

Rajesh Prasad, Surya Bali Prasad*
Cell and Tumor Biology Laboratory, Department of Zoology, North-Eastern Hill University, Umshing-Mawkynroh, Shillong-793022, India
*Address for Corresponding Author:
Surya Bali Prasad
Cell and Tumor Biology Laboratory,
Department of Zoology, North-Eastern Hill University, Umshing-Mawkynroh, Shillong-793022, India
Abstract
The name ‘Rutin’ has been obtained from the source plant Ruta graveolens. Rutin is a flavonoid found in a wide variety of plants and used as nutraceutical agent. It is nontoxic and naturally present in various food products of plant origin especially buckwheat seeds, apricots, tea, cherries, grapes, grapefruit, onion, plums, and oranges. Rutin is also known as vitamin P or rutoside and has been explored extensively for its various biological effects. This review describes the recent information details on the chemistry, pharmacology and various biological effects such as anticancer, antioxidant, antidiabetic, anti-inflammatory, antibacterial, antifungal, neuroprotective, cardioprotective, hepatoprotective, nephroprotective and haematoprotective properties, etc. of rutin with its possible mechanism(s). The anticancer effects of rutin may involve induction of apoptosis and arrest of cell cycle progression in cancer cell lines. Along with anticancer activity, chemopreventive and chemosensitizing properties of rutin were also found. Various studies have shown that rutin administration improved the antioxidant defense system by elevating antioxidant biochemicals/enzymes such as glutathione, superoxide dismutase, catalase etc. and antioxidant genes, and reducing the production of lipid peroxidation, reactive oxygen species, nitric oxide and pro-inflammatory cytokines interleukin-6 in the hosts which may be involved in its protective ability against different toxicities. Rutin also modulates several neuroprotective genes, including tyrosine hydroxylase and suppresses caspase-3 and ameliorates the levels of neuroprotective factors in the diabetic retina. The details of various biological properties of rutin depicted in this review establish its vital importance and provide further scope to use this nutraceutical compound to explore for more therapeutic benefits for human.
Keywords: Rutin, nutraceutical, pharmacology, biological properties
Introduction
Nature is an attractive source of new therapeutic agents and a variety of phytochemicals have proved to be very valuable in the prevention and treatment of various diseases. Many of the important drugs isolated from plants during the past 50 years have revolutionized modern medicinal practice (Dar et al., 2017). The use of herbal medicines and phytonutrients or nutraceuticals continues to expand rapidly across the world with many people now resorting to these products for the treatment of various health challenges in different national healthcare settings (WHO, 2004). It is estimated that up to 80% of the world’s population living in the developing world rely on herbal medicinal products as a primary source of healthcare and traditional medical practice as an integral part of the culture in their communities (Ekor, 2013). The medicinal compounds derived from various plants could be divided in to three major biochemical classes: alkaloids (vinblastine, vincristine, pilocarpine, berberine, caffeine, piperine etc.), flavonoids (quercetin, kaempferol,chrysin, naringenin, genistein, rutin etc.) and terpenoids (artemisinin, taxol, digitoxin, azadarachtin, camphor, limonene etc.) (Takshak, 2018). These compounds have also gained importance in the area of nutraceuticals, which have positive health effects and may prove to be advantageous in the treatment of diseases like cancer, cardiovascular diseases and diabetes (Crozier et al., 2006). Nutraceuticals are products derived from food sources that are purported to provide extra health benefits, in addition to the basic nutritional value found in foods.
Flavonoids, a group of low molecular weight polyphenolic substances, have attracted the attention of researchers because of their beneficial effects such as antibacterial, antiviral, anticancer, immune-stimulant, and antioxidant effects (Tiwari, 2017). More than 4,000 varieties of flavonoids have been found in herbs, vegetables, fruits, and beverages (Guardia et al., 2001). Chemically, the core structure of flavonoids is based upon a C6-C3-C6 (diphenyl propane structure) skeleton in which the three-carbon bridge is usually cyclized with oxygen. These compounds are considered as chemotaxonomic markers according to the biosynthesis pathway (a combination of phenylalanine with three malonyl‐CoA units to form a C‐15 chalcone), and they provide attractive color pigments such as yellow, red, blue, and purple in plants. The chemical nature of flavonoids depends on the degree of unsaturation and oxidation of the three-carbon chain. There are six main classes of flavonoid found in higher plants: i)flavones e.g. apigenin and chrysin, ii)flavonols i.e. quercetin and rutin, iii) flavanones i.e. hesperidin and naringenin, iv) flavanonol i.e. aromadedrin and taxifolin, v) isoflavones i.e. genistein and daidzein, and vi) flavan-3-ols i.e. catechin and gallocatechin (Rana and Gulliya, 2019).
The present review focuses on the collective information covering the chemistry, pharmacology and health-promoting effects of a type of flavonoid i.e. rutin as nutraceutical agent and its future prospects.
The name ‘rutin’ has been drawn from the plant Ruta graveolens (common Rue) (Figure 1) whose aerial parts contain rutin.

Figure 1. A- Pants of Ruta graveolens, B- its flowers, C- its leaves
Rutin is a kind of flavonoid glycoside, also known as vitamin P or purple quercitrin, present in more than seventy plants species and food products of plant origin especially buckwheat seeds, apricots, cherries, grapes, grapefruit, onion, plums, and oranges (Kreft et al., 1997; Calzada et al., 2018) was first discovered in buckwheat (Fagopyrum esculentum) in the 19th century (Yang et al., 2008). The leading plants containing up to 1.5% of rutin include Ruta graveolens L. (Rutaceae), Sophora japonica L. (Fabaceae), Maranta leuconeura E. Morren (Marantaceae), Orchidantha maxillarioides (Ridl.) Schum (Lowiaceae), Strelitzia reginae Banks ex Aiton (Strelitziaceae), Eucalyptus spp. (Myrtaceae), Canna indica L. (Cannaceae), Canna edulis Ker Gawl. (Cannaceae), and Labisia pumila (Blume) Mez (Primulaceae) (Calzada et al., 2018). Other plants and their parts commonly used for isolation of rutin are given in table 1.
Chemistry and pharmacology of rutin
Rutin (3, 3ʹ, 4ʹ, 5, 7-pentahydroxyflavone-3-rhamnoglucoside) (Figure 2) also called as rutoside, quercetin-3-rutinoside or sophorin is a flavonol category of flavonoid (Ganeshpurkar and Saluja, 2017).
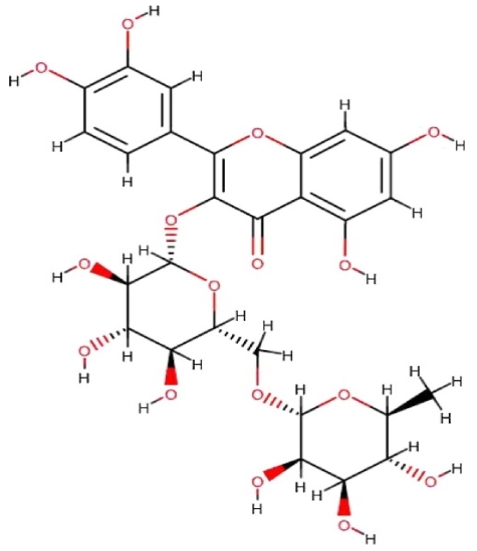
Figure 2. Chemical structure of rutin
Table 1. Different species of plants and their parts used for isolation of rutin
|
S. No |
Plant species |
Common name |
Indian name |
Rutin content (mg /100g) (Approx.) |
Part of plants used |
Reference |
|
1. |
Capparis spinosa, C. sicula and C. orientalis |
Capers |
Kabra |
332 |
flower buds |
Inocencio et al., 2000 |
|
2. |
Oleaeuropaea L. |
Olive |
Jaitoon |
45 |
fruits |
Romani et al., 1999 |
|
3. |
Fagopyrum esculentum Moench |
Buckwheat |
Kotu, Kuktu, Phaphra |
36 |
whole grain flour |
Dietrych-Szostak and Oleszek, 1999 |
|
4. |
Asparagus officinalis |
Sparrow grass |
Shatavari |
23 |
asparagus spears (stem) |
Makris and Rossiter, 2001 |
|
5. |
Rubus occidentalis |
Black raspberry |
|
19 |
Fruits |
Wada and Boxin, 2002 |
|
6. |
Rubus idaeus |
Red raspberry |
Rasabharii. |
11 |
Fruits |
Wada and Boxin, 2002 |
|
7. |
Fagopyrum esculentum Moench |
Buckwheat |
Kotu, Kuktu, Phaphra |
9 |
Groats (thermally treated) |
Dietrych-Szostak and Oleszek, 1999 |
|
8. |
Fagopyrum esculentum Moench |
Buckwheat |
Kotu, Kuktu, Phaphra |
6 |
Seeds |
Kreft et al., 1999 |
|
9. |
Ribes nigrum, Rhus chirindensis, Ribes rubrum |
Berries |
Jaamun |
6 |
Fruits |
Maatta et al., 2003 |
|
10. |
Prunus domestica, Prunus Mexicana, Prunus domestica subsp. syriaca |
Plums |
Ber |
6 |
Fruits |
Kim et al., 2003 |
|
11. |
Ribes nigrum |
Berry |
Jaamun |
5 |
Fruits |
Maatta et al., 2003 |
Chemically, rutin is a glycoside combining the flavonol quercetin with the disaccharide rutinose (rhamnose and glucose) (Sharma et al., 2013). Various physicochemical properties of rutin have been summarized in Table 2.
Table 2. The physicochemical properties of rutin (https://en.wikipedia.org/wiki/Rutin)
|
Chemical formula |
C27H30O16 |
|
Molar mass |
610.52 g·mol−1 |
|
Form and color |
Powder, yellow to green |
|
Melting point |
242 °C |
|
Solubility in water |
12.5 mg/100 mL |
|
Site of absorption |
Intestine (Ou-yang et al., 2013; Andlauer et al., 2001) |
|
Excretion |
Urine (about 10%); unchanged in faeces (rest) (Choudhury et al.,1999) |
Rutin is a common dietary flavonoid (Yang et al., 2008) and little or no dietary rutin is absorbed intact since gut microflora metabolizes it to a variety of compounds that may be absorbed. Microflora of the lower gut hydrolyzes rutin to its aglycone, quercetin, and the sugar residue, which are subsequently absorbed by the small intestine wall (Koval’skii et al., 2014). After peroral administration to rats and rabbits, the metabolism of rutin shows the presence of three rutin metabolites i.e. 3, 4-dihydroxyphenylacetic acid, 3-methoxy-4-hydroxyphenylaceticacid, and 3-hydroxyphenylacetic acid in the urine (Chen et al., 2005; Yang et al., 2005). Another metabolite, quercetin-3-omethyl ether is detected in bile for i.p. administration of rutin to rats (Graf et al., 2006). In a study in humans it has been noted that little or no dietary rutin is absorbed because gut microflora in the large intestine metabolize rutin to a variety of compounds that include quercetin and phenol derivatives such as 3, 4-dihydroxyphenylacetic acid, 3-hydroxyphenylacetic acid, 3-methoxy-4-hydroxyphenylacetic acid (homovanillic acid) and3, 4-dihydroxytoluene and they are devoid of significant antioxidant activity (Pashikanti et al., 2010). However, the level of absorption of quercetin is quite low as was evident from the findings that when doses of 50 mg of either rutin or quercetin were used in the diet of healthy humans, plasma levels of quercetin was found to be only 3.5 μmol/L (Erlund et al., 2000). Moreover, up to 50% of an ingested dose of 75 mg rutin was recovered as microbial metabolites from urine in human (Sawai et al., 1987). Glycation is a nonenzymatic condensation reaction between reducing sugars and amino groups of proteins that undergo rearrangements to stable ketoamines, leading to the formation of advanced glycation end products (AGEs) including argpyrimidine and Nε-carboxymethyllysine protein adducts and protein cross-links, and correlate with processes resulting in ageing and diabetes complications (Pashikanti et al., 2010). Rutin metabolites, 3, 4-dihydroxyphenylacetic acid and 3, 4-dihydroxytoluene which formed in the gut from dietary rutin consumed in the form of a variety of fruits and vegetables have been shown to be potent inhibitors of AGEs. Thus, it has been suggested that fruit- and vegetable-rich diets contribute to the prevention of processes resulting in ageing and diabetes complications and rutin consumption has been correlated with good health (Pashikanti et al., 2010).
Biological effects of rutin
Rutin, a common dietary flavonoid usually nontoxic naturally derived compound exhibits a diverse range of useful biological properties such as anticancer, antioxidant, antidiabetic, anti-inflammatory, antibacterial, antifungal, neuroprotective, cardioprotective, hepatoprotective, nephroprotective, haematoprotective, antiarthritis, anthelmintic, testicular protection, etc. (Ren et al., 2003; Ganeshpurkar and Saluja, 2017). The brief details of the main biological properties of rutin are described below.
