

G. Jyothi Reddy1*, K. Bhaskar Reddy2, G. V. Subba Reddy3
1Research Scholar, JNTUA, Ananthapuramu, Andhra Pradesh, India.
2Sri Venkateswara College of Pharmacy, RVS Nagar, Chittoor, Andhra Pradesh, India
3Department of Chemistry, JNTUA, Pulivendula, Andhra Pradesh, India
*Address for Corresponding Author
G. Jyothi Reddy,
Academic Consultant, Department of Pharmacology,
SVU College of Pharmaceutical Sciences, SV University, Tirupati, 517502, India.
Abstract
Background: Postprandial hyperglycemia (PPHG) has been proposed as an independent risk factor for cardiovascular diseases (CVD). Therefore, control of PPHG is suggested to be important in the management of diabetes mellitus and related secondary complications. Inhibition of α-amylase and α-glucosidase enzymes can be an important antidiabetic approach for reducing PPHG in type 2 diabetes melletus. Acarbose, voglibose and miglitol are the typical examples of inhibitors used in the clinical practice. However, these drugs have major drawbacks. Hence the purpose of the present study is to explore natural and safer α-amylase and α-glucosidase inhibitors. Objectives: To evaluate the in vitro antidiabetic activity of aqueous and methanolic extracts of Feronia elephantum fruit and Paspalum scrobiculatum grains. Materials and methods: aqueous and methanolic extracts of Feronia elephantum fruit and Paspalum scrobiculatum grains were assayed by using α-amylase and α-glucosidase enzyme inhibition assays. Results: All the extracts exhibited a dose-dependent inhibitory activity against and α-glucosidase enzymes. The α-amylase IC50 values of methanolic and aqueous extracts of F.elephantum fruit and P.scrobiculatum grains were 119.698, 167.505, 82.107 and 149.886μg/mL respectively which were comparable to that of standard drug acarbose, 109.722μg/mL. The α-glucosidase IC50 values of methanolic and aqueous extracts of F.elephantum fruit and P.scrobiculatum grains were 66.738, 84.548, 47.297 and 118.351μg/mL respectively, with Acarbose showing an IC50 of 52.645μg/ml. Conclusions: Findings of in vitro antidiabetic assay clearly indicate that F.elephantum fruit and P.scrobiculatum grains possesses considerable inhibitory activity against α-amylases and α-glucosidases, and can be of potential use in diabetic patients.
Keywords: α-amylase, α-glucosidase, Feronia limonia, Paspalum scrobiculatum, invitro antidiabetic
Introduction
Lifestyle of world’s population is influenced with modernization, changed dietary habits and concomitant rise in the stress level. They suffer from many life-threatening diseases like hypertension, cardiac diseases, cancer, diabetes mellitus (DM), obesity, depression and many others (Shomali, 2012). DM is characterized by circulating hyperglycemia resulting from either defect in insulin secretion or insulin action or both (Joslin, 2005). According to the international diabetes federation (IDF), it was estimated that more than 425 million people worldwide had diabetes in 2017. Its prevalence is increasing rapidly, particularly in developing countries, and is expected to rise to 552 million by 2030 (IDF, 2017).
Currently, antidiabetic therapy, other than insulin supplement includes usage of oral hypoglycemic agents viz., thiazolidines, sulfonylureas, meglitinides, biguanides, D-phenylalanine derivatives and α-glucosidase inhibitors along with diet, exercise and weight loss. However, none of them are considered to be ideal, due to prominent side-effects and sometimes diminished response after long term use (Chattopadhyay, 1999), and it has become the main reason for an increasing number of people seeking alternative therapies that may have no side-effects.
Postprandial hyperglycemia (PPHG) is a condition where there is elevation in blood glucose level after consuming meal, and has been proposed as an independent risk factor for cardiovascular diseases (CVD). Therefore, control of PPHG is suggested to be important in the management of diabetes mellitus and related secondary complications such as CVD, diabetic retinopathy, diabetic neuropathy, etc. (Gin and Rigalleau, 2000).
