

Sanman Samova*, Hetal Doctor, Dimple Damore, Ramtej Verma
Department of Zoology, BMTC and Human Genetics, School of Sciences, Gujarat University, Ahmedabad, India
*Address for Correspondence:
Sanman Samova
Department of Zoology,
University School of Sciences, Gujarat University, Ahmedabad-380009, India
Abstract
Objective: Bisphenol A is an endocrine disrupting chemical, widely used as a material for the production of epoxy resins and polycarbonate plastics. Food is considered as the main source of exposure to BPA as it leaches out from the food containers as well as surface coatings into it. BPA is toxic to vital organs such as liver kidney and brain. Quercetin, the most abundant flavonoid in nature, is present in large amounts in vegetables, fruits and tea. The aim of the present study was to evaluate the toxic effects of BPA in prostate gland and seminal vesicle of mice and its possible amelioration by quercetin. Material and methods: Inbred Swiss strain male albino mice were orally administered with BPA (80, 120 and 240 mg/kg body weight/day) for 45 Days. Oral administration of BPA caused significant, dose-dependent reduction in absolute and relative weights of prostate gland and seminal vesicle. Results and conclusion: Biochemical analysis revealed that protein content reduced significantly, whereas acid phosphatase activity increased significantly in prostate gland and reduction in fructose content was observed in seminal vesicle. Oral administration of quercetin (30, 60 and 90 mg/kg body weight/day) alone with high dose of BPA (240 mg/kg body weight/day) for 45 days caused significant and dose-dependent amelioration in all parameters as compared to BPA along treated group. Histopathological alterations were also ameliorated by quercetin. BPA causes toxicity in prostate gland and seminal vesicle of mice and quercetin is potent enough to ameliorate BPA induced toxicity.
Keywords: Bisphenol A, male accessory glands, prostate gland, seminal vesicle, biochemical analysis, histopathology
Introduction
Bisphenol A (2, 2-bis 4-hydroxyphenyl propane) is a xenoestrogenic organic endocrine-disrupting chemical (Dodds and Lawson, 1936) able to bind estrogen receptors (Takayanagi et al., 2006; Chapin et al., 2008). It is one of the highest volume chemical produced worldwide with a total of 2.8 million metric tons manufactured in 2002 that increased to 5.5 million metric tons in 2011 (Rochester, 2013). A large body of data has shown that BPA, is mainly used for the production of polycarbonate plastics, epoxy resins and non-polymer additives (Kuruto-Niwa et al., 2007; Grumetto et al., 2008). Food is considered as a main source of exposure to BPA as a consequence of its migration from food containers. (Ranjit et al., 2010) Bisphenol-A is toxic to vital organs such as liver, kidney and brain. (Kabuto et al., 2003; Bindhumol et al., 2003; Sangai et al., 2012) According to studies, even at very low doses, BPA mimic estrogen resulting in an array of health maladies including prostate (Prins et al., 2007) as well as breast cancers (Pupo et al., 2012), genotoxic effect (Karim and Husain, 2010) and other health problems. (Vom Saal et al., 2007).
Quercetin, the most abundant flavonoid in nature, is present in large amounts in vegetables, fruits, tea and olive oil, and contains a number of phenolic hydroxyl groups. (Wach et al., 2007) It was reported that quercetin has many beneficial effects in human health, including cardiovascular protection, (Grassi et al., 2010) anti-canceractivity, (Sak, 2014) anti-ulcer effects, (Suzuki et al., 1998) anti-allergy activity, (Bellik et al., 2013) cataract prevention, (Cornish et al., 2002) antiviral activity and anti-inflammatory effects. (Guardia et al., 2001) These effects of quercetin are due to its antioxidant properties of scavenging free radicals directly. (Afanas' et al., 1989; Robak et al., 1988)
The majority of studies on BPA have focused on the potential adverse effects on vital organs. However, there is insufficient data available on the effect of BPA on the accessory reproductive organs especially of males in mammals. The aim of the present study was to evaluate the toxic effects of BPA in prostate gland and seminal vesicle of mice and its possible amelioration by quercetin.
Materials and methods
Chemicals
Bisphenol A and quercetin was purchased from Hi Media Laboratories Pvt. Ltd., Mumbai, India and was of analytical grade. Olive oil was obtained from Figaro, Madrid, Spain. All the other chemicals used in the present study were of AR grade and procured from Hi Media Laboratories Pvt. Ltd., Sisco Research Laboratories Pvt. Ltd., Mumbai, India and Sigma-Aldrich St. Louis, MO, USA.
Experimental animals
In the experiment, inbred healthy adult Swiss strain male albino mice weighing 35-40 gm obtained from Cadila pharmaceutical Center, Ahmedabad, India, were kept in the Animal House of Zoology Department of Gujarat University, Ahmedabad, India. They were housed in an air-conditioned room at a temperature of 25±2ºC and 50-55% relative humidity with a 12 h light/dark cycle throughout the experiment. Animals were fed with certified pelleted rodent feed supplied by Amrut Feeds, Pranav Agro Industries Ltd., Pune, India and potable water ad libitum. All the experimental protocols were approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (Reg. – 167/1999/CPCSEA), New Delhi, India. Animals were handled According to the guidelines published by Indian National Science Academy, New Delhi, India (1991).