Anticancer property
Cancer is a multifactorial genetic disease that results from the accumulation of various mutations resulting in uncontrolled tumor cell proliferation eventually spreading through healthy tissues. Flavonoids are among the most studied anticancer and cancer preventing drugs. Various studies have well established the anticancer, chemopreventive and chemosensitizing property of rutin against a variety of cancers. Lin et al., (2012) demonstrated that rutin administered once i.p. per 4 days at 120 mg/kg was effective in reducing the growth of human leukemia HL-60 tumors in a xenograft mouse model. Chen et al. (2013) demonstrated the anticancer effect of rutin against human neuroblastoma LAN-5 cells. This anticancer activity was found to involve rutin-mediated G2/M phase cell cycle arrest and triggering apoptosis in LAN5 cells. It has been reported that rutin induces high cytotoxic effects in vitro against Human colon adenocarcinoma SW480 cells and also induces in vivo antitumor and anti-angiogenic effects, with no toxic effects on nude mice bearing SW480 tumors (Alonso-Castro et al., 2013). Rutin has also been shown to inhibit spleen leukemia tumor growth in a WEHI-3 leukemia murine model and promote immune response associated with the increase in the activity of macrophage phagocytosis (Lin et al., 2009).The antitumor activity of rutin-cisplatin combination against malignant murine ascites Dalton’s lymphoma has been reported with increased cytotoxicity and apoptosis in DL cells resulting in increase in hosts survivability. The treatment also caused a decrease in glutathione in tumor cells (Prasad and Prasad, 2018). Calzada et al. (2018) have reported that rutin obtained from Schinus molle showed anticancer activity against human leukemic monocyte lymphoma U‑937 cell line. Rutin isolated from the methanolic extract of Triticum aestivum straw has shown the anticancer and chemopreventive effect of rutin in dimethylbenz[a]anthracene (DMBA) and croton oil-induced skin carcinogenesis in Swiss albino mice (Dixit, 2014). In a different type of study, rutin encapsulated in low methoxyl pectin beads was found to exhibit anti-cancer activity higher than non-encapsulated rutin against 3 human cancer cell lines i.e. human colon adenocarcinoma (HT-29), human mouth epidermal carcinoma (KB) and hepatocellular carcinoma (HepG2) cells (Jantrawut et al., 2014). Rutin has also been used as a chemosensitizer for different cancer cell lines to increase the anticancer effect of different drugs on chemosensitize cells. Rutin was shown to sensitize human breast cancer cells to cyclophosphamide and successfully arrest cell cycle progression (Iriti et al., 2017). Quercetin as one of the main components of rutin has also shown the anticancer and apoptosis-inducing effects in vitro in nine tumor cell lines and in mice bearing MCF-7 and CT-26 tumors shown anticancer activity (Hashemzaei et al., 2017). Vadapalli et al. (2017) reported that rutin exhibited anti-proliferative activity on HeLa cell line by modulating haematological parameters, lipid peroxidation and augmenting antioxidant defense system in HeLa cell line induced cervical cancer in rats. Recently, it was found that rutin and orlistat exert an in vivo anticancer activity against Ehrlich ascites carcinoma (EAC) and caused a decrease in carcinoembryonic antigen level, cholesterol content, fatty acid synthase expression and exerted antioxidant action with reduced malondialdehyde level and increased glutathione content. Further, both the drugs were cytotoxic to breast cancer cell lines MCF-7 and the pancreatic cancer cell line, Panc-1 in vitro by promoting apoptosis (Saleh et al.,2019). Rutin has also been demonstrated for chemopreventative activity in many animal model such as azoxymethane-induced colonic tumorigenesis in mice and rats (Mahmoud et al., 2000), dimethylbenz[a]anthracene (DMBA) and N-nitrosomethylurea treated mammary glands of rats and DMBA-treated skin of cancer (Verma et al., 1988).
Antioxidant property
Antioxidants are compounds that inhibit oxidation of other molecules and balance the oxidative state in the cells. They can prevent or slow damage to cells caused by free radicals, unstable molecules that the body produces as a reaction to environmental and other factors. They are sometimes also called as "free-radical scavengers." It is well-known that excessive free radical production and lipid peroxidation in vivo may cause many kinds of diseases. Due to anti-oxidative nature, rutin shows a wide range of pharmacological actions and has been used as protective agent in a number of diseases. Yang et al., (2008) using different assays analyzed the antioxidant activity of rutin and found that it has powerful antioxidant capacity against various antioxidant systems exhibiting strong radical scavenging activity as well as effective inhibition of lipid peroxidation. The in vivo study showed that rutin administration improved the antioxidant defense systems against iron overload-induced hepatic oxidative stress in rats. This protective effect in liver of iron-loaded rats may be due to both antioxidant and metal chelation activities. (Aziza et al., 2014).The finding suggested that the antioxidant capacity of rutin may be attributed to its electron or hydrogen donating power orhydroxyl scavenging ability (Kaur et al., 2015). Rutin’s ability to reduce reactive oxygen species (ROS) generation and enhanced activity/levels of various antioxidants in the cells has also been reported (Gegotek et al., 2017). Wang et al.(2012) demonstrated that rutin acted as a multifunctional agent by inhibiting β-amyloid aggregation and cytotoxicity, preventing mitochondrial damage, reducing the production of malondialdehyde (MDA), ROS, nitric oxide(NO),glutathione disulfide(GSSG), inducible nitric oxide synthase (iNOS), and pro-inflammatory cytokines, and increasing catalase, superoxide dismutase, reduced glutathione (GSH), and glutathione peroxidase levels. Patil et al. (2017) reported the protective effect of rutin and quercetin against radiation‑induced DNA damage in human lymphocytes, which may be attributed to antioxidant capacity of rutin involving scavenging of radiation‑induced free radicals and also by the inhibition of radiation‑induced oxidative stress.
Antidiabetic property
Diabetes is a chronic disease that occurs either when the pancreas does not produce enough insulin or when the body cannot effectively use the insulin resulting in hyperglycaemia, or raised blood sugar level which may lead to serious damage to many of the body's systems. World health organization (WHO) in its fact sheets on diabetes 2018 indicated that the global prevalence of diabetes is rising every year and estimated that diabetes was the seventh leading cause of death in 2016. Flavonoids present in vegetables and medicinal plants have beneficial effects on diabetes by improving glycemic control, lipid profile, and antioxidant status. The probable reason and mechanisms for the anti-hyperglycemic effect of rutin include a decrease of carbohydrates absorption from the small intestine, inhibition of tissue gluconeogenesis, an increase of tissue glucose uptake, stimulation of insulin secretion from pancreatic beta cells, and protecting Langerhans islet against degeneration (Sattanathan et al., 2011; Ghorbani, 2017). Rutin treatment improved histo-architecture of beta islets and reversed hypertrophy of hepatocytes and exhibited significant antidiabetic activity by inhibiting inflammatory cytokines, improving antioxidant and plasma lipid profiles (Niture et al., 2014). Antihyperglycemic property of rutin and its protective effects against the development of diabetic complications has been associated with a rutin-mediated decrease in the formation of sorbitol, reactive oxygen species, advanced glycation end-product precursors, and inflammatory cytokines (Ghorbani, 2017). Antidiabetic effect of rutin has been suggested to involve enhanced peripheral glucose utilization either by direct stimulation of glucose uptake or via the mediation of enhanced insulin secretion and by inhibiting the glucose transporter activity (Jadhav et al., 2012). It has been reported that rutin as the major constituent of Morus alba leaf extract plays a key role in in vivo anti-diabetic activity in type II diabetic rats (Hunyadi et al., 2012). The dietary supplementation of rutin has the potential for the prevention and treatment of Type 2 diabetes and to suppress oxidative stress-mediated damage in diabetic pathophysiology (Dhanya et al., 2014). The rutin-mediated decrease in blood glucose levels in hyperglycaemic rats is also associated with the restoration of the depleted serum antioxidants enzymes and antioxidant capacity in the hosts (Tanko et al., 2017). In fact, it has been found that flavonoids of all sub-classes including rutin have antidiabetic properties via regeneration of pancreatic β-cells and enhancing insulin secretion, enhancing insulin-mediated glucose uptake by target cells, inhibiting aldose reductase and increasing Ca2+ uptake (Marella, 2017).
Anti-inflammatory property
Inflammation is not a synonym for infection. Inflammation is part of the complex biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants, and is a protective response involving immune cells, blood vessels, and molecular mediators. The flavonoids intake has been reported to be effective in reducing the risk of chronic and cardiovascular inflammatory diseases (Knekt et al., 2002). It has been reported that rutin by its radical scavenging and anti-inflammatory effect enhanced the healing of ulcerative colitis (Hussein et al., 2014). In vitro and in vivo study have shown that rutin can be used as candidate therapeutic agent for the treatment of various severe vascular inflammatory diseases via inhibition of the high mobility group box 1 (HMGB1) protein signalling pathway which acts as a late mediator of severe vascular inflammatory conditions (Yoo et al., 2014). The evaluation of the anti-inflammatory effects of flavonoid-rich common buckwheat sprout (CBS) and tartary buckwheat sprout (TBS) showed the approximately 3.6-fold higher content of rutin in TBS extracts than that in CBS extracts. As compared to CBS extracts, the TBS extract exhibited higher inhibitory activity on the macrophage-mediated inflammatory disorders and production of proinflammatory mediators such as nitric oxide and cytokines including tumor necrosis factor-α, interleukin-6, and interleukin-12 in BALB/c mice (Nam et al., 2017). Enhancement of anti-inflammatory activity of encapsulated rutin with the inhibition of IL-6 and NF-kB has also been reported (Jantrawut et al., 2017).
Anti-bacterial property
Bacterial infections have played a central role in the lives and deaths of humans. Bacterial infections may develop many diseases such as pneumonia, tetanus, cholera, tuberculosis, bloodstream infections (sepsis) and sexually transmitted diseases like gonorrhoea. Antibacterial usually refers to an antibiotic, a principal type of antimicrobial agent used mainly against bacteria to kill or inhibit their multiplication. Singh et al. (2008) studied the antimicrobial activity of methanolic extracts Pteris vittata, commonly known as ‘Brake fern’, on the growth of eight intestinal microorganisms, and showed that 70% aqueous methanolic extract has potent activity against Pseudomonas aeruginosa and Klebsiella pneumonia and the favonoid rutin was identified in the extract. In another study, the extract of Castanea sativa with an aqueous solution of sulfuric acid showed pronounced antibacterial effects against different strains of Gram-positive and Gram-negative bacteria and these effects were similar to that of pure rutin, quercetin and apigenin (Basile et al. 2000). It has also been reported that different flavonoid compounds have better antibacterial activity when used in combination. The combination of quercetin and quercitrin, quercetin and morin, morin and rutin, and quercetin and rutin was much more active than either flavonoid alone against Salmonella enteritidis and Bacillus cereus (Arima et al., 2002). The study on the effect of rutin in combination with other flavonoids and antibiotics in methicillin-resistant Staphylococcus aureus (MRSA) showed that quercetin + morin + rutin with amoxicillin, ampicillin, cephradine, ceftriaxone, imipenem, and methicillin showed synergism, while additive relationship was indicated between morin + rutin and amoxicillin, cephradine, ceftriaxone, imipenem, and methicillin. Quercetin alone had an additive effect with these antibiotics (Amin et al., 2015). Streptococcus suis form biofilms and cause severe diseases including meningitis and streptococcal toxic shock-like syndrome in pigs and humans. Biofilms are communities of microbes embedded in a matrix of extracellular polymeric substances such as capsular polysaccharides (CPS). CPS is involved in its adherence to influence bacterial biofilm formation. It was noted that rutin interferes with CPS synthesis in S. suis and thereby decreases biofilm formation without impairing its growth in vitro. Thus, rutin could be used as a novel natural inhibitor of biofilm and the prevention of S. suis biofilm-related infections (Wang et al., 2017). Rutin has been suggested to be a natural active antibacterial agent as it has shown potent antimicrobial activity against Streptococcus pyogenes, Enterococcus faecalis, Bacillus cereus, Pseudomonas aeruginosa, Klebsiella pneumoniae, and Escherichia coli (Danciu et al., 2018). The honey produced by the stingless bee Melipona compressipes manaosensis showed antibacterial activity against Gram-positive and Gram-negative bacteria and rutin was identified in these honey samples. (Pimentel et al., 2013).