The enzyme α-amylase, which is a digestive enzyme secreted from salivary gland and pancreas, is responsible for the hydrolysis of 1,4-glycosidic linkages of dietary starch and other oligosaccharides into maltose, maltriose and other simple sugars prior to absorption (Cazarolli et al., 2008). The enzyme α-glucosidase, is one among a number of glucosidases located in the brush-border surface on the membrane of intestinal cells and, is a key enzyme of carbohydrate digestion. α-glucosidase inhibitors block the action of enzyme in the small intestine, which is rate limiting in the conversion of oligosaccharides to monosaccharides, which is necessary for gastrointestinal absorption of carbohydrates. Thus, inhibition of these enzymes prolong the overall carbohydrate digestion time leading to a marked reduction in the rate of glucose absorption thereby blunting blood glucose level (Rhabasa and Chiasson, 2004), and hence, could be an important antidiabetic approach for reducing postprandial hyperglycemia in type 2 diabetes melletus (T2DM) patients. Acarbose, oglibose and miglitol are the typical examples of inhibitors used in the clinical practice (Bailey, 2003). However, these drugs have major drawbacks including various gastrointestinal side effects viz., abdominal discomfort, bloating, flatulence and diarrhoea (Cheng and Fantus, 2005). Thus, efforts are needed to explore natural and safer α-amylase and α-glucosidase inhibitors.
Ayurveda and other ancient Indian literature have reported the use of numerous herbal plants and their preparations in the treatment of diabetes (Randive et al., 2016; Grover et al., 2002). Some antidiabetic plants may exert the hypoglygemic action by either stimulating the function or number of pancreatic β-cells thus increasing the insulin release. In some other plants, the effect may be due to decrease in blood glucose synthesis by deferring the activity of enzymes. In other plants, the activity may be to slowdown absorption of carbohydrates and inhibition of glucose transport (Ali et al., 2006). Many plants with bioactive constituents like flavonoids, phenols, saponins etc exhibited potential α-amylase and α-glucosidase inhibitory activity, which is attributed to their antioxidant properties (Rathi Sre et al., 2012).
Feronia elephantum correa (Synonym: Limonia acidissima, Feronia limonia, or Schinus limonia), family Rutaceae, used not only to treat diabetes but also several other clinical conditions by traditional healers in rural areas (Joshi et al., 2009). The fruit (wood apple) contains flavanoids, glycosides, saponins and tannins (Saima, 2000). Some coumarins (Ghosh et al., 1982; Chatterjee et al., 1980) and tyramine derivatives (Parthasarathi et al., 1991) have also been isolated. Fruit pulp is a rich source of Beta carotene, which is a precursor of vitamin-A, and also contains significant amount of vitamins such as riboflavin and thiamine, and it had small quantities of ascorbic acid content (Sachan Kumar et al., 2004).
Paspalum scrobiculatum Linn., family Poaceae, commonly known as ‘Kodo millet’, contains phenolic acids like vanillic acid, cis-ferulic acid, melilotic acid, p-hydroxy benzoic acid, syringic acid, and flavonoids (Sanjay Jain et al., 2010). The grains are styptic, sweet and tonic and are effective in controlling haemorrhages, inflammation, general debility, and hepatopathy (Kiran et al., 2014). Ayurvedic texts like Charak Samhita and Sushruta Samhita, have reported the usefulness of these grains for the management of diabetes mellitus (The Wealth of India, 2003; Murty et al., 1989).
Thus, the present study aimed to evaluate the In vitro antidiabetic activity of aqueous and methanolic extracts of Feronia elephantum fruit and Paspalum scrobiculatum grains by using α-amylase and α-glucosidase enzyme inhibition assays.