Table 1. Experimental protocol
|
Experimental Groups |
Numbers of animal |
Duration of treatment (Days) |
Day of Necropsy |
|
|
Control groups |
||||
|
I |
Untreated control |
10 |
45 |
46th |
|
II |
Vehicle control (0.2 ml olive oil/animal/day) |
10 |
45 |
46th |
|
III |
Antidote control (90 mg quercetin /kg body weight/day) |
10 |
45 |
46th |
|
BPA -treated groups |
||||
|
IV |
BPA-Low dose (80 mg/kg body weight/day) |
10 |
45 |
46th |
|
V |
BPA-Medium dose (120mg /kg body weight/day) |
10 |
45 |
46th |
|
VI |
BPA-High dose (240 mg/kg body weight/day) |
10 |
45 |
46th |
|
HD BPA(240 mg/kg body weight/day) + quercetin treated groups |
||||
|
VII |
BPA-HD + Q30 (30 mg/kg body weight/day) |
10 |
45 |
46th |
|
VIII |
BPA-HD + Q60 (60 mg/kg body weight/day |
10 |
45 |
46th |
|
IX |
BPA-HD + Q90 (90 mg/kg body weight/day) |
10 |
45 |
46th |
Dose selection
Different doses of BPA was selected on the bases of LD50 value. (MSDS 2004) Animals of BPA-treated groups received three different doses of BPA, which is 1/10th, 1/20th and 1/30th of LD50 (240,120 and 80 mg/kg bw/day respectively) for 45 days for the amelioration of BPA-induced toxicity different doses of quercetin (30, 60 and 90 mg/kg bw/day) were selected on the basis of the study of Sangai and Verma (Sangai et al., 2012).
Experimental design
Experimental protocol is shown in table 1. Mice were randomly divided into nine groups, each containing10 animals. Treatment schedule of the animals was as follows. Animals from group I (untreated control) were kept untreated and given free access to feed and water. Group II (vehicle control) animals were administered with olive oil (0.2 ml/animal/day), which has been used as vehicle to dissolve BPA. Group III (antidote control) animals were treated with quercetin (90mg/kg bodyweight/day), which has been used for amelioration of BPA-induced toxicity.
Animals of group IV, V and VI received three different doses of BPA (80, 120 and 240 mg/kg bw/day) for 45 days. However animals of group VII, VIII and IX received three different doses of quercetin (30, 60 and 90 mg/kg bw/day) along with high dose of BPA (240 mg/kg bw/day). All treatments were given orally using a feeding tube attached to hypodermic syringe.
On completion of treatment, the animals were sacrificed by using anesthetic ether. The prostate gland and seminal vesicle were isolated, blotted free from blood, weighted and used for the biochemical analysis such as protein and ACP in prostate and Fructose in seminal vesicle. Tissue was stored in 10% formalin for histopathological study.
Analysis of biochemical parameters
Protein content in prostate gland was estimated by the method of Lowry et al. (1951) and expressed as mg/100 mg tissue weight. The acid phosphatase (ACP) activity in the prostate gland was assayed by the method as described in Sigma Technical Bulletin (Sigma Technical Bulletin, MO, USA. The enzyme activity was expressed as μmoles p-nitrophenol released/mg protein/30 min. The concentration of fructose in tissue of seminal vesicle was estimated by the method of Foreman et al. (1973). The fructose content was expressed as μg/mg tissue weight.
Histopathological study
Histopathological studies were carried out using the standard technique of hematoxylin and eosin (H & E) staining. Prostate and seminal vesicle tissues of all control and treated animals were preserved in 10% neutral buffered formalin immediately after the autopsy. The tissues were dehydrated by passing through ascending grades of alcohol, cleared in xylene and embedded in paraffin wax (58 to 60°C mp). 5 μm thick sections were cut on a rotary microtome and stained with H & E, dehydrated in alcohol, cleared in xylene, mounted in DPX and examined under a light microscope.
Statistical analysis
The results were expressed as the means ± SEM. The data were statistically analyzed using one way Analysis of Variance (ANOVA) followed by Turkey’s post hoc test in Graph pad prism 5 (graph pad, software, USA). Statistical significance was accepted with p< 0.05. Correlation Coefficient was measured to estimate the strength of linear association between two Variables. Pearson’s correlation analysis was used to find the correlation between vehicle control and BPA toxicant-treated groups.
Results
Absolute and relative weights
Table 2 shows absolute and relative weights of Prostate gland and seminal vesicle in BPA-treated as well as quercetin along with BPA-HD-treated animals.
No significant difference was observed in absolute and relative weights of prostate gland and seminal vesicle in between different control groups of animals (Groups I-III). BPA treatment (Groups IV-VI) caused significant (p<0.05) reduction in absolute and relative weights of prostate gland as compared to vehicle control group of animals (Group II). These effects were dose dependent (r= 0.906, 0.812 respectively). The maximum reduction in absolute and relative weights was 35.26 % and 28.31% respectively.
As compared to vehicle control (Group II), oral administration of BPA (Groups IV-VI) for 45 days caused significant (p<0.05), dose-dependent decrease in absolute (r=0.967) and relative (r=0.765) weights of seminal vesicle by BPA-LD: 9.11%; BPA-MD: 18.87%and BPA-HD: 25.55% respectively.