Antifungal property
An infection caused by a fungus (fungal infection) can develop different diseases such as candidiasis, aspergillosis, ringworm, blastomycosis, etc. An antifungal agent is a drug that selectively eliminates fungal pathogens and the development of antifungal agents has lagged behind that of antibacterial agents. The evaluation of the antifungal effect of the combination of a fungicidal substance amphotericin B, with quercetin or rutin, showed that this combination improves the antifungal activity of amphotericin B against Cryptococcus neoformans. The toxicity tests showed that quercetin protects red blood cells from amphotericin B-mediated hemolysis and toxicity. It was suggested that quercetin and rutin are potential agents to combine with amphotericin B in order to reduce the amphotericin dose to lessen side effects and improve antifungal efficacy (Oliveira et al., 2016). Rutin and aurapten isolated from Polygala paniculata plants common in Brazil has shown antifungal activity against Cryptococcus gattii (Johann et al., 2011). ). Han (2009) studied the effect of rutin on septic arthritis due to Candida albicans, a major etiological agent that causes fungal arthritis. It was found that after treatment approximately 45% of the edema was reduced at the peak day of septic arthritis, growth of C. albicans was inhibited and hemolysis was not observed, therefore, suggested that rutin has both anti-arthritic and antifungal effects. In another study rutin isolated from tobacco leaves revealed that rutin is a good antifungal antibacterial agent as it showed antifungal activity against Candida albicans and prominent antibacterial activity against Staphylococcus aureus, Bacillus subtilis. Escherichia coli and Klebsiella oxytoca, and suggested that phytochemicals such as rutin serve as important therapeutic agents (Dubey et al.2013). The study to assess the antifungal and antibacterial activity of the entire olive leaf aqueous extract showed high antifungal activity against Candida albicans and Cryptococcus neoformans, and antibacterial activity against B. cereus, B. subtilis, S. aureus (Gram +), E. coli, P. aeruginosa, K. pneumoniae (Gram -) bacteria. It was suggested that extracts could be more beneficial than isolated constituents since a bioactive individual component can change its properties in the presence of other compounds present in the extract. Rutin was identified as one of the seven phenolic compounds quantified in the extract. It was suggested that the use of olive leaves as nutraceuticals may lower the risk of microbial infections (Pereira et al., 2007).
Neuroprotective property
Neuroprotection refers to the relative preservation of neuronal structure and/or function and the neuroprotective potential of rutin has been widely studied. Neurodegenerative diseases such as Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, and multiple sclerosis are defined as disorders with loss and damages of neurons. It has been proven that chronic inflammation is a part of the cause of neurodegenerative diseases. Methanol and water extracts of above-ground tissues Glaucium corniculatum significantly inhibited pro-inflammatory IL-6 secretion and increased anti-inflammatory cytokine IL-10 secretion in a dose-dependent manner in neuronal pheochromocytoma PC12 cells. Rutin and quercetin flavonoids found to be as major constituents in the extract may involve in its neuroprotective effect (Koçanci et al., 2017). The neuroprotective properties of rutin have been well demonstrated against multiple disease states, including cancer, cardiovascular disease, ischaemia–reperfusion brain injury and neurodegenerative disorders (Pu et al., 2007). These therapeutic benefits of rutin have been proposed due to its antioxidant and free radical–scavenging properties. Rutin has been demonstrated to decrease inducible nitric oxide and cytokines activity in an experimental model of Alzheimer diseases. In a study to explore the neuroprotective mechanism of rutin against immobilization stress-induced anxiety-like behavioral and oxidative damage in mice showed that nitric oxide modulator L-NAME pre-treatment significantly potentiated the protective effect of rutin suggesting the involvement of nitric oxide mechanism (Machawal and Kumar, 2014). In a study designed to establish the neuroprotective role of rutin as well as to elucidate the antioxidant mechanism of rutin in 6-hydroxydopamine (6-OHDA)-induced toxicity in PC-12 neuronal cells, significant cytoprotective activity was observed in rutin pretreated cells in a dose-dependent manner. Furthermore, there was marked activation of antioxidant enzymes including superoxide dismutase, catalase, glutathione peroxidase, and total glutathione (GSH) in rutin pretreated cells compared to cells incubated with 6-OHDA alone. Rutin significantly reduced lipid peroxidation in 6-OHDA-induced PC-12 cells. On the basis of these observations, it was concluded that the bioflavonoid rutin inhibited 6-OHDA-induced neurotoxicity in PC-12 cells by improving antioxidant enzyme levels and inhibiting lipid peroxidation (Magalingam et al., 2013). Prion disorders are progressive neurodegenerative diseases characterized by extensive neuronal loss and accumulation of the abnormal form of the scrapie prion protein (PrP). Rutin treatment blocked PrP (106–126)-mediated increases in reactive oxygen species production and nitric oxide release and helped slowing the decrease of neurotrophic factors that results from PrP accumulation. Additionally, rutin treatment significantly decreased the expression of the death receptor Fas and its ligand Fas-L, thus, these results demonstrated that rutin protects against the neurodegenerative effects of prion accumulation and inhibits apoptotic pathway activation in neuronal cells (Na et al., 2014). It has been demonstrated that rutin pretreatment attenuated the expression of Parkin 5, Parkin 7, Caspase 3, Caspase 7, and Ataxin 2 gene expression that was highly expressed 6-hydroxydopamine (6-OHDA) treated neuronal PC12 rat pheochromocytoma cells. Moreover, rutin upregulated many neuroprotective genes, including tyrosine hydroxylase (Th), neuron-specific gene family member 1 (Nsg1), N-ethylmaleimide-sensitive factor (Nsf), and optic atrophy 1 homolog (Opa1) genes (Magalingam et al., 2015a; Magalingam et al. 2015b). Diabetes-induced oxidative stress is believed to be the central factor in damaging the retina. In the analysis of the neuroprotective effects of rutin, in the diabetic rat retina, it was found that rutin possesses antidiabetic activity, as blood glucose level decreased and insulin level increased in diabetic rats. In the diabetic retina, rutin supplementation enhanced the reduced levels of brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and glutathione (GSH), and reduced the lipid peroxidation. In addition, rutin treatment showed antiapoptotic activity by decreasing the level of caspase-3 and increasing the level of Bcl-2 in the diabetic retina. These results suggest rutin’s effectiveness to prevent the retinal damage and subsequently the development of diabetic retinopathy (Ola et al., 2015). Dexamethasone, a synthetic glucocorticoid receptor agonist, causes neuronal death in the CA3 layer of the hippocampus, which has been associated with learning and memory impairments. The study aimed to evaluate the neuroprotective effect of okra (Abelmoschus esculentus Linn.) extract and its derivatives quercetin and rutin okra in dexamethasone-treated mice showed that quercetin, rutin, and okra extract pre-treatments reversed cognitive memory deficits, including impaired dentate gyrus (DG) cell proliferation, and protected against morphological changes in the CA3 region in dexamethasone-treated mice. The pre-treatment with quercetin, rutin or okra also prevented dexamethasone-induced neuronal damage and changes in N-Methyl-D-aspartate (NMDA) -receptor positive cells in the hippocampal CA3 area thus, propose their neuroprotective effects (Tongjaroenbuangam et al., 2011). Cisplatin is a widely used chemotherapeutic agent for cancer treatment with limited uses due to its neurotoxic side effects. The neurotoxic effect of cisplatin was determined by measuring the lipid peroxidation, GSH and antioxidant genes levels in the brain tissue of the rat. Cisplatin treatment decreased the expression levels of paraoxonase-1 (PON-1), PON-3, peroxisome proliferator-activated receptor delta (PPAR-δ) and glutathione peroxidase (GPx) whereas significantly increased PON-2 expression levels in the brain of rats. The cisplatin treatment also significantly increased the lipid peroxidation and decline the GSH levels in the brain. However, co-administration of rutin with cisplatin prevented the cisplatin-induced toxicity by restoring the changes in lipid peroxidation, GSH level and the studied genes to normal values as in the control group (Almutairi et al., 2017). The effects of rutin on differentiated human neuroblastoma cells (IMR32) in vitro unveiled that the high (100 μM) and low (100 nM and 10μM) rutin concentrations significantly avert ROS generation by two different mechanisms, by enhancing apoptosis through the modulation of levels of Bcl2, Caspase-3, survivin and its antioxidant activity via stress-related proteins, c-jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (p38MAPK). The findings suggest that rutin is a multi-targeted therapeutic and preventive agent that may act as an adjuvant complementary therapeutic molecule to treat oxidative stress-mediated neurodegeneration (Banudevi et al., 2018). Fluorides have been known to induce neurotoxicity. The rats orally co-treated by gavage with rutin and sodium fluoride showed a reversal of the NaF- induced neurobehavioral deficits. Further, the co-treatment attenuated the NaF–induced inhibition of antioxidant enzymes and acetylcholine esterase activity and inhibits lipid peroxidation, neuro-inflammation, and apoptosis in the cerebrum and striatum of the rats, thus, rutin may be used as a neuroprotective agent against fluoride–induced neurotoxicity (Nkpaa and Onyeso, 2018).
Cardioprotective property
Cardioprotection includes all mechanisms and means that contribute to the preservation of the heart by reducing or even preventing myocardial damage and rutin has been reported to have a cardioprotective effect also. Lipopolysaccharide (LPS) gram-negative bacteria endotoxin is widely used for the induction of sepsis-induce heart injury. Rutin attenuates LPS-induced heart injury with its antioxidative and anti-inflammatory effects. Pre-treatment of rats with rutin improved LPS-induced sepsis associated with morphological changes of the myocardium and relieved cardiac marker enzymes (Creatinine kinase and lactate dehydrogenase). The pre-treatment mitigated fibrosis related matrix metalloproteinase-2 and matrix metalloproteinase-9 genes expression in the heart to prevent against LPS-induced cardiac fibrosis. The rutin treatment led to increased antioxidant enzymes to balance the oxidation and antioxidation systems in the heart and also ameliorated TNF-α and IL-6 activity to restrain inflammatory responses in the heart. Thus, it was suggested that rutin could be used as a potential cardioprotective in sepsis (Xianchu et al. 2018). Pirarubicin, an anthracycline antibiotic, frequently used for the treatment of various human cancers very often develops cardiac side effects. Pretreatment with rutin attenuated pirarubicin-induced myocardial histopathological injury, electrocardiogram abnormalities, and cardiac dysfunction. The pre-treatment with rutin also resulted in significantly reduced serum levels of MDA, BNP, CK-MB, CTnT, and LDH and increased serum SOD levels. The rutin treatment also showed a reduction in JNK and Caspase-3 protein levels, compared to the pirarubicin group and it is suggested that the antioxidant and anti-apoptotic properties of rutin may be responsible for the cardioprotective effects observed (Wang et al., 2018). Myocardial infarction (MI), commonly known as a heart attack, occurs when blood flow stops to a part of the heart causing damage to the heart muscle. The examination of the cardioprotective actions of quercetin and rutin in ischaemia–reperfusion-induced myocardial infarction in both normal and diabetic rats, showed that quercetin and rutin significantly limit the infarct size in both the groups of animals in a similar fashion with attenuation of the lipid peroxidation (Annapurna et al., 2009). Study on the effects of rutin on isoproterenol-induced myocardial infarction showed that pretreatment with rutin enhanced survival of the rats and decreased the level of protein kinase C and malonylaldehyde enzyme while the elevation of glutathione and superoxide dismutase level as compared to that in isoproterenol-treated group. The results indicate that rutin significantly reduces myocardial infarction and emphasize the beneficial action in the prevention of myocardial infarction (Malaviya et al., 2011). Diabetic cardiomyopathy (DCM) is a dreadful complication of diabetes. Rutin administration by oral gavage in diabetic rats significantly decreased the blood glucose level, glycated hemoglobin HbA1c, and reduced expression of tumor necrosis factor-alpha (TNF-α), C-reactive protein, and B-type natriuretic peptide compared to diabetic control rats. In addition, rutin provided significant protection against diabetes-associated oxidative stress, prevented degenerative changes in heart, and improved ECG parameters compared to diabetic control rats. Thus, rutin ameliorates DCM through its antioxidant and anti-inflammatory actions on the heart (Saklani et al., 2016). The cardioprotective effect of co-treatment of quercetin or rutin with isoproterenol (ISO) in ISO-induced cardiac fibrosis in rats showed the decreased cardiac weight index and myocardial enzyme activity, increased the activity of superoxide dismutase in the serum, and inhibited the ISO-induced increase in angiotensin II and aldosterone in the plasma. Furthermore, overexpression of transforming growth factor β1 (TGF-β1), connective tissue growth factor (CTGF), and excessive deposition of extracellular matrix (ECM) in isoproterenol-treated myocardial tissues were normalized by quercetin and rutin. The results suggest that both quercetin and rutin exhibited cardioprotective effects in cardiac fibrosis induced by ISO in the rat heart (Li et al., 2013).