Materialas and methods
Chemicals and reagents
3,5-Dinitro Salicylic Acid (DNSA), p-nitrophenyl-α-D-glucopyranoside (pNPG), Acarbose, α-amylase and α-glucosidase enzymes were purchased from Sigma–Aldrich Chemicals Pvt Limited, (Bangalore, India). All other chemicals and reagents used were of high purity analytical grade and purchased from Merck Pvt. Ltd. (India).
Collection and authentication of plants
The grains of P.scrobiculatum and ripe fruits of F.elephantum were collected from a local market in Tirupati, Andhra pradesh, during the month of September 2017. The plants were authenticated by Dr. K. Madhava Chetty, Department of Botany, Sri Venkateswara University (SVU), Tirupathi, Andhra Pradesh, and voucher specimens were deposited at that Institute (Voucher number: 2032 and 1328). The fruits of F.elephantum were washed thoroughly and separated into pulp and rind, the fruit pulp with embedded seeds was shade-dried. The grains of P.scrobiculatum and fruit pulp of F.elephantum were coarsely powdered and furthur used for extraction.
Preparation of extracts
The aqueous extracts of P.scrobiculatum grains and F.elephantum fruit were prepared by cold maceration of 150 g of powdered materials individually in 500 mL of distilled water for 7 days. The methanolic extracts of P.scrobiculatum grains and F.elephantum fruit were prepared by soxhlet extraction method by taking 100 g of the powdered material and extracting with 400 mL of methanol. Both the extracts were filtered, concentrated, dried in incubator (37◦C). The extracts were stored in a refrigerator at 2–8°C for use in subsequent experiments.
In vitro α-amylase inhibitory assay
The In vitro α-amylase inhibition assay of all extracts was performed with slight modification of a previously reported method which is based on the spectrophotometric assay using acarbose as the reference compound (Barapatre et al., 2015). Stock solutions of plant extracts of F.elephantum and P. scrobiculatum and positive control sample of acarbose were prepared in distilled water. A total of 500μL of different concentrations of each sample (25, 50, 100, 200 and 250µg/mL) were added to a 500μL α-amylase solution (0.5 mg/mL in 0.02M sodium phosphate buffer, pH 6.9) and incubated for 10 min at. Then 500 μL of 1% (w/v) starch solution was added and incubated for another 10 min at 25°C. Then the colouring reagent, DNSA (1 mL) was added, and the reaction mixture was heated in a boiling water bath for 5 minutes and cooled to room temperature. It was diluted with 10 mL distilled water and the absorbance was measured at 540 nm using a UV-VIS spectrophotometer (UV-1800; Shimadzu). The absorbance of blank was prepared by replacing enzyme solution with 500μl buffer. A positive control, representing 100% enzyme activity was prepared using acarbose (100 μg/mL–500 μg/mL) in a similar procedure, without plant extract as mentioned above. Repeat the experiments thrice using the same protocol.
In vitro α-glucosidase inhibition assay
Inhibition of α-glucosidase activity was determined using p-nitrophenyl-α-D-glucopyranoside (pNPG) as described before (Kim 2011). 100μL of plant extract or acarbose (25, 50, 100, 200 and 250µg/mL) was added to 50μL of α-glucosidase (effective concentration 3.2.1.20; 1 U/mL) prepared in 0.1 M phosphate buffer (pH 6.9). Then, add 250μL of 0.1 M phosphate buffer to get the final concentrations. The mixture was pre-incubated at 37°C for 20 min. After pre-incubation, 10μl of 10mM pNPG prepared in 0.1M phosphate buffer (pH 6.9) was added, and incubated at 37°C for 30 min. The reactions were stopped by adding 650μl of 1M sodium carbonate, and the absorbance was measured in a spectrophotometer at 405nm. The absorbance of blank with 100% enzyme activity (only the solvent with the enzyme) was measured. Acarbose was used as positive control. Repeat the experiments thrice using the same protocol.
Method for calculation of α-amylase and α-glucosidase inhibitory concentration (IC50):
The percentage enzyme inhibition of the extracts/standard was calculated using the following formula shown below, and the values were presented as mean±standard error mean of three replicates.