Co-treatment with different doses of quercetin along with BPA-HD (Groups VII-IX) caused significant, dose-dependent increase in absolute (r=0.978) as well as relative weights of prostate gland (r=0.941) as compared to BPA-HD alone treated animals (Group VI). Table 2 also shows organoprotective index. Maximum protection was observed with 90 mg/kg bw/day dose of quercetin along with BPA-HD. No significant difference was observed between different control groups of animals (Groups I-III).
Co treatment with quercetin along with BPA-HD (Groups VII-IX) caused significant (p<0.05), dose-dependent increase in absolute and relative weights of seminal vesicle as compared to BPA-HD alone treated group (Group VI). These effects were dose-dependent (r= 0.954, 955 respectively). As shown in table 2 protection percentage of quercetin was maximum with 90 mg/kg bw/day along with BPA-HD (65.36%, 71.60 %, respectively) as calculated by organo protective index.
Table 2. Effect of Bisphenol-A treatment ion changes in weight of prostate gland and seminal vesicle of mice and its amelioration by quercetin
|
Sr. No. |
Experimental groups |
Prostate Gland |
Seminal Vesicle |
||
|
Absolute weight |
Relative weight |
Absolute weight |
Relative weight |
||
|
Control groups |
|||||
|
I |
Untreated control |
18.53±0.16 |
46.32±0.40 |
305.14±1.80 |
762.84±4.50 |
|
II |
Vehicle control (0.2 ml olive oil/animal/day) |
18.57±0.13 |
46.48±0.57 |
306.69±1.86 |
767.70±9.50 |
|
III |
Antidote control (0.2 ml quercetin/animal/day) |
18.52±0.11 |
46.36±0.62 |
304.73±1.88 |
761.4.68±4.68 |
|
BPA-treated groups (Percent change in BPA-treated) |
|||||
|
IV |
BPA-Low dose (80 mg/kg bodyweight/day) |
15.47±0.22a (16.70) |
40.43±0.73 a (12.99) |
278.74±1.85 (9.11) |
728.67±8.93 (5.08) |
|
V |
BPA-Medium dose (120 mg/kg bodyweight/day) |
14.17±0.19 a (23.68) |
38.23±0.77 a (17.74) |
248.82±1.47 (18.87) |
672.38±15.36 (12.41) |
|
VI |
BPA-High dose (240 mg/kg bodyweight/day) |
12.02±0.39 a (35.27) |
33.32±0.90 a (28.31) |
228.32±1.14 (25.55) |
635.22±11.70 (17.26) |
|
BPA (HD) + quercetin -treated group (Organoprotective index from BPA-HD) |
|||||
|
VII |
BPA HD + quercetin (30 mg/kg body weight/day) |
12.96±0.34 b (14.32) |
35.22±0.86 b (14.46) |
234.55±1.24 (7.94) |
652.96±9.52 (13.40) |
|
VIII |
BPA HD+ quercetin (60 mg/kg body weight/day) |
15.30±0.23 b (75.17) |
40.64±0.75 b (55.64) |
257.54±1.15 (37.17) |
684.55±14.52 (37.24) |
|
IX |
BPA HD + quercetin (90 mg/kg body weight/day) |
18.55±0.17 b (99.86) |
46.00±0.48 b (96.41) |
279.55±1.62 (65.37) |
730.09±10.33 (71.61) |
Values are mean ± S.E.M., n=10. Significance at the level of; ap<0.05, as compared to vehicle control. bp<0.05, as compared to BPA-HD-treated. Units: Absolute weight- mg; Relative weight- mg/100 gm body weight.
Biochemical analysis
No significant difference was observed between different control groups of animals (Group I-III). Oral administration of BPA (Groups IV-VI) for 45 days caused significant decrease in protein content (LD: 13.94 %, MD: 25.78% and HD: 38.06%) as compared to vehicle control. The effect was dose-dependent (r= 0.931). Concurrent administration of quercetin along with BPA-HD (Groups VII-IX) significantly ameliorated protein content as compared to BPA-HD alone treated group (Group VI). The effect was dose-dependent (r=0.978). Organoprotective index for protein content was 14.47%, 34.10% and 69.25% for Q30, Q60 and Q90 respectively. BPA also caused significant (p<0.05), dose-dependent (r=0.994) increase in acid phosphatase activity by 41.37%, 87.93% and 153.44% for LD , MD and HD of BPA respectively as compared to vehicle control group of animals. Co treatment with different doses of quercetin along with BPA-HD caused significant, dose-dependent (r=-0.989) decrease in acid phosphatase activity as compared to BPA-HD alone treated animals. Organoprotective index for acid phosphatase activity was 22.09%, 47.67% and 88.37% for Q30, Q60 and Q90 along with BPA HD respectively (table 3).
No significant changes were observed in fructose content of all different control groups of animals (Groups I-III). As shown in table 3, BPA treatment (Groups IV-VI) for 45 days caused significant, dose- dependent decrease in fructose (r=0.982) content in seminal vesicle, as compared to vehicle control (Group II). The reduction in fructose content was 28.79%, 37.25% and 49.47% for LD, MD, and LD dose of BPA respectively. However, co administration with different doses of quercetin along with BPA-HD (Group VII-IX) for 45 days caused significant dose-dependent (r=0.891) increase in fructose content as compared to BPA-HD alone treated group (Group VI). The organoprotective index calculated for fructose content was 36.49%, 55.99% and 73.95% for Q30, Q60 and Q90 respectively.