Hepatoprotective property
Hepatoprotection is the ability to prevent damage to the liver. High-cholesterol diet (HCD) causing hypercholesterolemia increases the oxidative stress in liver tissues to develop hepatotoxicity. Rats fed with diet containing rutin plus HCD demonstrated significant protective effect against hypercholesterolemia-induced hepatotoxicity with improved liver functions and lipid profile. Transforming growth factor beta (TGF-𝛽)/ mothers against decapentaplegic homolog (Smad) signaling pathway is highly expressed in HCD-induced hepatotoxicity and rutin restored this pathway to nearly their normal levels in hepatic cells. (AlSharari et al., 2016). Isoniazid (INH) is considered one of the most important first-line anti-tuberculosis drugs but it induces severe hepatotoxicity. INH treatment of rats caused hepatotoxicity associated increase in the activity of serum aspartate and alanine aminotransferases and alkaline phosphatases which are indicative to the liver functions. There was also a decrease in the hepatic GSH and SOD activity with elevated liver MDA concentration after INH treatment. However, the oral administration of rutin 1h prior to INH treatment resulted in the amelioration of these alterations suggesting a hepatoprotective role of rutin which could be via antoxidative property (Abdel-Ghaffar et al., 2017). Carbon tetrachloride (CCl4) is a well-known hepatotoxicity inducing agent. The rutin administered intragastrically after 48 h of CCl4-treatment to rats showed significant protection with the depletion of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma glutamyl transpeptidase (γ-GT) in serum which was raised by the induction of CCl4. Concentration of serum triglycerides, total cholesterol and low density lipoproteins was increased while high-density lipoprotein was decreased with rutin treatment in a dose dependent manner. Activity level of endogenous liver antioxidant enzymes i.e. catalase, superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione-S-transferase (GST) and glutathione reductase (GSR) and glutathione (GSH) contents were increased while lipid peroxidation was decreased dose dependently with rutin. Moreover, increase in DNA fragmentation and 8-Oxo-2'-deoxyguanosine (8-oxo-dG) damages while decrease in p53 and CYP 2E1 expression induced with CCl4 was restored with the treatment of rutin. Thus, it was suggested that administration of rutin may be useful in the treatment and prevention of hepatic stress (Khan et al., 2012). 5-Fluorouracil (5-FU) a cancer chemotherapeutic agent is known to cause damages in the liver. In a study on the effects of quercetin and rutin on 5-FU-induced hepatotoxicity, it was reported that 5-FU administration (i.p.) in rats caused a significant increase in serum ALT, LDH, AST and ALP enzyme levels compared to that in control whereas prior intragastric administration of quercetin and rutin significantly restored these enzyme levels. Thus, these results suggest that quercetin and rutin have protective effects on 5-FU-induced hepatotoxicity (Gelen et al., 2017). In another study, rutin alone as well as rutin nano-complexes (rutin combined with egg phosphatidylcholine) were used to assess their comparative protective effects on the CCl4-induce hepatotoxicity in rats. CCl4-intoxicated rats showed increase in SGOT, SGPT, SALP and total bilirubin confirming the hepatic damages. The prior treatment of rats orally with rutin or rutin nano-complexes restored these liver function enzymes and histo-pathological changes induced by CCl4. It was found that rutin nano-complexes showed better solubility, bioavailability, antioxidant activity and hepatoprotective activity as compared to pure rutin at the same dose levels. Thus, the finding indicate the better efficacy of the rutin nano-complex compared to the pure rutin (Ravi et al., 2018). Nonalcoholic fatty liver disease (NAFLD) is characterized by excessive accumulation of hepatic lipids and oxidative injury of hepatocytes. A study on rutin's hypolipidemic and hepatoprotective effects in NAFLD in rats has been reported. Rutin treatment of mice maintained on a high-fat diet and HepG2 cells in vitro showed to significantly lower triglyceride content and the abundance of lipid droplets and also found to reduce cellular malondialdehyde level and restore superoxide dismutase activity in hepatocytes. In addition, rutin treatment also restored the expression of peroxisome proliferator-activated receptor alpha (PPAR-α) and its downstream targets, carnitine palmitoyltransferase 1 and 2 (CPT-1 and CPT-2). Furthermore, it was shown to repress the autophagic function of liver tissues by down-regulating key autophagy biomarkers, including tumor necrosis factor alpha (TNF-α), interleukin 1 beta (IL-1β). Therefore, it was suggested that rutin could serve as an ideal therapeutic agent for NAFLD (Liu et al., 2017). In Ethiopian traditional medicine the aqueous decoction of the leaves of Cineraria abyssinica (Asteraceae) is used for the treatment of various ailments including liver diseases. Pretreatment of the rats orally with the crude extracts of leaves of C. abyssinica, methanol fraction or rutin as well as the standard drug, silymarin ameliorated CCl4-induced hepatocellular damage with the significant reduction in serum ALP, ALT and AST levels. The leaves extracts found to have rutin as a phytoconstituent. The study revealed for the presence of a hepatoprotective and antioxidant phytochemical in the leaves of C. abyssinica that scientifically validates the traditional use of the plant and its potential for the treatment of liver disorders (Sintayehu et al., 2012). The protective effects of rutin and its aglycone quercetin against CCl4-induced liver damage in BALB/cN mice showed that intraperitoneally pretreatment with rutin and with quercetin significantly reduced the activity of plasma transaminases and improved the histological signs of acute liver damage in CCl4-intoxicated mice. Quercetin and rutin attenuated the inflammation in the liver by down-regulating the CCl4-induced activation of nuclear factor-kappa B (NF-κB), tumor necrosis factor-α (TNF-α) and cyclooxygenase (COX-2). In addition, treatment with both flavonoids significantly increased NF-E2-related factor 2 (Nrf2) and heme oxygenase (HO-1) expression in injured livers. Thus, it was suggested that rutin exerts stronger protection against nitrosative stress and hepatocellular damage than quercetin which may be attributed to the presence of a rutinoside moiety in position 3 of the C ring (Domitrović et al., 2012). Acetaminophen (N-acetyl-p-aminophenol/APAP) is known to cause hepatic damages. The rats were treated with APAP to induce hepatotoxicity. The APAP treated rats were administered with rutin and examination of various histological changes and liver function tests showed improved hepatic function by regenerating the histo-architecture and functioning of liver with decreased ALT, total bilirubin, and glucose while increased GSH level, activities of SOD and CAT indicating its protective activity. These changes were comparable to that of the well-known hepatoprotective silymarin-treated group. The hepatoprotective effects of rutin are attributed to its antioxidant and free radical scavenging properties (Reddy et al., 2017). The protective effect of rutin against carbon tetrachloride induced hepatotoxicity in Swiss albino mice showed that rutin pretreatment to mice decreased the level of lipid peroxidation (LPO) significantly as compared to CCl4 treated group and also restored the activity of liver antioxidant enzymes like superoxide dismutase and catalase, glutathione-S-transferase (GST) significantly. Overall, pretreatment of rutin showed the ability to reverse the altered biochemical profile towards normalization and also stabilizes the plasma membrane as well as increases the regenerative potential of the liver in mice (Ashraf et al., 2012). Rats fed with diet containing rutin plus HCD induced a significant protective effect against the hepatotoxicity by reducing the plasma level of alanine transaminase (ALT), aspartate aminotransferase (AST), triglyceride (TG), total cholesterol (TC), and low-density lipoprotein (LDL). The treatment with rutin combination also showed the amelioration of oxidative stress genes with an increase in glutathione peroxidase (GPx), glutathione reductase (GR) and increase in glutathione S transferase α (GSTα), sulfiredoxin-1(Srx1), glutamate-cysteine ligase (GCL) and paraoxonase-1(PON-1) genes expression levels (Al-Rejaie et al., 2013).
Nephroprotective property
Nephroprotection involves the measures which protect the kidney against many aggressive factors and nephroprotective agents are the substances which possess protective activity against nephrotoxicity. Gentamicin, an aminoglycoside antibiotic, is commonly used against gram-negative microorganisms and its therapeutic use is mainly limited by nephrotoxicity. It has been reported that rutin pretreatment in rats attenuated nephrotoxicity induced by gentamicin by reducing the urea, creatinine, and MDA levels and increasing the superoxide dismutase, catalase, and glutathione peroxidase activity, and the glutathione levels. The rutin also inhibited inducible nitric oxide synthase (iNOS), cleaved caspase-3 and light chain 3B (LC3B). Thus, it was suggested that rutin could attenuate gentamicin-induced nephrotoxicity in rats. (Kandemir et al., 2015). Cisplatin, a cancer chemotherapeutic drug is a well-known for developing nephrotoxicity in the hosts. It has been reported that anti-oxidant and anti-inflammatory effect of rutin play important role in preventing cisplatin-induced nephrotoxicity via decreasing the oxidative stress, inhibiting the interconnected ROS/JNK/TNF/P38 MAPK signalling pathways, and repairing the histo-pathological changes against cisplatin-induced nephrotoxicity in rats (Alhoshani et al., 2017). In another cisplatin-induced nephrotoxicity study in rats, rutin pretreatment prevented nerphrotoxic deteriorative effects induced by cisplatin through a protective mechanism that involved reduction of increased oxidative stress as well as caspase-3, TNF-α and NFκB protein expression levels and showed restoration of histopathological changes (Arjumand et al., 2011). Hexachlorobutadiene (HCBD) extensively used in industry is a potent nephrotoxin which may contaminate human foods and water. Pretreatment with rutin decreased serum creatinine and urea as well as urine protein concentrations when compared with HCBD treated rats. Rutin also reversed the HCBD-induced depletion in thiol content and elevation in lipid peroxidation in the kidney. Thus, the results suggest that rutin has the potential to be a good nephroprotective agent against HCBD-induced nephrotoxicity (Sadeghnia etal., 2013). In a study, to investigate the protective effect of rutin and low dose of irradiation (LDR) on cisplatin-induced nephrotoxicity in rats showed that LDR and rutin ameliorated various cisplatin-induce nephrotoxic parameters such as elevation in serum creatinine and urea, disturbance in blood count, elevation in gene expression of tumor necrosis factor alpha, nuclear factor kappa B, interleukin-1β, caspase-3, mitochondrial cytochrome C and apoptosis-inducing factor in renal tissue. Thus, it was suggested that LDR may augment the nephroprotective effect of rutin and hence might be valuable in improving the therapeutic index of cisplatin (Radwan et al., 2017). Kamel et al. (2014) also demonstrated that pretreatment with rutin ameliorated cisplatin-induced alterations in serum sodium, serum and urinary creatinine, creatinine clearance, blood urea nitrogen (BUN), total sodium and potassium excreted in the urine and relative kidney weight. In addition, rutin significantly mitigated the lipid peroxidation in the rat kidney induced by cisplatin. Ansar et al., (2016) showed that rutin has the beneficial impact on lead-induced toxicity also. The pretreatment of rats with rutin reversed various biochemical changes such as increase in the uric acid, creatinine and blood urea nitrogen levels and a decrease in glutathione, superoxide dismutase, catalase and glutathione peroxidase activities observed after only lead acetate treatment. Thus, rutin was suggested to have a beneficial impact on lead-induced toxicity due to its scavenging and antioxidant effect in rats. Fluoride is a well-known soil, water, and air contaminant, and it’s toxicity to humans has been widely studied. In a study to evaluate the protective effect of rutin against sodium fluoride (NaF)-induced nephrotoxicity in rats it was found that co-administration of sodium fluoride and rutin thorough drinking water attenuated the NaF intoxication and decreased the specific gravity, urea, uric acid, protein and creatinine clearance while increased the pH and creatinine of urine suggesting the normalisation of the kidney function (Umarani et al., 2015). Carfilzomib (CFZ), a proteasome inhibitor approved by the FDA to treat multiple myeloma, may cause nephrotoxicity. Significant increase in MDA levels, caspase-3 enzyme and CAT activity and a significant decrease in GSH level and GR activity was observed in the renal tissue of CFZ-treated animals. Co-administration of carfilzomib with rutin significantly reversed their levels toward the normal range. The findings clearly demonstrate that rutin ameliorates CFZ-induced oxidative stress and inflammation in nephrotoxicity (Al‑Harbi et al. 2019)
Haematoprotective property
Hematotoxicity is the study of blood and blood forming tissues as a target organ for drugs, chemicals in the environment, and factors such as stress, exercise, and ionizing radiation. A substance that destroy red blood cells, disrupt blood clotting, and/or cause organ degeneration and generalized tissue damage is referred as hemotoxins, haemotoxins or hematotoxins. Isoniazid (INH) treatment of rats resulted in normocytic anemia associated with leukopenia which was a direct consequence of the reduction in hemoglobin content, packed cell volume, erythrocytes, neutrophil, eosinophil and lymphocyte counts. The oral administration of rutin 1h prior to INH treatment prevented or ameliorated these alterations. It seems likely that rutin protected maure erythrocytes as well as the erythropoietic tissues and factors against the oxidative stress induced by isoniazid or its metabolites (Abdel-Ghaffar et al., 2018). Prasad and Prasad (2018) reported that cisplatin treatment of the tumor-bearing mice caused a decrease in erythrocytes and leukocytes count and hemoglobin content which could lead to develop hematotoxicity in the hosts. However, the combination of rutin and cisplatin treatment showed a noteworthy improvement in erythrocytes and leukocytes count and hemoglobin content indicating the protective ability of rutin against cisplatin-induced hematotoxicity.The in vitro oxidative hemolysis of human red blood cells (RBCs) was induced by adding a water-soluble free radical initiator 2, 2′-azobis (2-methylpropionamidine) dihydrochloride (AAPH) to the suspension of RBCs. However, the AAPH-induced hemolysis in RBCs was significantly suppressed by the addition of various flavonols and their glycosides i.e., quercetin, rutin, quercetin galactopyranoside etc. with AAPH in the suspension. These flavonols bear an ortho-dihydroxyl functionality and showed much more effective anti-hemolysis activity than that of other flavonols i.e., morin, kaempferol, kaempferol glucopyranoside which bear no such functionality (Dai et al., 2006). In another study, the antioxidant effect of pure flavonoids such as kaempferol, quercetin, morin and rutin in the presence of AAPH was studied on red blood cell hemolysis and their -SH capacity. Rutin showed the highest inhibitory effect on hemolysis among the tested flavonoids. It was also found that except quercetin, other flavonoids increased the –SH capacity. This study suggested that tested flavonoids exert beneficial effects in preventing oxidative damage to membranes of RBCs (Asgary et al., 2005). The antioxidative action of the flavonoids quercetin and rutin in normal human erythrocytes, subjected to in vitro oxidative stress induced by tert-butylhydroperoxide (tBHP) was studied. It was shown that with the increase in tBHP concentration, the GSH concentration, G6-PD and glutahione reductase activities were decreased and methemoglobin and Heinz bodies (HB) formation increased. The pretreatment of RBCs suspension with flavonoids prevented the GSH decrease and the formation of HB from oxidative damage by tBHP suggesting haematoprotective role of rutin through reduction in the oxidative stress caused by tBHP (Krukoski et al., 2009). Bothrops snake bites are frequent throughout South and Central Americas and usually lead to inflammatory and bleeding manifestations in patients. In Brazil, Bothrops jararaca snake venom (BjV) evokes hemostatic disturbances, bleeding manifestations, and redox status imbalance. Sachetto et al. (2018) demonstrated the modulatory activity of rutin on the hematological, hemostatic and redox status markers altered by BjV injection in mice. It was found that rutin attenuated local hemorrhage, and the increase in reactive species, prevented the fall in RBC counts and fibrinogen levels, diminished tail bleeding and shortened prothrombin time evoked by envenomation. Furthermore, rutin reduced tissue factor (TF) activity and altered the protein expression of TF in liver, lungs, heart and skin suggesting it has a great potential to be used as an ancillary therapeutic agent for snakebites. Further, Al‑Harbi et al. (2019) demonstrated that co-administration of carfilzomib with rutin significantly reversed the carfilzomib-induced decrease in RBCs, WBCs, platelets count, haemoglobin content, and haematocrit concentration in a dose-dependent manner in rats.