% inhibition = (A control – A sample / A control) x 100
Where “A control” is the absorbance of the control and “A sample” is the absorbance of the sample. The concentration of the extracts required to inhibit 50% of α-amylase and α-glucosidase activity under the assay conditions was defined as the IC50 value (Sium et al., 2017). IC50 was calculated by using the percentage inhibition activities at five different concentrations of the extract, by using probit regression (dose–response curve) using SPSS20.0 software.
Results
Evaluation of in vitro α-amylase inhibitory activity of plant extracts
Four extracts prepared from two plants were screened by in vitro α-amylase inhibition assay, which might be extrapolated to detect their antidiabetic potential. There was a concentration dependent increase in the percentage inhibitory activity against α-amylase enzyme (Table 1). Methanolic extract of F.elephantum fruit (MEFE), Aqueous extract of F.elephantum fruit (AEFE), Methanolic extract of P.scrobiculatum grains (MEPS), Aqueous extract of P.scrobiculatum grains (AEPS) and acarbose at a concentration 100μg/mL showed a percentage inhibition of 39.05±1.02, 26.21±0.75, 50.86±1.03, 29.02±0.64 and 38.72±1.08 respectively, and at a maximum concentration of 250μg/mL it was 86.92±2.32, 72.36±1.93, 92.87±2.96, 79.12±1.72, and 94.67±1.89 respectively (Figure 1). The IC50 values of MEFE, AEFE, MEPS and AEPS were 119.698, 167.505, 82.107 and 149.886μg/mL respectively. The IC50 value of standard drug acarbose was found to be 109.722μg/mL.
Table 1. α -Amylase and α-glucosidase inhibition assay of various extracts of F.elephantum and P.scrobiculatum
|
Sample |
Concentration (μg/mL) |
% inhibition of α-amylase activity |
IC50 (𝜇g/mL) |
% inhibition of α-glucosidase activity |
IC50 (𝜇g/mL) |
|
Methanolic extract of F.elephantum (MEFE) |
25 50 100 200 250 |