Table 3. Effect of bisphenol-A treatment on biochemical changes in prostate gland and seminal vesicle of mice and its amelioration by quercetin
|
Sr. No. |
Experimental groups |
Prostate Gland |
Seminal Vesicle |
|
|
Protein |
ACP |
Fructose |
||
|
Control groups |
||||
|
I |
Untreated control |
8.83±0.10 |
0.53±0.02 |
46.89±0.81 |
|
II |
Vehicle control (0.2 ml olive oil/animal/day) |
8.72±0.11 |
0.52±0.06 |
46.06±0.36 |
|
III |
Antidote control (0.2 ml quercetin/animal/day) |
8.83±0.76 |
0.52±0.02 |
46.48±0.74 |
|
BPA-treated groups (Percent change in BPA-treated) |
||||
|
IV |
BPA-Low dose (80 mg/kg bodyweight/day) |
7.51±0.06 a (13.94) |
0.86±0.02 a (25.55) |
32.80±0.36 a (28.79) |
|
V |
BPA-Medium dose (120 mg/kg bodyweight/day) |
6.48±0.16 a (25.78) |
1.20±0.07 a (64.70) |
28.90±0.16 a (37.25) |
|
VI |
BPA-High dose (240 mg/kg bodyweight/day) |
5.40±0.12 a (38.06) |
1.43±0.04 a (69.88) |
23.27±0.22 a (49.47) |
|
BPA (HD) + quercetin -treated group (Organoprotective index) |
||||
|
VII |
BPA HD + quercetin (30 mg/kg body weight/day) |
6.11±0.11 b (21.94) |
1.17±0.03 b (76.58) |
31.58±0.58 b (36.49) |
|
VIII |
BPA HD+ quercetin (60 mg/kg body weight/day) |
7.46±0.05 b (61.76) |
0.89±0.04 b (86.45) |
36.03±0.51 b (55.99) |
|
IX |
BPA HD + quercetin (90 mg/kg body weight/day) |
8.52±0.07 b (93.96) |
0.58±0.05 b (99.10) |
40.12±0.67 b (73.95) |
Values are mean ± S.E.M., n=10. Significance at the level of; ap<0.05, as compared to vehicle control. bp<0.05, as compared to BPA-HD-treated. Units: Protein- mg/100 mg tissue weight; ACP- μmoles p-nitrophenol released/mg protein/30 min. Fructose- μg/mg tissue weight.
Table 4. Effect of Bisphenol-A treatment on testosterone level in serum of mice and its amelioration by quercetin
|
Sr. No. |
Experimental groups |
Serum |
|
|
Testosterone |
|||
|
Control groups |
|||
|
I |
Untreated control |
2.728 ± 0.044 |
|
|
II |
Vehicle control (0.2 ml olive oil/animal/day) |
2.730 ± 0.052 |
|
|
III |
Antidote control (0.2 ml quercetin/animal/day) |
2.679 ± 0.053 |
|
|
BPA-treated groups (Percent change in BPA-treated) |
|
||
|
IV |
BPA-Low dose (80 mg/kg bodyweight/day) |
2.243 ± 0.047a (18.09) |
|
|
V |
BPA-Medium dose (120 mg/kg bodyweight/day) |
1.335 ± 0.024 a (51.24) |
|
|
VI |
BPA-High dose (240 mg/kg bodyweight/day) |
0.723 ± 0.067 a (73.61) |
|
|
BPA (HD) + quercetin -treated group (Organoprotective index) |
|||
|
VII |
BPA HD + quercetin (30 mg/kg body weight/day) |
1.498± 0.033b (36.97) |
|
|
VIII |
BPA HD+ quercetin (60 mg/kg body weight/day) |
2.145± 0.089b (69.87) |
|
|
IX |
BPA HD + quercetin (90 mg/kg body weight/day) |
2.694± 0.036b (97.79) |
|
Values are mean ± S.E.M., n=10. Significance at the level of; ap<0.05, as compared to vehicle control. bp<0.05, as compared to BPA-HD-treated. Units: Testosterone: ng/dl.
Histopathological analysis
The transverse section of prostate gland of all control groups (Groups I-III) of animals showed normal smooth muscle layer and prominent luminal folds (Figure 1). Oral administration of BPA (Groups IV- VI) for 45 days caused degeneration in luminal folds and epithelial cells (Figure 2). This effect was more pronounced in BPA-HD groups of animals (Group-VI) than that of BPA-LD treated group of animals (Group IV). Co-treatment with quercetin along with BPA-HD (Groups VII-IX) caused amelioration in BPA-induced changes in histo-architecture of prostate gland (Figure 3).
The seminal vesicle of all control groups (Groups I-III) of animals showed normal mucosal epithelium, muscle layer and highly folded mucosa (figure 4). However, oral administration of BPA (Groups IV-VI) for 45 days caused degeneration in mucosal epithelium and mucosal folds in seminal vesicle (Figure 5). These changes were more pronounced in BPA-HD treated group (Group-VI). Co administration of quercetin along with BPA (Group VII-IX) showed recovery with normal mucosal epithelial cells in seminal vesicle (Figure 6). The recovery in both the organs was almost complete at the dose of 90 mg/kg bw/day of quercetin along with BPA-HD (Group IX).