Antiarthritis property
Arthritis is a term often used to describe any disorder that affects joints and use of dietary flavonoids have been reported to control joint inflammation and lessen arthritis in both human Rheumatoid arthritis and animal models (Hughes et al., 2017). Rheumatoid arthritis is an autoimmune disorder characterized by synovial joint inflammation, hyperplasia, irreversible cartilage and bone damage, and skeletal disorders. Rheumatoid arthritis induced by collagen-induced arthritis (CIA) is a widely studied animal model of inflammatory polyarthritis. Nuclear factor-κB (NF-κB) and inducible nitric oxide synthase (iNOS) are important mediators of inflammatory response in human and animal models of arthritis and its over expression leads to the extracellular matrix degradation, and excessive cartilage and bone resorption, ultimately leading to the irreversible damage to joints. The intraperitoneal treatment with rutin and rutin-conjugated gold nanoparticles (R-AuNPs) in collagen-induced arthritis rats showed a significant down-regulation in the NF-κB and iNOS expression in the treatment groups compared to the arthritic control group. Further, reduction in the arthritic score as well as in the nitric oxide and peroxide levels was observed in the treated groups. The findings suggested potential clinical role of rutin and R-AuNPs in the treatment of rheumatoid arthritis (Gul et al., 2018). In another study, it was found that rutin markedly inhibited joint swelling and significantly decreased the free radical load in CIA rats, which suggested that rutin was effective in treating inflammatory disorders like rheumatoid arthritis. The pretreatment of rutin in CIA-induced rats replenished GSH and SOD level significantly and suppressed the accumulation of lipid peroxidation, nitric oxide, probably by scavenging free radicals and as a result helped to maintain the integrity of cellular membranes in the injured cartilage. Thus, it was suggested that the administration of rutin have potential value in the treatment of rheumatoid arthritis (Umar et al., 2012). In a study the septic arthritis was induced by injecting Complete Freund's Adjuvant mixed with C. albicans cell wall into the hind footpads of the mice. After 24 h of the final injection and induction of septic arthritis in mice groups having the swollen footpad were treated intraperitoneally (i.p.) with rutin and it was found that about 45% of the edema was reduced which was mediated by rutin's ability to inhibit nitric oxide production from macrophages and T-cells proliferation. Furthermore, this flavonoid also inhibited the growth of C. albicans and resulted in no hemolysis. It was suggested that the dual effects of rutin, anti-arthritic and anti-candidal may be helpful as an all-in-one treatment for septic arthritis (Han, 2009). Osteoarthritis (OA) is a disease characterized by fibrillations, fissures, and disappearance of cartilage. Cartilage degradation is associated with structural and metabolic changes in joint tissues such as subchondral bone sclerosis and synovial membrane inflammation. The protective effects of three polyphenols oleuropein, rutin and curcumin were studied on spontaneously developed osteoarthritis in guinea pigs. These polyphenols feeding with standard guinea pig diet significantly reduced the cartilage degradation score. Biomarkers of osteoarthritis such as Coll2-1 was decreased in serum by rutin and the combination of rutin/curcumin. However, the biomarker fibulin-3 fragments (Fib3-1 and Fib3-2) only decreased by the rutin/curcumin mixture, while Coll2-1NO2 was significantly decreased by all treatments. Thus, it was suggested that oleuropein and rutin ± curcumin significantly slowed down the progression of spontaneous OA lesions in guinea pigs (Horcajada et al., 2015). The effect of rutin in complete Freund's adjuvant-induced arthritic rats has shown the significant upsurge in the level of superoxide dismutase, glutathione peroxidase and glutathione and decrease in the level of malondialdehyde after treatment in a dose-dependent manner. Furthermore, the rutin treatment caused lowering of cytokines tumor necrosis factor-α and interleukin-1β, and transcription factors NF-κBp65 (Ser536) in arthritic rats. In addition, histopathological examination showed that the inflammatory cells infiltration, synovial hyperplasia, pannus formation and cartilage and bone erosion had considerably improved on administration of rutin. Thus, the findings demonstrated the protective effect of rutin against the rheumatoid arthritis via suppression of NF-κB p65 protein expression (Sun et al., 2017).
Anthelmintic property
Anthelmintics or antihelminthics are a group of antiparasitic drugs that expel parasitic worms (helminths) and other internal parasites from the body by either stunning or killing them without causing significant damage to the host. In a study, rutin isolated from tobacco leaves revealed that rutin has anthelmintic and larvicidal potential against roundworms (Ascaridia galli) and larvae of the yellow fever mosquitoes Aedes aegypti (Dubey et al., 2013). In a study three flavonol glycosides i.e. rutin, nicotiflorin and narcissin were isolated from the extracts of tanniferous legume forage Sainfoin i.e. Onobrychis viciifolia. Larval migration inhibition (LMI) assay on the nematode Haemonchus contortus 3rd-stage larvae showed that these flavonol glycosides inhibited significantly the larval migration suggesting the anthelmintic property of these glycosides (Barrau et al., 2005). Strongyle parasites are commonly found in the large intestine of ruminants. An in vitro study on anthelmintic activity of four plant-derived pure compounds i.e., mangiferin, rutin, quercetin and β-sitosterol on sheep gastrointestinal strongyles (GIS) showed that all tested compounds at all concentrations significantly inhibited the hatching of the eggs when compared to the untreated controls and also significantly increased the death of 3rd-stage larvae compared to the untreated controls, except for β-sitosterol (Giovanelli et al., 2018).
Protective ability against testicular toxicity
Testicular toxicity is defined as adverse impacts on sexual function/fertility in adult males because of toxicants and pharmaceutical compounds which adversely affect the male reproductive systemt by targeting different cell types of the testis (ie, germ, Leydig, and Sertoli cells) and altering hormone levels. Cyclophosphamide (CYC), an anticancer alkylating agent has been known as a male reproductive toxicant. The protective effect of rutin on CYC-induced reproductive toxicity in rats showed that rutin prevented lower sperm counts, sperm motility, daily sperm production, and higher abnormal sperm numbers induced by CYC. Rutin decreased superoxide dismutase (SOD), lactate dehydrogenase (LDH), and sorbitol dehydrogenase (SDH) and increased catalase (CAT), 3β-hydroxysteroid dehydrogenase (3β-HSD), 17β-HSD, alkaline phosphatase (ALP), and acid phosphatase (ACP) induced by CYC in testes. In epididymis, rutin increased SOD, CAT, GSH, GSH-Px, GR, GST SDH, ALP and ACP and decreased MDA and LDH induced by CYC. Thus, it was suggested that rutin ameliorated CYC-induced reproductive toxicity in rats (Abarikwu et al., 2012). Exposure to cadmium (Cd) reduces sperm quality and induces oxidative stress in the testis. In a study, the effect of ethanol intake on CdCl2-induced testicular toxicity with or without rutin pre-treatment in rats showed that co-treatment with CdCl2- ethanol decreased the activities of glutathione (GSH), GSH-peroxidase and superoxide dismutase resulting to slight increase in the testicular MDA level compared to CdCl2-treated rats. These animals had higher levels of abnormal spermatozoa, decreased epididymal sperm number and serum testosterone levels compared to the Cd-treated animals. Rutin administration protected against these EtOH effects in a dose-dependent manner and animals had higher GSH and GSH-Px activities beyond the control values. Therefore, it was suggested that rutin induces GSH and GSH-Px activities to protect against Cd+EtOH-induced testis oxidative stress in rats (Abarikwu et al., 2017). Gentamycin is an antibiotic used in neonatal sepsis and other systemic infections caused by gram negative microorganisms and is known to reduce sperm count, sperm motility, and sperm viability. In a study on the effects of bioflavonoids rutin and naringin on gentamycin-induced testicular oxidative stress in rats showed that rutin and naringin pretreatment caused significant and dose dependent reduction in MDA levels and increase in levels of antioxidant enzymes, SOD and catalase when compared to control animals suggesting its protective ability. Sperm count, motility, viability were also protected and normalized with rutin and naringin treatment along with improved testicular architecture. Thus, it was suggested that both the bioflavonoids were effective in reducing the gentamycin-induced testicular toxicity in rats (Akondi et al. 2011). Cisplatin treatment causes damage in the male reproductive system. The effects of rutin against cisplatin-induced reproductive toxicity in male rats showed that only cisplatin treatment resulted increased damages on some spermatological parameters (i.e. motility, cauda epididymal sperm density, dead sperm percentage and morphological sperm abnormalities), the oxidative stress and testicular degeneration and apoptosis. However, rutin pretreatment mitigated these side effects when compared to that of cisplatin alone. It was suggested that rutin treatment reduces cisplatin-induced reproductive toxicity as a potential antioxidant compound (Aksu et al., 2017). In another cisplatin-induced testicular toxicity study it was found that only cisplatin treatment resulted in a significant decrease in daily sperm production, head length and % DNA in head, reduction of epithelial cells height, tubular diameter, number of spermatogonia, spermatocytes and spermatids, the intra-testicular testosterone concentrations, increase in the thiobarbituric acid reactive substances (TBARS) and oxidative stress in testicular tissues. Rutin treatment to cisplatin-treated rats resulted in reversing cisplatin’s effects on DNA damage, sperm count, histological and biochemical parameters, thus suggested the shielding effects of rutin against cisplatin-induced testicular toxicity (Jahan et al., 2018). The effects of rutin on diabetic-induced impairments in sexual behaviour, spermatogenesis and oxidative testicular damage have also been reported. Diabetic-induced alterations in male sexual behaviour, sperm count, motility and viability were markedly corrected following rutin treatment to the diabetic rats. Rutin also attenuated the diabetic associated decrease in serum testosterone and penile cGMP content, while improved diabetic-associated condition of inflammation and testicular lipid peroxidation and oxidative stress. Histopathological evaluation revealed damaged testicular tissues in diabetic rats, which was protected following rutin treatment. Thus, it was suggested that rutin treatment improves sexual functionality and also protects against diabetic-induced testicular damage in rats (Al-Roujeaie et al., 2017). The pathophysiology of testicular torsion-detorsion is an ischemia-reperfusion injury caused by over-generation of reactive oxygen species (ROS). Unilateral testicular torsion-detorsion caused a significant increase in malondialdehyde level and caused significant decreases in superoxide dismutase, catalase activities, and spermatogenesis in ipsilateral testes. However, the rats treated with rutin had a significant decrease in malondialdehyde level and had significant increases in superoxide dismutase, catalase activities, and spermatogenesis in ipsilateral testes, compared with torsion-detorsion group, thus, suggesting rutin’s protective effects in testes from ischemia-reperfusion injury (Wei et al., 2011). Ansar et al. (2016) studied the effect of rutin on lead acetate-induced testis tissue damages and antioxidant enzyme activities in rats and suggested that rutin possesses significant potential to reduce lead acetate-induced testicular toxicity. Further it has been demonstrated that rutin as a flavonoid could restore motility of other metal (AlCl3, CdCl2, and PbCl4)-exposed sperms from human and protect it against metal-induced lipid peroxidation (Jamalan et al., 2016). Restraint (immobilization) stress is an amalgamation of both physical and psychological stress, whereby movement is confined to a restricted area and the individual is isolated from its group. It is an experimental procedure developed for biomedical studies of stress. Restraint stress impedes male reproductive capacity by hampering the hypothalamic-pituitary-testicular axis (Rai et al., 2004). In a study it was found that restraint stress in mice significantly decreases body weights, testis and epididymis weights, thymus weights, visceral fats, serum concentrations of testosterone, sperm counts, sperm motility and sperm viability, while it increases serum epinephrine levels, adrenal gland weights and abnormal sperms. In addition, restraint stress severely damages the testicular histoarchitecture and spermatogenesis and also shows broken seminiferous tubules, few spermatozoa in lumen and less population of Leydig cells. In this study, expression levels of cleaved Poly (ADP-ribose) polymerase-1 (PARP1) and cleaved caspase-3 are significantly increased in testes. The rutin treatment to restrained mice demonstrated that rutin significantly ameliorates the side effects induced by restraint stress (Mehfooz et al., 2018). Idarubicin as a cancer chemotherapeutic drug, has side effects on testicular tissue. Pre-treatment of mice with rutin showed protective effects on the idarubicin-induced testicular damages. The protective capacity of rutin was attributed to its ability to reduce apoptosis by under-expression of caspase-3 and overexpression of Bcl2 genes (Deihimi et al., 2017).