9.16± 0.61 17.29± 0.59 39.05±1.02 60.97± 1.37 86.92± 2.32 |
119.698 |
21.14 ± 1.75 33.99 ± 1.84 68.41 ± 2.30 79.04 ± 1.95 92.34 ± 3.31 |
66.738 |
|
Aqueous extract of F.elephantum (AEFE) |
25 50 100 200 250 |
7.89± 0.35 13.91± 0.43 26.21± 0.75 51.02± 1.09 72.36± 1.93 |
167.505 |
18.89±0.59 26.50±0.85 49.45±1.12 75.32±1.29 91.60±0.97 |
84.548 |
|
Methanolic extract of P.scrobiculatum (MEPS) |
25 50 100 200 250 |
15.67± 0.84 28.53± 1.21 50.86± 1.03 79.24± 1.68 92.87± 1.96 |
82.107 |
28.07±1.03 49.08±1.40 79.27±1.76 91.04±1.32 93.65±2.35 |
47.297 |
|
Aqueous extract of P. scrobiculatum (AEPS) |
25 50 100 200 250 |
5.49± 1.05 14.93± 0.75 29.02± 0.64 53.36± 1.32 79.12± 1.72 |
149.886 |
11.57±0.87 19.98±0.93 39.45±1.72 68.04±1.24 77.60±1.96 |
118.351 |
|
Acarbose |
25 50 100 200 250 |
17.90± 1.05 20.21± 1.24 38.72± 1.08 79.93± 1.43 94.67± 1.89 |
109.722 |
22.12±0.93 43.58±1.09 81.23±1.02 90.64±1.13 97.17±1.35 |
52.645 |
All determinations were carried out in triplicate manner and values are expressed as the mean ± SEM
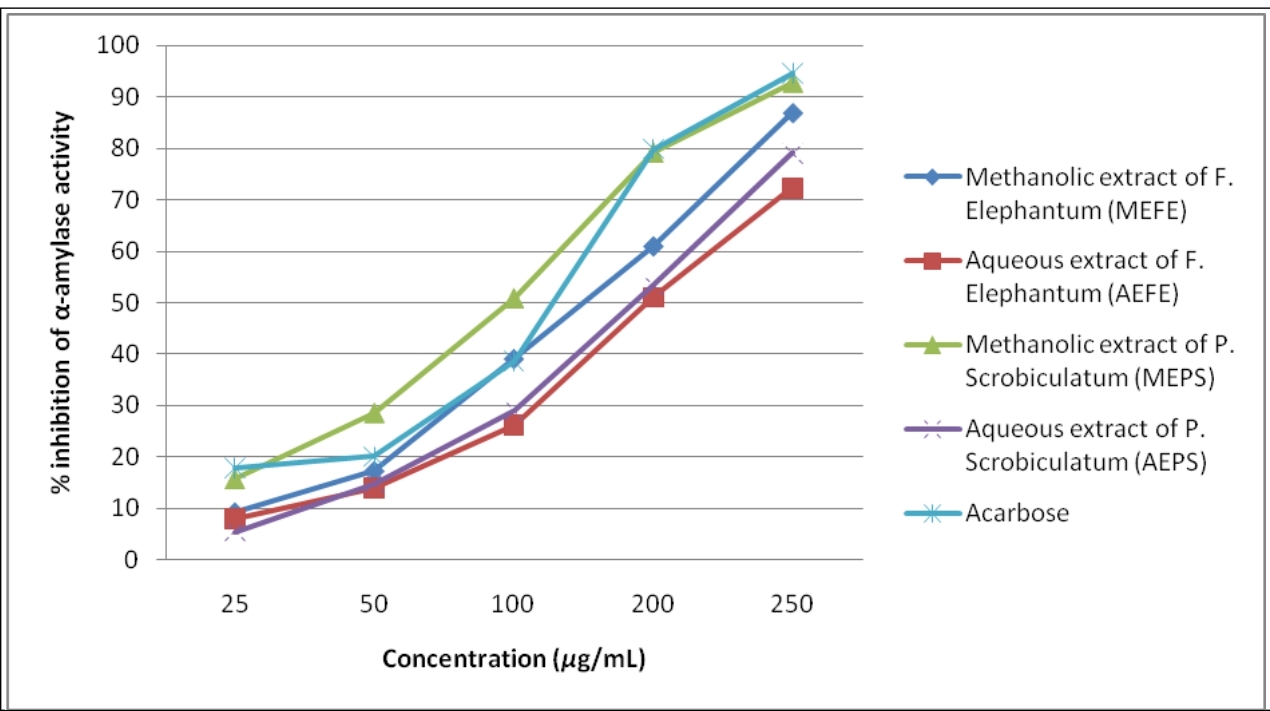
Figure 1. α-Amylase inhibitory activity of plant extracts
Evaluation of in vitro α-glucosidase inhibitory activity
There was a dose-dependent increase in the percentage inhibitory activity against α-glucosidase enzyme. MEFE, AEFE, MEPS and AEPS and acarbose at a concentration 100μg/mL showed a percentage inhibition of 68.41 ± 2.30, 49.45±1.12, 79.27±1.76, 39.45±1.72 and 81.23±1.02 respectively (Table 1). The IC50 values of MEFE, AEFE, MEPS and AEPS were 66.738, 84.548, 47.297 and 118.351μg/mL respectively. The standard positive control, acarbose showed an IC50 of 52.645μg/ml. In this study, significant differences were found between the IC50 values of all extracts (Figure 2). However, the IC50 of acarbose and the methanol extracts of both the plants did not show a significant difference.
