange F, De Muret A. 2015. Blastic NK-Cell Lymphomas (AgranularCD4+ CD56+ Hematodermic Neoplasms) A Review. American Journal of Clinical Pathology, 123(5):662-675.
Poirot L, Duchateau P. Cellectis.2015. Method for in situ inhibition of regulatory t cells. U.S. Patent Application, 15/120: 060.
Prakash V, Moore M, Yáñez-Muñoz RJ. 2016. Current progress in therapeutic gene editing for monogenic diseases. Molecular Therapy, 24(3):465-474.
Rabinovich S, Yuan M, Wilson AJ, Phogat S, Lindsay RW, Chiuchiolo MJ, Parks CL. 2017. International Aids Vaccine Initiative. Recombinant viral vectors. U.S. Patent, 9(610):346.
Ramamoorth M, Narvekar A. 2015. Non viral vectors in gene therapy-an overview. Journal of Clinical and Diagnostic Research, 9(1):1-11.
Richter A, Niemoller M, Nolle V, Miltenyi S, Assenmacher M, MiltenyiBiotec Gmb H. 2017. Assay for Detection of Chimeric Antigen Receptor T Cells. U.S. Patent Application,15/417: 292.
Salani R, Billingsley CC, Crafton SM. 2014. Cancer and pregnancy: an overview for obstetricians and gynecologists. American journal of obstetrics and gynecology, 211(1):7-14.
Slivac I, Guay D, Mangion M, Champeil J, Gaillet B. 2017. Non-viral nucleic acid delivery methods. Expert Opinion on Biological Therapy, 17(1):105-118.
Soni G, Yadav KS. 2015. Applications of nanoparticles in treatment and diagnosis of leukemia. Materials Science and Engineering C, 47:156-164.
Stefani L, Galanti G, Klika R. 2017. Clinical implementation of exercise guidelines for cancer patients: Adaptation of ACSM’s guidelines to the Italian model. Journal of Functional Morphology and Kinesiology, 2(1):1- 4.
Swisher L, Patton KT, Thibodeau GA. 2017. Study Guide for The Human Body in Health &Disease-E-Book. Elsevier Health Sciences.
Taylor RB. 2017. Medical Words from Various Languages. In The Amazing Language of Medicine. Springer International Publishing, pp 53-76.
Tran E, Turcotte S, Gros A, Robbins PF, Lu YC, Dudley ME, Wunderlich JR, Somerville RP, Hogan K, Hinrichs CS, Parkhurst MR. 2014. Cancer immunotherapy based on mutation-specific CD4+ T cells in a patient with epithelial cancer. Science, 344(6184):641-645.
Valecha GK, Ibrahim U, Ghanem S, Asti D, Atallah JP, Terjanian T. 2017. Emerging role of immunotherapy in precursor B-cell acute lymphoblastic leukemia. Expert Review of Hematology, 10(9):783-799.
Vineis P, Wild CP. 2014. Global cancer patterns: causes and prevention. The Lancet, 383(9916):549-557.
Wells J, Jain N, Konopleva M. 2017. Philadelphia chromosome-like acute lymphoblastic leukemia: progress in a new cancer subtype. Clinical Advances in Hematology & Oncology, 15:554-561.
Zani IA, Stephen SL, Mughal NA, Russell D, Homer-Vanniasinkam S, Wheatcroft SB, Ponnambalam S. 2015. Scavenger receptor structure and function in health and disease. Cells, 4(2):178-201
Zhang C, Liu J, Zhong JF, Zhang X. 2012. Engineering CAR-T cells. Biomarker Research, (1):22.
Zhao L, Li J, Liu Y, Kang L, Chen H, Jin Y, Zhao F, Feng J, Fang C, Zhu B, Ding S. 2018.Cholesterol Esterification Enzyme Inhibition Enhances Antitumor Effects of Human Chimeric Antigen Receptors Modified T Cells. Journal of Immunotherapy, 41(2):45-52.