Figure 3. Summary of various biological properties of rutin
Conclusion
The findings from the various reports covered in the review establish that rutin is an important nutraceutical agent naturally present in various plants and plant-derived fruits, vegetables etc. and has no toxic effects. Moreover, it has the large beneficial effects (Figure 3) in the body by treating various diseases and also protecting against various side effects induced by different drugs, chemicals, metals etc. The intake of rutin through the consumption of natural food and fruits should be included in the regular diet to cure various diseases and toxicities, and also to prevent their possible development.
Conflicts of interest
There are no conflicts of interest.
References
Abarikwu SO, Olufemi PD, Lawrence CJ, Wekere FC, Ochulor AC, Barikuma AM. 2017. Rutin, an antioxidant flavonoid, induces glutathione and glutathione peroxidase activities to protect against ethanol effects in cadmium-induced oxidative stress in the testis of adult rats. Andrologia, 49:1-12.
Abarikwu SO, Otuechere CA, Ekor M, Monwuba K, Osobu D. 2012. Rutin ameliorates cyclophosphamide-induced reproductive toxicity in male rats. Toxicology International, 19(2): 207-214.
Abdel-Ghaffar O, Mahmoud ST, Said AA, Sanad A-AFY. 2017. Hepatoprotective effect of rutin against oxidative stress of isoniazid in albino rats. International Journal of Pharmacology, 13: 516-528.
Abdel-Ghaffar O, Mahmoud ST, Said AA, Sanad A-AFY. 2018. Ameliorative effect of rutin against isoniazid-induced alterations in certain hematological and biochemical parameters of albino rats. International Journal of Pharmacology, 14: 39-51.
Akondi RB, Akula A, Challa SR. 2011. Protective effects of rutin and naringin on gentamycin induced testicular oxidative stress. European Journal of General Medicine 8(1): 57-64.
Aksu EH, Kandemir FM, Ozkaraca M, Omur AD, Kucukler S, Comaki S. 2017. Rutin ameliorates cisplatin-induced reproductive damagevia suppression of oxidative stress and apoptosis in adult male rats. Andrologia, 49: 1-8.
Al‑Harbi NO, Imam F, Al‑Harbi MM, Al‑Shabanah OA, Alotaibi MR, Sobeai HMS, Afzal M, Kazmi I, Rikabi ACA. 2019. Rutin inhibits carfilzomib‑induced oxidative stress and inflammation via the NOS‑mediated NF‑κB signaling pathway. Inflammopharmacology, 1-11.
Alhoshani AR, Hafez MM, Husain S, Al-sheikh AM, Alotaibi MR, Rejaie SSA, Alshammari MA, Almutairi MM, Al-Shabanah OA. 2017. Protective effect of rutin supplementation against cisplatin-induced nephrotoxicity in rats. BMC Nephrology, 18: 194-204.
Almutairi MM, Alanazi WA, Alshammari MA, Alotaibi MR, Alhoshani AR, Al-Rejaie SS, Hafez MM, Al-Shabanah OA. 2017. Neuro-protective effect of rutin against cisplatin-induced neurotoxic rat model. BMC Complementary and Alternative Medicine, 17: 472-480.
Alonso-Castro AJ, Dominguez F, Garcia-Carranca A. 2013. Rutin exerts antitumor effects on nude mice bearing SW480 tumor. Archives of Medical Research, 44 (2013): 346-351.
Al-Rejaie SS, Aleisa AM, Sayed-Ahmed MM, AL-Shabanah OA, Abuohashish HM, Ahmed MM, Al-Hosaini KA, Hafez MM. 2013. Protective effect of rutin on the antioxidant genes expression in hypercholestrolemic male Westar rat. BMC Complementary and Alternative Medicine, 13:136-144.
Al-Roujeaie AS, Abuohashish HM, Ahmed MM, Alkhamees OA. 2017. Effect of rutin on diabetic-induced erectile dysfunction: Possible involvement of testicular biomarkers in male rats. Andrologia, 49: 1-9.
AlSharari SD, Al-Rejaie SS, Abuohashish HM, Ahmed MM, Hafez MM. 2016. Rutin attenuates hepatotoxicity in high-cholesterol-diet-fed rats. Oxidative Medicine and Cellular Longevity, 2016: 1-11.
Amin MU, Khurram M, Khattak B, Khan J. 2015. Antibiotic additive and synergistic action of rutin, morin and quercetin against methicillin resistant Staphylococcus aureus. BMC Complementary and Alternative Medicine, 15: 59-72.
Andlauer W, Stumpf C, Fürst P. 2001. Intestinal absorption of rutin in free and conjugated forms.Biochemical Pharmacology, 62(3): 369-374.
Annapurna A, Reddy CS, Akondi RB, Rao SRC. 2009. Cardioprotective actions of two bioflavonoids, quercetin, and rutin, in experimental myocardial infarction in both normal and streptozotocin-induced type I diabetic rats. Journal of Pharmacy and Pharmacology, 61: 1365-1374.
Ansar S, AlGhosoon HT, Hamed S. 2016. Evaluation of the protective effect of rutin on lead acetate-induced testicular toxicity in Wistar rats. Toxin Reviews, 34(4): 195-199.
Ansar S, Hamed S, AlGhosoon HT, AlSaedan RA, Iqbal M. 2016. The protective effect of rutin against renal toxicity induced by lead acetate. Toxin Reviews, 35: 58-62.
Arima H, Ashida H, Danno G. 2002. Rutin-enhanced antibacterial activities of flavonoids against Bacillus cereus and Salmonella enteriditis. Bioscience, Biotechnology, and Biochemistry, 66(5): 1009-1014.
Arjumand W, Seth A, Sultana S. 2011. Rutin attenuates cisplatin-induced renal inflammation and apoptosis by reducing NFκB, TNF-α and caspase-3 expression in Wistar rats. Food and Chemical Toxicology, 49(9): 2013-2021.
Asgary S, Naderi GH, Askari N. 2005.Protective effect of flavonoids against red blood cell hemolysis by free radicals. Experimental & Clinical Cardiology, 10(2): 88-90.
Ashraf J, Nagma, Siddique J, Mirani N, Rub A. 2012. Protective effect of rutin against carbon tetrachloride-induced hepatotoxicity in mice. International Journal of Drug Development and Research, 4(2): 352-357.
Aziza SAH, Azab ME, El-Shall SK. 2014. Ameliorating role of rutin on oxidative stress induced by iron overload in hepatic tissue of rats. Pakistan Journal of Biological Sciences,17(8): 964-977.
Banudevi S, UmaMaheswari K, Vignesh R. 2018. Amelioration of oxidative stress in differentiated neuronal cells by rutin regulated by a concentration switch. Biomedicine & Pharmacotherapy, 108: 15-26.
Barrau E, Fabre N, Fouraste I,Hoste H. 2005. Effect of bioactive compounds from sainfoin (Onobrychis viciifolia Scop.) on the in vitro larval migration of Haemonchus contortus: Role of tannins and flavonol glycosides. Parasitology, 131: 531-538.
Basile A, Sorbo, S, Giordano S, Ricciardi L, Ferrara S, Montesano D, Cobianchi RC, Vuotto ML, Ferrara L. 2000. Antibacterial and allelopathic activity of extract from Castanea sativa leaves. Fitoterapia, 71: S110-S116.
Calzada F, Solares-Pascasio JI, Valdes M, Garcia-Hernandez N, Velázquez C, Ordoñez-Razo RM, Barbosa E. 2018. Antilymphoma potential of the ethanol extract and rutin obtained of the leaves from Schinus molle linn. Pharmacognosy Research, 10(2): 119-123.
Chen H, Miao Q, Geng M, Liu J, Hu Y, Tian L, Pan J, Yang Y. 2013.Anti-tumor effect of rutin on human neuroblastoma cell lines through inducing G2/M cell cycle arrest and promoting apoptosis. The ScientificWorld Journal, 2013: 1-8.
Chen X, Yin OQP, Zuo Z, Chow MSS. 2005. Pharmacokinetics and modeling of quercetin and metabolites. Pharmaceutical Research, 22(6): 892-901.
Choudhury R, Srai SK, Debnam E, Rice-Evans CA. 1999. Urinary excretion of hydroxycinnamates and flavonoids after oral and intravenous administration. Free Radical Biology and Medicine, 27 (3-4): 278-286.
Crozier A, Clifford MN, Ashihara H. 2006. Plant secondary metabolites: occurrence, structure and role in the human diet. Wiley-Blackwell, 384 Pages.
Dai F, Miao Q, Zhou B, Yang L, Liu Z-L. 2006. Protective effects of flavonols and their glycosides against free radical induced oxidative hemolysis of red blood cells. Life Sciences, 78(21): 2488-2493.
Danciu C, Pinzaru IA, Dehelean CA, Hancianu M, Zupkó I, Navolan D, Licker M, Ghuilai RM, Şoica CM. 2018. Antiproliferative and antimicrobial properties of pure and encapsulated rutin. Farmacia, 66(2): 302-308.
Dar RA, Shahnawaz M, Qazi PH. 2017. Natural product medicines: A literature update. Journal of Phytopharmacology,6(6): 349-351.
Deihimi M, Moghbelinejad S, Najafipour R, Parivar K, Nassiri-Asl M. 2017. The effects of rutin on the gene expression of dazl, bcl2, and caspase3 in idarubicin-induced testicular damages in mice. Iranian Red Crescent Medical Journal, 19(4): 1-5.
Dhanya R, Arun KB, Syama HP, Nisha P, Sundaresan A, Santhosh KTR, Jayamurthy P. 2014. Rutin and quercetin enhance glucose uptake in L6 myotubes under oxidative stress induced by tertiary butyl hydrogen peroxide. Food Chemistry, 158: 546-554.