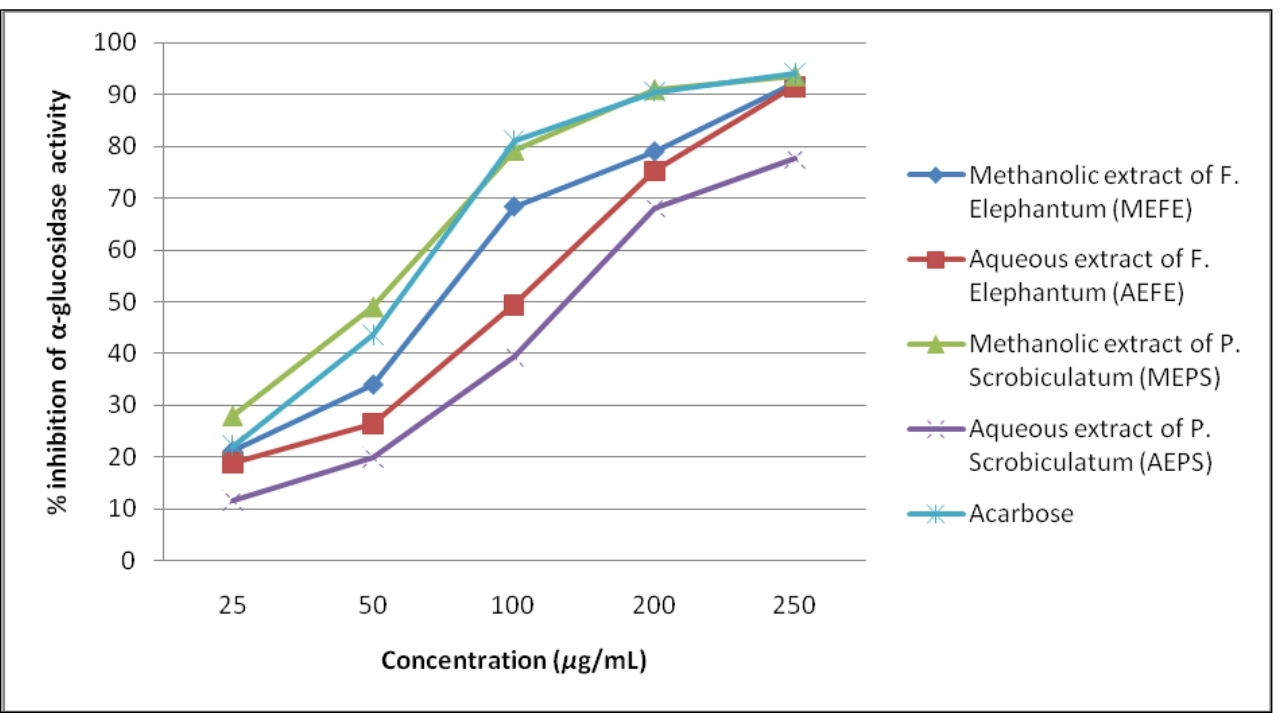
Figure 2. α-Glucosidase inhibitory activity of plant extracts
Discussion
In diabetic patients, high postprandial blood glucose level is mainly responsible for micro and macro-vascular complications than that of fasting blood glucose levels. It is well known that suppression of starch digestive enzymes reduce elevation of postprandial blood glucose level (Truscheit et al., 1981). Hence, one of the therapeutic approaches for minimizing postprandial (PP) blood glucose level in diabetic patients is to reduce glucose production by impeding its breakdown and intestinal absorption, by inhibiting the enzymes α-amylases and α-glucosidases after food intake.
The present study demonstrated that both methanolic and aqueous extracts of F.elephantum fruit and P.scrobiculatum grains had significant α-amylase and α-glucosidase inhibitory potentials. The IC50 values of all the extracts were comparable to that of acarbose, which is a marketed anti-diabetic drug.