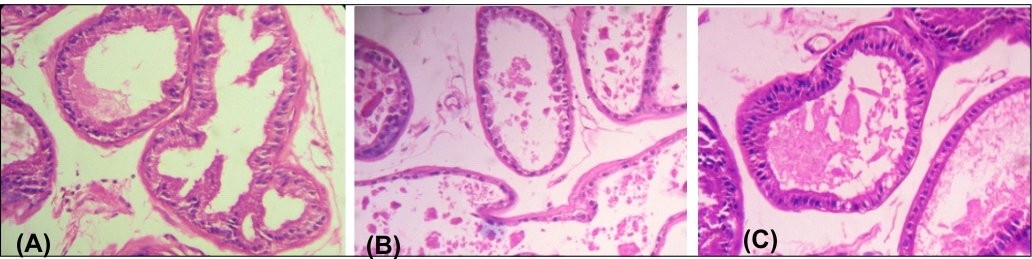
Figure 1. Photomicrograph showed T.S. of prostate gland of: (A) Vehicle control mice showing normal histoarchitecture as mentioned, (B) BPA-HD-treated mice showing highly degenerated epithelial cells and luminal folds, (C) BPA-HD+Q90-treated mice showing almost complete recovery with normal epithelial cells and prominent luminal folds (H & E staining, X225).
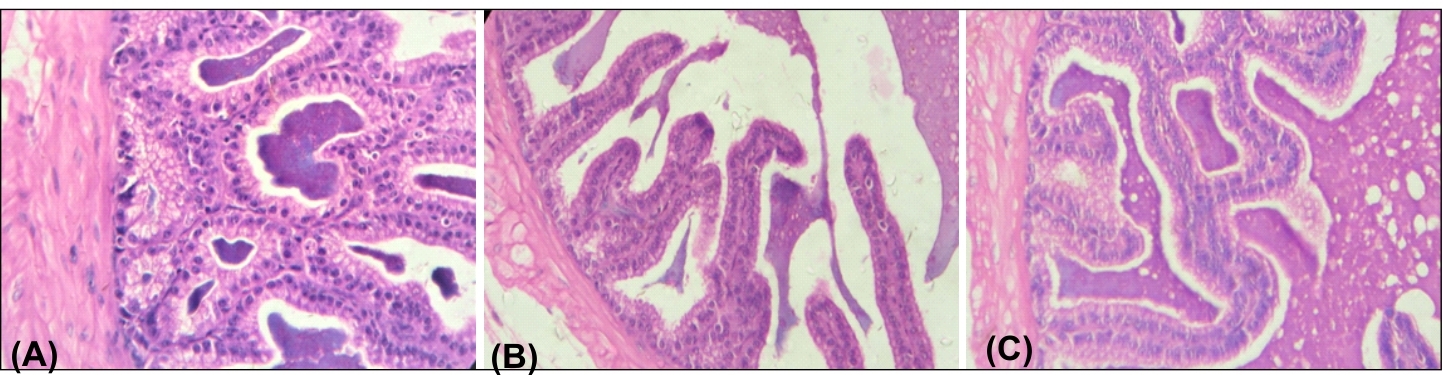
Figure 2. Photomicrograph showed T.S. of seminal vesicle of: (A) Vehicle control mice showing normal histoarchitecture as mentioned, (B) BPA-HD-treated mice showing highly degenerated mucosal epithelium and mucosal folds, (C) BPA-HD+Q50-treated mice showing almost complete recovery with normal mucosal epithelium and highly folded mucosa (H & E staining, X225).
Discussion
Bisphenol A has been shown to have estrogenic activity. (Laws et al., 2000) Testicular toxicity of BPA has been studied by Takahashi and Oishi, (2003). The molecular biology and toxicology of BPA in prostate cells is not well understood. To better understand the role of BPA in reduction in infertility, it is important to determine how chronic exposure to BPA affects prostate gland and seminal vesicle. According to our previous study oral administration of BPA for 45 days causes reduction in sperm motility, viability as well as sperm count (Samova et al., 2016). Biochemical studies have demonstrated that, apart from furnishing an osmotically balanced ambiance for the spermatozoa, the seminal plasma also contains substances such as sugars, enzymes, hormones, inorganic ions and polyamines. The function of these substances in relation to sperm transport and motility is still speculative (Brandes et al., 2012; Dewsbury, 1982).
Oral administration of BPA for 45 days caused lethargy and dullness. In addition, it caused dose-dependent, significant decrease in absolute and relative weights of prostate gland and seminal vesicle (Table 2). Reduction in weights could be due to reduced secretion or significant biochemical changes as observed in the present study (Table 3). Prostate is androgen dependent gland (Bieberich et al., 1996). Reduction in the hormone level leads to reduction in the gland function. In the prostate, the predominant ligand for the AR is DHT, which is reduced from testosterone. Supplementation of quercetin along with BPA significantly increased absolute and relative weights (Cicero et al., 1976; Hermo et al., 1994; Kumar and Majumder, 1995).
In present study we have studied acid phosphatase activity and protein content in prostate gland. Prostatic secretions are generally composed of simple sugars and are often slightly alkaline (Huggins et al., 1942). In prostatic secretions, the protein content is less than 1% and includes proteolytic enzymes, prostatic acid phosphatase, beta-microsemino protein, and prostate-specific antigen. Secretions of the accessory sex organs play an important role in the motility and viability of the spermatozoa in man (Lindholmer, 1974; Eliasson and Lindholmer, 1975).