Dietrych-Szostak D, Oleszek W. 1999. Effect of processing on the flavonoid content in buckwheat (Fagopyrum esculentum Moench) grain. Journal of Agricultural and Food Chemistry, 47:4384-4387.
Dixit S. 2014. Anticancer effect of rutin isolated from the methanolic extract of Triticum aestivum straw in mice. Medical Sciences, 2: 153-160.
Domitrović R, Jakovac H, Marchesi VV, Vladimir-Knežević S, Cvijanović O, Tadić Z, Romić Z, Rahelić D. 2012. Differential hepatoprotective mechanisms of rutin and quercetin in CCl4 -intoxicated BALB/cN mice. ActaPharmacologicaSinica, 33(10): 1260-1270.
Dubey S, Ganeshpurkar A, Bansal D, Dubey N. 2013. Experimental studies on bioactive potential of rutin. Chronicles of Young scientists, 4: 153-157.
Ekor M. 2014. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Frontiers in Pharmacology, 4: 1-10.
Erlund I, Kosonen T, AlfthanG, Maenpaa J, Perttunen K, Kenraali J, Parantainen J, Aro A. 2000. Pharmacokinetics of quercetin from quercetin aglycone and rutin in healthy volunteers. European Journal of Clinical Pharmacology, 56: 545-553.
Ganeshpurkar A, Saluja AK. 2017. The pharmacological potential of rutin. Saudi Pharmaceutical Journal, 25: 149-164.
Gegotek A, Rybaltowska-Kawalko P, Skrzydlewska E. 2017. Rutin as a mediator of lipid metabolism and cellular signaling pathways interactions in fibroblasts altered by UVA and UVB radiation. Oxidative Medicine and Cellular Longevity, 2017: 1-20.
Gelen V, Sengul E, Gedikli S, AtilaG, Uslu H, Makav M. 2017. The protective effect of rutin and quercetin on 5-FU-induced hepatotoxicity in rats. Asian Pacific Journal of Tropical Biomedicine, 7(7): 647-653.
Ghorbani A. 2017. Mechanisms of antidiabetic effects of flavonoid rutin. Biomedicine & Pharmacotherapy, 96: 305-312.
Giovanelli F, Mattellini M, Fichi G, Flamini G, Perrucci S. 2018. In vitro anthelmintic activity of four plant-derived compounds against sheep gastrointestinal nematodes. Veterinary Sciences, 5: 78-85.
Graf BA, Ameho C, Dolnikowski GG, Milbury PE, Chen CY, Blumberg JB. 2006. Rat gastrointestinal tissues metabolize quercetin. Journal of Nutrition, 136(1): 39 – 44.
Guardia T, Rotelli AE, Juarez AO, Pelzer LE. 2001. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Farmaco, 56(9): 683-687.
Gul A, Kunwar B, Mazhar M, Faizi S, Ahmed D, Shah MR, Simjee SU. 2018. Rutin and rutin-conjugated gold nanoparticles ameliorate collagen-induced arthritis in rats through inhibition of NF-κB and iNOS activation. International Immunopharmacology, 59: 310-317.
Han Y. 2009. Rutin has therapeutic effect on septic arthritis caused by Candida albicans. International Immunopharmacology, 9: 207-211.
Hashemzaei M, Far AD, Yari A, Heravi RE, Tabrizian K, Taghdisi SM, Sadegh SE, Tsarouhas K, Kouretas D, Tzanakakis G, Nikitovic D, Anisimov NY, Spandidos DA, Tsatsakis AM, Rezaee R. 2017. Anticancer and apoptosis‑inducing effects of quercetin in vitro and in vivo. Oncology Reports, 38: 819-828.
Horcajada MN, Sanchez C,MembrezScalfo F, Drion P, Comblain F, Taralla S, Donneau AF, Offord EA, Henrotin Y. 2015. Oleuropein or rutin consumption decreases the spontaneous development of osteoarthritis in the Hartley guinea pig. Osteoarthritis Cartilage, 23: 94-102.
Hughes SD, Ketheesan N, Haleagrahara N. 2017. The therapeutic potential of plant flavonoids on rheumatoid arthritis. Critical Reviews in Food Science and Nutrition, 57(17): 3601-3613.
Hunyadi A, Martins A, Hsieh T-J, Seres A, Zupko I. 2012. Chlorogenic acid and rutin play a major role in the in vivo anti-diabetic activity of Morus alba leaf extract on type II diabetic rats. PLoS ONE, 7(11): 1-6.
Hussein SA, Zaid OARA, Abdel-Maksoud HA, Khadija AAA. 2014. Anti-inflammatory and anti-oxidant effect of rutin on 2, 4, 6-Trinitrobenzene sulfonic acid induced ulcerative colitis in rats. Benha Veterinary Medical Journal, 27(1): 208‐220.
Inocencio C, Rivera D, Alcaraz F, Tomas-Barberan FA. 2000. Flavonoid content of commercial capers (Capparis spinosa, C. sicula and C. orientalis) produced in Mediterranean countries. European Food Research and Technology, 212:70-74.
Iriti M, Kubina R, Cochis A, Sorrentino R, Varoni EM, Kabała-Dzik A, Azzimonti B, Dziedzic A, Rimondini L, Wojtyczka RD. 2017. Rutin, a quercetin glycoside, restores chemosensitivity in human breast cancer cells. Phytotherapy Research, 31(10):1529-1538.
Jadhav R, Puchchakayala G. 2012. Hypoglycemic and antidiabetic activity of flavonoids: Boswellicacid, ellagic acid, quercetin, rutin on streptozotocin-nicotinamide induced type2 diabetic rats. International Journal of Pharmacy and Pharmaceutical Sciences, 4 (2): 251-256.
Jahan S, Munawar A, Razak S, Anam S, Ain QU, Ullah H, Afsar T, Abulmeaty M, Almajwal A. 2018. Ameliorative effects of rutin against cisplatin-induced reproductive toxicity in male rats. BMC Urology, 18: 107-117.
Jamalan M, Ghaffari MA, Hoseinzadeh P, Hashemitabar M, Zeinali M. 2016. Human sperm quality and metal toxicants: protective effects of some flavonoids on male reproductive function. International Journal of Fertility & Sterility, 10(2): 215–223.
Jantrawut P, Akazawa H, Ruksiriwanich W. 2014. Anti-cancer activity of rutin encapsulated in low methoxyl pectin beads. International Journal of Pharmacy and Pharmaceutical Sciences, 6(3): 199-202.
Jantrawut P, Phongpradist R, Muller M, Viernstein H. 2017. Enhancement of anti-inflammatory activity of polyphenolic flavonoid rutin by encapsulation. Pakistan Journal of Pharmaceutical Sciences, 30(5): 1521-1527.
Johann S, Mendes BG, Missau FC, de Resende MA, Pizzolatti MG. 2011. Antifungal activity of five species of Polygala. Brazilian Journal of Microbiology, 42(3): 1065-1075.
Kamel KM, El-Raouf OMA, Metwally SA, El-Latif HAA, El-sayed ME. 2014. Hesperidin and rutin, antioxidant citrus flavonoids, attenuate cisplatin-induced nephrotoxicity in rats. Journal of Biochemical and Molecular Toxicology, 28(7): 312-319.
Kandemir FM, Ozkaraca M, Yildirim BA, Hanedan B, Kirbas A, Kilic K, Aktas E, Benzer F. 2015. Rutin attenuates gentamicin-induced renal damage by reducing oxidative stress, inflammation, apoptosis, and autophagy in rats. Renal Failure, 37(3): 518-525.
Kaur R, Arora S, Thukral AK. 2015. Evaluation of antimutagenic and antioxidant activities of rutin. International Journal of Pharma and Bio Sciences, 6(1): 816-825.
Khan RA, Khan MR, Sahreen S. 2012. CCl4-induced hepatotoxicity: protective effect of rutin on p53, CYP2E1 and the antioxidative status in rat. BMC Complementary and Alternative Medicine, 12: 178-183.
Kim D-O, Chun OK, Kim YJ, Moon H-Y, Lee CY. 2003. Quantification of polyphenolics and their antioxidant capacity in fresh plums. Journal of Agricultural and Food Chemistry, 51:6509-6515.
Knekt P, Kumpulainen J, Järvinen R, Rissanen H, Heliövaara M, Reunanen A, Hakulinen T, Aromaa A. 2002. Flavonoid intake and risk of chronic diseases. The American Journal of Clinical Nutrition, 76: 560-568.
Koçanci FG, Hamamcioğlu B, Aslim B. 2017. Neuroprotective effects of rutin and quercetin flavonoids in Glaucium corniculatum methanol and water extracts. International Journal of Secondary Metabolite, 4(3): 85-93.
Koval’skii IV, Krasnyuk II, Krasnyuk Jr. II, Nikulina OI, Belyatskaya AV, Kharitonov Yu Ya, Feldman NB, Lutsenko SV. 2014. Mechanisms of rutin pharmacological action (review). Pharmaceutical Chemistry Journal, 48(2): 73-76.
Kreft S, Knapp M, Kreft I. 1997. Extraction of rutin from buckwheat (Fagopyrum esculentum Moench) seeds and determination by capillary electrophoresis. Journal of Agricultural and Food Chemistry, 47(11): 4649-4652.
Kreft S, Knapp M, Kreft I. 1999. Extraction of rutin from buckwheat (Fagopyrum escuentum Moench) seeds and determination by capillary electrophoresis. Journal of Agricultural and Food Chemistry, 47:4649-4652.
Krukoski DW, Comar SR, Claro LM, Leonart MSS, Nascimento AJ. 2009. Effect of vitamin C, deferoxamine, quercetin and rutin against tert-butyl hydroperoxide oxidative damage in human erythrocytes. Hematology, 14(3): 168-172.
Li M, Jiang Y, Jing W, Sun B, Miao C, Ren L. 2013. Cardioprotective effect than its glycoside derivative rutin on isoproterenol-induced cardiac fibrosis in the rat. Canadian Journal of Physiology and Pharmacology, 91: 951-959.
Lin J-P, Yang J-S, Lin J-J, Lai K-C, Lu H-F, Ma C-Y, Wu RS-C, Wu KC, Chueh FS, Wood WG, Chung JG. 2012. Rutin inhibits human leukemia tumor growth in a murine xenograft model in vivo. Environmental Toxicology, 27(8): 480-484.
Lin JP, Yang JS, Lu CC, Chiang JH, Wu CL, Lin JJ, Lin HL, Yang MD, Liu KC, Chiu TH, Chung JG. 2009. Rutin inhibits the proliferation of murine leukemia WEHI-3 cells in vivo and promotes immune response in vivo. Leukemia Research, 33 (2009): 823–828.
Liu Q, Pan R, Ding L, Zhang F, Hu L, Ding B, Zhu L, Xia Y, Dou X. 2017. Rutin exhibits hepatoprotective effects in a mouse model of non-alcoholic fatty liver disease by reducing hepatic lipid levels and mitigating lipid-induced oxidative injuries. International Immunopharmacology, 49: 132-141.
Maatta KR, Kamal-Eldin A, Torronen AR. 2003. High-performance liquid chromatography (HPLC) analysis of phenolic compounds in berries with diode array and electrospray ionization mass spectrometric (MS) detection: Ribes species. Journal of Agricultural and Food Chemistry, 51:6736-6744.
Machawal L, Kumar A. 2014. Possible involvement of nitric oxide mechanism in the neuroprotective effect of rutin against immobilization stress induced anxiety like behaviour, oxidative damage in mice. Pharmacological Reports, 66: 15-21.
Magalingam KB, Radhakrishnan A, Haleagrahara N. 2013. Rutin, a bioflavonoid antioxidant protects rat pheochromocytoma (PC-12) cells against 6-hydroxydopamine (6-OHDA)-induced neurotoxicity. International Journal of Molecular Medicine, 32: 235-240.
Magalingam KB, Radhakrishnan A, Ramdas P, Haleagrahara N. 2015a. Quercetin glycosides induced neuroprotection by changes in the gene expression in a cellular model of Parkinson’s disease. Journal of Molecular Neuroscience, 55(3): 609–617.
Magalingam KB, Radhakrishnan AK, Haleagrahara N. 2015b. Protective mechanisms of flavonoids in Parkinson’s disease. Oxidative Medicine and Cellular Longevity, 2015: 1-14.
Mahmoud NN, Carothers AM, Grunberger D, Bilinski RT, Churchill MR, Martucci C, Newmark HL, Bertagnolli MM. 2000. Plant phenolics decrease intestinal tumors in an animal model of familial adenomatous polyposis. Carcinogenesis, 21(5): 921-927.
Makris DP, Rossiter JT. 2001. Domestic processing of onion bulbs (Allium cepa) and asparagus spears (Asparagus officinalis): effect on flavonol content and antioxidant status. Journal of Agricultural and Food Chemistry, 49:3216-3222.