The inhibitory potential of the extracts on α-amylase activity may be a possible mechanism by which the extracts exert antidiabetic activity. Since α-amylases play vital role in starch assimilation in human beings and animals, the presence of such inhibitors in foodstuffs or plant extracts may be responsible for impaired starch digestion (Puls et al., 1973). This α-amylase inhibitory activity of the extracts may be of value as novel therapeutic agents.
Similarly, the α-glucosidase inhibitory effect exhibited by all the extracts indicates their potential effectiveness at managing T2DM attributed by reduction in both postprandial glycemic levels and the total range of postprandial glucose levels (Rhabasa and Chiasson, 2004).
F.elephantum fruit efficiently inhibits α-amylase and α-glucosidase enzymes in vitro, which might be due to the presence of many phenolic compounds, flavonoids, coumarins etc (Kumar and Sachitra, 2004).
P. scrobiculatum has been reported for the presence of many phenolic acids, dietary fibres and flavonols like quercetin. quercetin was found to possess in vitro antidiabetic property, by reducing intestinal glucose absorption (Hoda et al., 2017). These phytoconstituents might be responsible for exerting a good inhibitory action against α-amylase and α-glucosidase enzymes, thus preventing diabetes mellitus. Hence these plants can be included as part of dietary management of the desease.
Conclusion
Findings of the in vitro antidiabetic assay clearly indicate that F.elephantum fruit and P.scrobiculatum grains possesses considerable inhibitory activity against α-amylases and α-glucosidases, with remarkable activity in the methanolic extracts. This observed antidiabetic activity might be attributed to bioactive compounds like phenols, flavonoids, etc. F.elephantum fruit and P.scrobiculatum grains could be greatly beneficial in reducing post-prandial blood glucose levels in diabetic patients and thus, can be effectively used in ayurvedic treatments in the management of diabetics. This needs further in-vivo investigations in animal models and identification of specific bioactive compounds to develop novel effective anti-diabetic drugs that are free from harmful side effects.
Conflicts of interest: None
References
Ali H, Houghton PJ, Soumyanath A. 2006. α-Amylase inhibitory activity of some Malaysian plants used to treat diabetes; with particular reference to phyllanthus amarus. Journal of Ethnopharmacology 107: 449-55.
Bailey CJ. 2003. New Approaches to the Pharmacotherapy of Diabetes. Volume 2, 3rd ed. 73.1-73.21, UK, Blackwell Science Ltd.
Barapatre A, Aadil KR, Tiwary BN, Jha H. 2015. In vitro antioxidant and antidiabetic activities of biomodified lignin from Acacia nilotica wood. International Journal of Biological Macromolecules 75:81-9.
Cazarolli LH, Zanatta L, Alberton EH, Figueiredo MS, Folador P, Damazio RG. 2008. Flavonoids: prospective drug candidates. Mini -Reviews in Medicinal Chemistry 8:1429-1440.
Chatterjee A, Sankar S, Shoolery JN. 1980. 7-Phenyl acetoxy Coumarin from Limonia crenulata. Phytochemistry 19: 2219-2220.
Chattopadhyay RR. 1999. A comparative evaluation of some blood sugar lowering agents of plant origin. Journal of Ethnopharmacology 67(3): 367-372.
Cheng AYY, Fantus IG. 2005. Oral antihyperglycemic therapy for type 2 diabetes mellitus. Canadian Medical Association Journal 172 (2): 213-226.
Ghosh P, Sil P, Majumdar SG, Thakur S. 1982. A Coumarin from Limonia acidissima, Phytochemistry 21: 240-241.
Gin H, Rigalleau V. 2000. Post-prandial hyperglycemia. Post-prandial hyperglycemia and diabetes. Diabetes & Metabolism 4:265-72.
Grover JK, Yadav S, Vats V. 2002. Medicinal plants of India with antidiabetic potential. Journal of Ethnopharmacology 81(1): 81-100.
Hoda M. Eid, Pierre S. Haddad. 2017. The Antidiabetic Potential of Quercetin: Underlying Mechanisms. Current Medicinal Chemistry 24(4): 355-364.