Bisphenol A treatment for 45 days caused significant decrease in protein content in prostate gland of mice. As previously reported, BPA can cause the generation of free radicals and increased susceptibility to oxidative damage (Hassan et al., 2012). Proteins are major targets of free radicals (Du and Gebicki, 2004). Stadman (2001) reported that oxidative modifications of protein by elevated oxidative free radicals. Acid phosphatase is a marker enzyme for the lysosomal integrity and important for the tissue reorganization and tissue repair (Bullet et al., 2002). Oral administration of BPA for 45 days caused significant dose-dependent increase in ACP activity in prostate gland (Table 2) of mice. This might be due to increased release of lysosomal enzyme by damaging lysosomal integrity causing lysis of cell. Histopathology studies revealed necrosis in prostate gland of BPA treated mice.
BPA treatment significantly decreased fructose content in seminal vesicle (table 3) Fructose provides nutrient energy for the ejaculated spermatozoa. So the reduction in fructose level affects the motility and viability of spermatozoa (Mann, 1948; Cheah et al., 2011). Prostate gland of control animals showed normal smooth muscle layer and prominent luminal folds, which is required for the normal secretion. Acid phosphatase is deposited in lysosomes and functions when these fuse with endosomes, which are acidified while they function; therefore, it has an acid pH optimum (Eskelinen et al., 2003). Degeneration in luminal folds and epithelial cells is seen in BPA treated groups. Degeneration of epithelium will affect the lysosomal integrity, which leads to the increased the acid phosphatase activity (Trelstad et al., 1982). Co treatment with quercetin caused amelioration in BPA-induced changes in histo-architecture of prostate gland.
Previous in vivo studies show that BPA reduces steroidogenesis and spermatogenesis (Samova et al., 2016; Samova et al., 2018). Prostate gland is androgen-dependent tissues. So the biochemical changes observed here could also be due to reduced testosterone level by BPA treatment (Vom Saal et al., 1998). Increased testosterone level by quercetin co-treatment might be the reason for recovery in prostate gland and seminal vesicle. Ejaculate appear to be important in fertility (Cicero et al., 1976; Kidd et al., 2001). Prostate secretion allow sperm to survive more by neutralizing the veginal acidic medium to neutral. According to the study of Martin and Gould the secretions of the accessory sex organs are the main contribution to the volume of ejaculates, (Martin and Gould, 1981) moreover it allow sperm to survive more by neutralizing the vaginal acidic medium (Creasy et al., 2005; Robertson et al., 2007). Removal of the prostate glands and seminal vesicles may greatly alter the concentration of spermatozoa or the volume of ejaculate in mice and thus may account for the lower fertility in the mice (Pang et al., 1979; Queen et al., 1981; Peitz and Olds-Clarke P, 1986: Mann and Lutwak-Mann, 2012).
Oral administration of quercetin to BPA-HD treated groups of mice caused significant, dose dependent amelioration. Absolute and relative weights, histopathology as well as biochemical changes in prostate gland and seminal vesicle of mice were observed. This could be due to antioxidative effect of quercetin on seminal vesicle as shown in present study. This could be due to antioxidative property of quercetin. In addition, it may also due to increased testosterone level in quercetin treated animals as these are androgen dependent organs.
Conclusion
In conclusion, the present investigation has shown that BPA is capable of producing alterations in biochemical parameters investigated in prostate gland. Moreover administration of BPA caused dose-dependent, significant increases in the activity of acid phosphatase. Treatment with quercetin along with BPA caused significant amelioration in all parameters as compared to BPA alone treated groups in prostate gland of mice. The importance of the accessory sex organs on fertility in mice was confirmed in the present study.
Conflicts of interest
The authors declare no conflict of interest.
Acknowledgements
Sanman Samova, and Hetal Doctor acknowledge financial assistance from University Grants Commission (UGC), Govt. of India as Rajiv Gandhi National Fellowship. The authors are sincerely thankful to the Department of Zoology, B.M.T.C. and Human Genetic, Gujarat University for providing an opportunity to access the bioinformatics research facilities.
References
Afanas' ev IB, Dcrozhko AI, Brodskii AV, Kostyuk VA, Potapovitch AI. 1989. Chelating and free radical scavenging mechanisms of inhibitory action of rutin and quercetin in lipid peroxidation. Biochemical pharmacology, 38(11):1763-9.
Akingbemi BT, Soitas CM, Koulova AI, Kleinfelter GR, Hardy MP. 2004. Inhibition of testicular steroidogenesis by the xenoestrogenbisphenol A is associated with reduced pituitary luteinizing hormone secretion and decreased steroidogenic enzyme gene expression in rat Leyding cells. Endocrinology, 145:592-603.
Bellik Y, Boukraâ L, Alzahrani H, Bakhotmah B, Abdellah F, Hammoudi S, Iguer-Ouada M. 2013. Molecular mechanism underlying anti-inflammatory and anti-allergic activities of phytochemicals: an update. Molecules, 18(1):322-53.
Bieberich CJ, Fujita K, He WW, Jay G. 1996. Prostate-specific and androgen-dependent expression of a novel homeobox gene. Journal of Biological Chemistry, 271(50):31779-82.
Bindhumol V, Chitra KC, Mathur PP. 2003. Bisphenol A induces reactive oxygen species generation in the liver of male rats. Toxicology, 188(2-3):117-24.
Brandes D. 2012. Male Accessory Sex Organs: structure and function in mammals. Elsevier; 2012 Dec 2.