Malaviya S, Bhalodia Y, Vaghasiya J, Manek R, Sheth N, Jivani N. 2011. Cardioprotective effect of rutin considering cardiac enzyme level in induced myocardial infarction. Latin American Journal of Pharmacy, 30 (2): 397-401.
Marella S. 2017. Flavonoids-The most potent poly-phenols as antidiabetic agents: An overview. Modern Approaches in Drug Designing, 1(3): 1-5.
Mehfooz A, Wei Q, Zheng K, Fadlalla MB, Maltasic G, Shi F. 2018. Protective roles of rutin against restraint stress on spermatogenesis in testes of adult mice. Tissue and Cell, 50: 133-143.
Na J-Y, Kim S, Song K, Kwon J. 2014. Rutin alleviates prion peptide-induced cell death through inhibiting apoptotic pathway activation in dopaminergic neuronal cells. Cellular and Molecular Neurobiology, 34: 1071-1079.
Nam TG, Lim T-G, Lee BH, Lim S, Kang H, Eom SH, Yoo M, Jang HW, Kim D-O. 2017. Comparison of anti-inflammatory effects of flavonoid-rich common and tartary buckwheat sprout extracts in lipopolysaccharide-stimulated RAW 264.7 and peritoneal macrophages. Oxidative Medicine and Cellular Longevity, 2017: 1-12.
Niture NT, Ansari AA, Naik SR. 2014. Anti-hyperglycemic activity of rutin in streptozotocin-induced diabetic rats: an effect mediated through cytokines, antioxidants and lipid biomarkers. Indian Journal of Experimental Biology, 52(7): 720-727.
Nkpaa KW, Onyeso GI. 2018. Rutin attenuates neurobehavioral deficits, oxidative stress, neuro-inflammation and apoptosis in fluoride treated rats. Neuroscience Letters, 682:92-99.
Ola MS, Ahmed MM, Ahmad R, Abuohashish HM, Al-Rejaie SS, Alhomida AS. 2015. Neuroprotective effects of rutin in streptozotocin-induced diabetic rat retina. Journal of Molecular Neuroscience, 56: 440-448.
Oliveira VM, Carraroa E, Aulerb ME, Khalil NM. 2016. Quercetin and rutin as potential agents antifungal against Cryptococcus spp. Brazilian Journal of Biology, 76(4): 1029-1034.
Ou-yang Z, Caoa X, Weia Y, Zhanga W-W-Q, Zhaoa M, Duan J-a. 2013. Pharmacokinetic study of rutin and quercetin in rats after oral administration of total flavones of mulberry leaf extract. RevistaBrasileira de Farmacognosia, 23: 776-782.
Pashikanti S, de Alba DR, Boissonneault GA, Cervantes-Laurean D. 2010. Rutin metabolites: Novel inhibitors of nonoxidative advanced glycation end products. Free Radical Biology and Medicine, 48: 656-663.
Patil SL, Swaroop K, Kakde N, Somashekarappa HM. 2017. In vitro protective effect of rutin and quercetin against radiation induced genetic damage in human lymphocytes. Indian Journal of Nuclear Medicine, 32:289-295.
Pereira AP, Ferreira ICFR, Marcelino F, Valentão P, Andrade PB, Seabra R, Estevinho L, Bento A, Pereira JA. 2007. Phenolic compounds and antimicrobial activity of olive (Oleaeuropaea L. Cv. Cobrançosa) leaves. Molecules, 12: 1153-1162.
Pimentel RBQ, Costa CA, Albuquerque PM, Junior SD. 2013. Antimicrobial activity and rutin identification of honey produced by the stingless bee Melipona compressipes manaosensis and commercial honey. BMC Complementary and Alternative Medicine, 13:151-163.
Prasad R, Prasad SB. 2018. Antitumor activity of rutin-cisplatin in combination and its protective effect against hematotoxicity. Research Journal of Life Sciences, Bioinformatics, Pharmaceutical and Chemical Sciences, 4(6): 42-56.
Pu F, Mishima K, Irie K, Motohashi K, Tanaka Y, Orito K, Egawa T, Kitamura Y, Egashira N, Iwasaki K, Fujiwara M. 2007. Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. Journal of Pharmacological Sciences, 104(4): 329–334.
Radwan RR, Fattah SMA. 2017. Mechanisms involved in the possible nephroprotective effect of rutin and low dose γ irradiation against cisplatin-induced nephropathy in rats. Journal of Photochemistry and Photobiology, 169: 56-62.
Rai J, Pandey S, Srivastava R. 2004. Testosterone hormone level in albino rats following restraint stress of long duration. Journal of the Anatomical Society of India, 53: 17-19.
Rana AC, Gulliya B. 2019. Chemistry and pharmacology of flavonoids-A review. Indian Journal of Pharmaceutical Education and Research, 53(1): 8-20.
Ravi GS, Charyulu RN, Dubey A, Prabhu P, Hebbar S, Mathias AC. 2018. Nano-lipid complex of rutin: development, characterisation and in vivo investigation of hepatoprotective, antioxidant activity and bioavailability study in rats. AAPS PharmSciTech, 19(8): 3631-3648.
Reddy MK, Reddy AG, Kumar BK, Madhuri D, Boobalan G, Reddy MA. 2017. Protective effect of rutin in comparison to silymarin against induced hepatotoxicity in rats. Veterinary World, 10(1): 74-80.
Ren W, Qiao Z, Wang H, Zhu L, Zhang L. 2003. Flavonoids: Promising anticancer agents. Medicinal Research Reviews, 23(4): 519-534.
Romani A, Mulinacci N, Pinelli P, Vincieri FF, Cimato A. 1999. Polyphenolic content in five Tuscany cultivars of Oleaeuropaea L. Journal of Agricultural and Food Chemistry, 47:964-967.
Sachetto ATA, Rosa JG, Santoro ML. 2018. Rutin (quercetin-3-rutinoside) modulates the hemostatic disturbances and redox imbalance induced by Bothrops jararaca snake venom in mice. PLOS Neglected Tropical Diseases, 12(10): 1-22.
Sadeghnia HR, Yousefsani BS, Rashidfar M, Boroushaki MT, Asadpour E, Ghorbani A. 2013. Protective effect of rutin on hexachlorobutadiene-induced nephrotoxicity. Renal Failure, 35(8): 1151-1155.
Saklani R, Gupta SK, Mohanty IR, Kumar B, Srivastava S, Mathur R. 2016. Cardioprotective effects of rutin via alteration in TNF-a, CRP, and BNP levels coupled with antioxidant effect in STZ-induced diabetic rats. Molecular and Cellular Biochemistry, 420: 65-72.
Saleh A, ElFayoumi HM, Youns M, Barakat W. 2019. Rutin and orlistat produce antitumor effects via antioxidant and apoptotic actions. Naunyn-Schmiedeberg's Archives of Pharmacology, 392: 165-175.
Sattanathan K, Dhanapal CK, Umarani R, Manavalan R. 2011. Beneficial health effects of rutin supplementation in patients with diabetes mellitus. Journal of Applied Pharmaceutical Science, 01 (08): 227-231.
Sawai Y, Kohsaka K, Nishiyama Y, Ando K. 1987. Serum concentrations of rutoside metabolites after oral administration of a rutoside formulation to humans. Arzneimittelforschung, 37: 729-732.
Sharma S, Ali A, Ali J, Sahni JK, Baboota S. 2013. Rutin: therapeutic potential and recent advances in drug delivery. Expert Opinion on Investigational Drugs, 22(8): 1063-1079.
Singh M, Govindarajan R, Rawat AKS, Khare PB. 2008. Antimicrobial flavonoid rutin from Pteris vittata L. against pathogenic gastrointestinal microflora. American Fern Journal, 98(2): 98-103.
Sintayehu B, Bucar F, Veeresham C, Asres K. 2012. Hepatoprotective and free radical scavenging activities of extracts and a major compound isolated from the leaves of Cineraria abyssinica Sch. Bip. exA. Rich. Pharmacognosy Journal, 4(29): 40-46.
Sun CL, Wei J, Bi LQ. 2017. Rutin attenuates oxidative stress and proinflammatory cytokine level in adjuvant induced rheumatoid arthritis via inhibition of NF-kB. Pharmacology, 100 (1-2): 40-49.
Takshak S. 2018.Bioactive compounds in medicinal plants: A condensed review. SEJ Pharmacognosy, (1)1: 1-35.
Tanko Y. Salisu AI, Mohammed KA, Musa SA, Jimoh A, Yusuf R. 2017. Anti-hyperglycaemic effects of rutin on blood glucose, oxidative stress biomarkers and lipid peroxidation in Alloxan-induced hyperglycaemic wistar rats. Nigerian Journal of Physiological Sciences,32: 91-96.
Tiwari SC, Husain N. 2017. Biological activities and role of flavonoids in human health- A review. Indian Journal of Scientific Research, 12 (2): 193-196.
Tongjaroenbuangam W, Ruksee N, Chantiratikul P, Pakdeenarong N, Kongbuntad W, Govitrapong P. 2011. Neuroprotective effects of quercetin, rutin and okra (Abelmoschus esculentus Linn.) in dexamethasone-treated mice. Neurochemistry International, 59: 677-685.
Umar S, Mishra NK, Pal K, Sajad M, Neha, Ansari MM, Ahmad S, Katiyar CK, Khan HA. 2012. Protective effect of rutin in attenuation of collagen-induced arthritis in Wistar rat by inhibiting inflammation and oxidative stress. Indian Journal of Rheumatology, 7(4): 191-198.
Umarani V, Sudhakar M, Ramesh A, Lakshmi BVS,Mahipal K. 2015. Protective effects of rutin against sodium fluoride induced nephrotoxicity and blood toxicity in rats. International Journal of Advances in Pharmacy Medicine and Bioallied Sciences, 3(3): 116-121.
Vadapalli U, Muvvala S, Alluri R, Lakshmi B.V.S. 2017. Anti-proliferative activity of rutin on Hela cell line induced cervical cancer in rats. International Journal of Pharmaceutical Sciences and Research, 8(11): 4803-4811.
Verma AK, Johnson JA, Gould MN, Tanner MA. 1988. Inhibition of 7, 12-dimethylbenz (a) anthracene- and N-nitrosomethylurea-induced ratmammary cancer by dietary flavonolquercetin. Cancer Research, 48: 5754-5758.
Wada L, Boxin O. 2002. Antioxidant activity and phenolic content of Oregon caneberries. Journal of Agricultural and Food Chemistry, 50:3495-3500.
Wang S, Wang C, Gao L, Cai H, Zhou Y, Yang Y, Xu C, Ding W, Chen J, Muhammad I, Chen X, He X, Liu D, Li Y. 2017. Rutin inhibits Streptococcus suis biofilm formation by affecting CPS biosynthesis. Frontiers in Pharmacology, 8(379): 1-12.
Wang S, Wang Y, Su Y, Zhou W, Yang S, Zhang R, Zhao M, Li Y, Zhang Z, Zhan D, Liu R. 2012. Rutin inhibits β-amyloid aggregation and cytotoxicity, attenuates oxidative stress, and decreases the production of nitric oxide and pro-inflammatory cytokines. Neurotoxicology, 33(3): 482-490.
Wang YD, Zhang Y, Sun B, Leng XW, Li YJ, Ren LQ. 2018. Cardioprotective effects of rutin in rats exposed to pirarubicin toxicity. Journal of Asian Natural Products Research, 20(4): 361-373.
Wei S-M, Yan Z-Z, Zhou J. 2011. Protective effect of rutin on testicular ischemia-reperfusion injury. Journal of Pediatric Surgery, 46(7): 1419-1424.
WHO. (2004). WHO Guidelines on Safety Monitoring of Herbal Medicines in Pharmacovigilance Systems. Geneva, Switzerland: World Health Organization.
Xianchu L, Lan Z, Ming L, Yanzhi M. 2018. Protective effect of rutin on lipopolysaccharide-induced heart injury in mice. Journal of Toxicological Sciences, 43(5): 329-337.
Yang C-H, Hsiu S-L, Wen K-C, Lin S-P, Tsai S-Y, Hou Y-C, Chao P-DL. 2005. Bioavailability and metabolic pharmacokinetics of rutin and quercetin in rats. Journal of Food and Drug Analysis, 13(3): 244-250.
Yang J, Guo J, Yuan J. 2008. In vitro antioxidant properties of rutin. LWT - Food Science and Technology, 41(6): 1060-1066.
Yoo H, Ku S-K, Baek Y-D, Bae J-S. 2014. Anti-inflammatory effects of rutin on HMGB1-induced inflammatory responses in vitro and in vivo. Inflammation Research, 63: 197-206.