IDF Diabetes Atlas. 2017. 8th ed. International Diabetes Federation.
Joshi RK, Patil PA, Muzawar MHK, Kumar D, Kholkute SD. 2009. Hypoglycemic activity of aqueous leaf extract of feronia elephantum in normal and streptozotocin-induced diabetic rats. Pharmacologyonline 3: 815-821.
Joslin's Diabetes Mellitus. 2005. Lippincott Williams & Wilkins. pp 1-17. 4. Boston: Massachusetts.
Kim JS, Hyun TK, Kim MJ. 2011. The inhibitory effects of ethanol extracts from sorghum, foxtail millet and proso millet on α-glucosidase and α-amylase activities. Food Chemistry 124: 1647-51.
Kiran P, Denni. M, Daniel M. 2014. Antidiabetic Principles, Phospholipids and Fixed Oil of Kodo Millet (Paspalum scrobiculatum Linn.). Indian Journal of Applied Research 4(2): 13-15.
Rhabasa L, Chiasson JL. 2004. Alpha-Glucosidase Inhibitors, 13rd ed, pp. 901-904, UK, John Wiley & Sons Ltd.
Murty AVS, Subramanyam NSA. 1989. N.S.A. Textbook of Economic Botany: New Delhi: Wiley Eastern Limited.
Parthasarathi G, Prabal S, Srabani D, Swapnadip T, Kokke WCMC, Akihisa T. 1991. Tyramine Derivatives from the Fruit of Limonia acidissima. Journal of Natural Products 54: 1389-1393.
Puls W, Keup U. 1973. Influence of an α-amylase inhibitor (BAY d 7791) on blood glucose, serum insulin and NEFA in starch loading tests in rats, dogs and man. Diabetologia 9: 97-101.
Randive DS, Sayyad SF, Bhinge SD, Bhutkar MA. 2016. Preparation of Arjunarishta using microbes isolated from Woodfordia fruticosa flowers (Dhayati). Ancient Science of life 36 (1):42-47.
Rathi Sre PR, Sheila T, Kandaswamy M. 2012. Phytochemical screening and In vitro antioxidant activity of methanolic root extract of Erythrina indica. Asian Pacific Journal of Tropical Biomedicine 2: 1696-700.
Rhabasa L, Chiasson JL. 2004. Alpha-Glucosidase Inhibitors. Volume 1, 3rd ed, pp. 901-904, UK, John Wiley & Sons Ltd.
Kumar S, Sachitra P. 2004. Ayurveda. p. 374-376, Patna, Shri Baidyanath Ayurved bhavan Pvt. Ltd.
Saima Y, Das AK, Sarkar KK, Sen AK, Sur P. 2000. An antitumor pectic polysaccharide from Feronia limonia. International Journal of Biological Macromolecules 27: 333-335.
Jain S, Bhatiaa G, Barika R, Kumarb P, Jain A, Dixit VK. 2010. Antidiabetic activity of Paspalum scrobiculatum Linn. in alloxan induced diabetic rats, Journal of Ethnopharmacology 127:325-328.
Shomali M. 2012. Diabetes treatment in 2025: can scientific advances keep pace with prevalence? Therapeutic Advances in Endocrinology and Metabolism 3:163-173.
Sium M, Kareru P, Kiage-Mokua B, Sood K, Langley J, Herniman J. 2017. In vitro Anti- Diabetic Activities and Phytochemical Analysis of Bioactive Fractions Present in Meriandra dianthera, Aloe camperi and a Polyherb. American Journal of Plant Sciences 8: 533-548.
The Wealth of India, 2003. P. scrobiculatum. In: A Dictionary of Indian Raw Materials and Industrial Products. p. 284. CSIR, New Delhi, India,.
Truscheit E, Frommer W, Junge B, Muller L, Schmidt DD. 1981. Chemistry and biochemistry of microbial a-glucosidase inhibitors. Angewandte Chemie International Edition 20: 744-761.