Bull H, Murray PG, Thomas D, Fraser AM, Nelson PN. 2002. Acid phosphatases. Molecular Pathology, 55(2):65.
Chapin RE, Adams J, Boekelheide K, Gray Jr LE, Hayward SW, Lees PS, McIntyre BS, Portier KM, Schnorr TM, Selevan SG, Vandenbergh JG. 2008. NTP‐CERHR expert panel report on the reproductive and developmental toxicity of bisphenol A. Birth Defects Research Part B: Developmental and Reproductive Toxicology, 83(3):157-395.
Cheah Y, Yang W. 2011. Functions of essential nutrition for high quality spermatogenesis. Advances in Bioscience and Biotechnology, 2(04):182.
Cicero Tj, Meyer Er, Bell Rd, Koch Ga. 1976. Effects of morphine and methadone on serum testosterone and luteinizing hormone levels and on the secondary sex organs of the male rat. Endocrinology, 98(2):367-72.
Cornish KM, Williamson G, Sanderson J. 2002. Quercetin metabolism in the lens: role in inhibition of hydrogen peroxide induced cataract. Free Radical Biology and Medicine, 33(1):63-70.
Creasy DM. The male reproductive system. In Target organ pathology: a basic text 2005 (pp. 334-365). Taylor & Francis London, Bristol.
Dewsbury DA. 1982. Ejaculate cost and male choice. The American Naturalist, 119(5):601-10.
Dodds EC, Lawson W. 1936 Synthetic estrogenic agents without the phenanthrene nucleus Nature 137:996.
Du J, Gebicki JM. 2004. Proteins are major initial cell targets of hydroxyl free radicals. The international journal of biochemistry & cell biology, 36(11):2334-43.
Eliasson R, Lindholmer C. 1975. Human seminal plasma and sperm survival and transport. Inthe Biology of spermatozoa (pp. 83-92). Karger Publishers.
Eskelinen EL, Tanaka Y, Saftig P. 2003. At the acidic edge: emerging functions for lysosomal membrane proteins. Trends in cell biology, 13(3):137-45.
Foreman D, Gaylor L, Evans E, Trella C. 1973. A modification of the Roe procedure for determination of fructose in tissues with increased specificity. Analytical biochemistry, 56(2):584-90.
Grassi D, Desideri G, Ferri L, Aggio A, Tiberti S, Ferri C. 2010. Oxidative stress and endothelial dysfunction: say NO to cigarette smoking!. Current pharmaceutical design, 16(23):2539-50.
Grumetto L, Montesano D, Seccia S, Albrizio S, Barbato F. 2008. Determination of bisphenol A and bisphenol B residues in canned peeled tomatoes by reversed-phase liquid chromatography. Journal of agricultural and food chemistry, 56(22):10633-7.
Guardia T, Rotelli AE, Juarez AO, Pelzer LE. 2001. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Il farmaco, 56(9):683-7.
Hassan ZK, Elobeid MA, Virk P, Omer SA, ElAmin M, Daghestani MH, AlOlayan EM. 2012. Bisphenol A induces hepatotoxicity through oxidative stress in rat model. Oxidative Medicine and Cellular Longevity. 24;2012.
Hermo L, Oka R, Morales CR. 1994. Secretion and endocytosis in the male reproductive tract: a role in sperm maturation. In International review of cytology Vol. 154:105-189
Huggins C, Scott WW, Heinen JH. 1942. Chemical composition of human semen and of the secretions of the prostate and seminal vesicles. American Journal of Physiology-Legacy Content, 136(3):467-73.
Kabuto H, Hasuike S, Minagawa N, Shishibori T. 2003. Effects of bisphenol A on the metabolisms of active oxygen species in mouse tissues. Environmental research, 93(1):31-5.
Karim Z, Husain Q. 2010. Application of fly ash adsorbed peroxidase for the removal of bisphenol A in batch process and continuous reactor: Assessment of genotoxicity of its product. Food and Chemical Toxicology, 48(12):3385-90.
Kidd SA, Eskenazi B, Wyrobek AJ. 2001. Effects of male age on semen quality and fertility: a review of the literature. Fertility and sterility, 75(2):237-48.
Kumar VL, Majumder PK. 1995. Prostate gland: structure, functions and regulation. International urology and nephrology, 27(3):231-43.
Kuruto-Niwa R, Tateoka Y, Usuki Y, Nozawa R. 2007. Measurement of bisphenol A concentrations in human colostrum. Chemosphere. 66(6):1160-4.
Laws SC, Carey SA, Ferrell JM, Bodman GJ, Cooper RL. 2000. Estrogenic activity of octylphenol, nonylphenol, bisphenol A and methoxychlor in rats. Toxicological Sciences, 54(1):154-67.
Levine RL, Stadtman ER. 2001. Oxidative modification of proteins during aging. Experimental gerontology, 36(9):1495-502.
Lindholmer CH. 1974. The importance of seminal plasma for human sperm motility. Biology of reproduction, 10(5):533-42.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. 1951. Protein measurment with the Folin phenol reagent. Journal of Biological Chemistry, 4:492-501.
Mann T, Lutwak-Mann C. 2012. Male reproductive function and semen: themes and trends in physiology, biochemistry and investigative andrology. Springer Science & Business Media; 2012
Mann T. 1948. Fructose content and fructolysis in semen. Practical application in the evaluation of semen quality. The Journal of Agricultural Science 38(3):323-31.
Martin DE, Gould KG. 1981. The male ape genital tract and its secretions. Reproductive biology of the great apes: comparative and biomedical perspectives. Published by Elsevier BV, p.p.127-61.
MSDS 2004. Bisphenol A Material safety Data Sheet. In http://terpconnect.umd.edu/-choi/MSDS/Sigma. Aldrich/ BISPHENOL A %20A.Pdf (SIGMA-ALDRICH).
Pang SF, Chow PH, Wong TM. 1979. The role of the seminal vesicles, coagulating glands and prostate glands on the fertility and fecundity of mice. Journal of Reproduction and Fertility, 56(1):129-32.
Peitz B, Olds-Clarke P. 1986. Effects of seminal vesicle removal on fertility and uterine sperm motility in the house mouse. Biology of Reproduction, 35(3):608-17.
Prins GS, Birch L, Tang WY, Ho SM. 2007. Developmental estrogen exposures predispose to prostate carcinogenesis with aging. Reproductive Toxicology, 23(3):374-82.
Pupo M, Pisano A, Lappano R, Santolla MF, De Francesco EM, Abonante S, Rosano C, Maggiolini M. 2012. Bisphenol A induces gene expression changes and proliferative effects through GPER in breast cancer cells and cancer-associated fibroblasts. Environmental Health Perspectives, 120(8):1177.
Queen K, Dhabuwala CB, Pierrepoint CG. 1981. The effect of the removal of the various accessory sex glands on the fertility of male rats. Journal of Reproduction and Fertility, 62(2):423-6.
Ranjit N, Siefert K, Padmanabhan V. 2010. Bisphenol-A and disparities in birth outcomes: a review and directions for future research. Journal of Perinatology, 30(1):2.
Robak J, Gryglewski RJ. 1988. Flavonoids are scavengers of superoxide anions. Biochemical pharmacology, 37(5):837-41.
Robertson SA, Sharkey DJ, Bromfield JJ. 2007. Seminal Plasma and Male Factor Signalling in the Female Reproductive Tract: Is 19. American Journal of Reproductive Immunology, 58(3):188-9.
Rochester JR. 2013. Bisphenol A and human health: a review of the literature. Reproductive toxicology, 42:132-55.
Sak K. 2014. Site-specific anticancer effects of dietary flavonoid quercetin. Nutrition and cancer, 66(2):177-93.
Samova S, Doctor H, Verma RJ. 2016. Spermatotoxic effect of bisphenol A and its amelioration using quercetin. . world journal of pharmacy and pharmaceutical sciences, 5(5):1161-1175
Samova S, Patel CN, Doctor H, Pandya HA, Verma RJ. 2018. The effect of bisphenol A on testicular steroidogenesis and its amelioration by quercetin: an in vivo and in silico approach. Toxicology Research, 7(1):22-31.
Sangai NP, Verma RJ. 2012. Quercetin ameliorates bisphenol A-induced toxicity in mice. Acta Poloniae Pharmaceutical n Drug Research, 69(3):557-63.
Sangai NP, Verma RJ.2012. Quercetin ameliorates bisphenol A-induced toxicity in mice. Acta Poloniae Pharmaceutical n Drug Research, 69(3):557-63.
Sigma Technical Bulletin No. 104, Sigma Chemical Co., 3500, Dekoib St. Louis 18, MO, USA.
Stadtman ER. 2001. Protein oxidation in aging and age‐related diseases. Annals of the new York Academy of Sciences, 928(1):22-38.
Suzuki Y, Ishihara M, Segami T, Ito M. 1998. Anti-ulcer effects of antioxidants, quercetin, α-tocopherol, nifedipine and tetracycline in rats. The Japanese Journal of Pharmacology, 78(4):435-41.
Takahashi O, Oishi S. 2003. Testicular toxicity of dietarily or parenterally administered bisphenol A in rats and mice. Food and chemical toxicology. 41(7):1035-44.
Takayanagi S, Tokunaga T, Liu X, Okada H, Matsushima A, Shimohigashi Y. 2006. Endocrine disruptor bisphenol A strongly binds to human estrogen-related receptor γ (ERRγ) with high constitutive activity. Toxicology letters, 1;167(2):95-105.
Trelstad RL, Hayashi A, Hayashi K, Donahoe PK. 1982. The epithelial-mesenchymal interface of the male rat Mullerian duct: loss of basement membrane integrity and ductal regression. Developmental biology, 92(1):27-40.
Vom Saal FS, Akingbemi BT, Belcher SM, Birnbaum LS, Crain DA, Eriksen M, Farabollini F, Guillette Jr LJ, Hauser R, Heindel JJ, Ho SM. Chapel Hill. 2007. Bisphenol A expert panel consensus statement: integration of mechanisms, effects in animals and potential to impact human health at current levels of exposure. Reproductive toxicology (Elmsford, NY), 24(2):131.
Vom Saal FS, Cooke PS, Buchanan DL, Palanza P, Thayer KA, Nagel SC, Parmigiani S, Welshons WV. 1998. A physiologically based approach to the study of bisphenol A and other estrogenic chemicals on the size of reproductive organs, daily sperm production, and behavior. Toxicology and Industrial Health, 14(1-2):239-60.
Wach A, Pyrzyńska K, Biesaga M. 2007. Quercetin content in some food and herbal samples. Food Chemistry, 100(2):699-704.