

Mithun Singh Rajput*, Devashish Rathore, Rashmi Dahima
School of Pharmacy, Devi Ahilya Vishwavidyalaya, Takshashila Campus, Khandwa Road, Indore-452001, M.P., India
*Address for Correspondence
Dr. Mithun Singh Rajput,
Postdoctoral Fellow, School of Pharmacy, Devi Ahilya Vishwavidyalaya, Takshashila Campus, Khandwa Road, Indore-452001, M.P., India.
Abstract
Background: Neutrophils are one of the components of innate immune system that defend against infectious microorganisms through various antimicrobial capabilities, including microorganism phagocytosis and by releasing bactericidal agents from intracellular secretory granules into the phagosome or to the cell exterior. Activated neutrophils represent the main source of myeloperoxidase (MPO), superoxide (SO) and subsequently derived oxygen metabolites. Objective: The current study was carried out with the aim to investigate the effect of cardamonin on N-formyl-methionyl-leucyl-phenyl-alanine (fMLP) and opsonized zymosan (OZ) stimulated SO generation in neutrophils and their degranulation measured as MPO release. Materials and Methods: Spectrophotometry was used to evaluate the effect of cardamonin (0.1 to 100.0μmol/l) on OZ (0.5 mg/ml) or fMLP (0.1μmol/l) stimulated SO generation and MPO release in neutrophil cells. Superoxide formation was measured in isolated neutrophils as superoxide dismutase inhibitable reduction of cytochrome c and the activity of MPO was assayed by determining the oxidation of o –dianisidine in the presence of hydrogen peroxide. Results: It is evident from one way ANCOVA followed by post-hoc test that cardamonin dose-dependently (1.0 to 100.0μmol/l) significantly decreased (p < 0.05) SO generation and MPO release after each stimulus (fMLP or OZ). Conclusion: Our results indicate that cardamonin could support resolution of inflammation through decreased activity of neutrophils, i.e. respiratory burst and degranulation by inhibiting MPO release and by decreasing the generation of SO and the subsequently derived reactive oxygen species.
Keywords: Cardamonin, Myeloperoxidase, Neutrophils, PMA, Superoxide generation
Introduction
Neutrophils are one of the components of innate immune system that defend against infectious microorganisms through various antimicrobial capabilities, including microorganism phagocytosis and by releasing bactericidal agents from intracellular secretory granules into the phagosome or to the cell exterior (Mantovani et al., 2011). Neutrophils produce microbicidal oxidants to destroy the invading pathogens using nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, a membrane-associated enzyme complex that generates superoxide anion (O2−) (Lominadze et al., 2005). Activated neutrophils represent the main source of myeloperoxidase (MPO), superoxide (SO) and subsequently derived oxygen metabolites. MPO, the most abundant enzyme in neutrophils, is a major nitric oxide (NO) scavenger and a marker of oxidative stress as well as a powerful auto and paracrine stimulator of neutrophil activation (Pecivova et al., 2012) Superoxide, NO and MPO seem to be coupled through complex and interdependent pathways. The biological consequences of NO-peroxidase interactions may have broad implications for the regulation of local infectious, inflammatory and cardiovascular events in vivo (Eiserich et al., 1998). Overproduction of reactive oxygen species (ROS), prolonged or excessive liberation of MPO and other effective yet also toxic substances from neutrophils may participate in disturbed apoptosis, intensify the inflammatory processes and result in serious human diseases (Abu-Soud et al., 2000).
Cardamonin, chemically known as (2’, 4’-dihydroxy-6’-methoxychalcone) is a naturally occurring chalcone, foremost isolated from seeds of Amomum subulatum and later from other plant species, such as Alpinia henryi, Alpinia katsumadai, Alpinia rafflesiana, Boesenbergia pandurata and Campomanesia adamantium (Bheemasankara et al., 1976). Cardamonin is known to exhibit many pharmacological activities including anti-inflammatory, anti-mutagenic, anti-neoplastic, anti-proliferative, anti-tumor, in vitro anti-HIV, vasorelaxant, hypoglycemic, anti-infectious and anti-platelet activities (Ping et al., 2018). It is also known to possess potential antioxidant effect. Treatment of induced RAW 264.7 cells with cardamonin showed dose-dependent inhibition of intracellular ROS generation (Ahmad et al., 2006). The quest for potential health applications of cardamonin particularly in relation to its antioxidant property seems to grow in the near future, as ROS generation is one of the root causes of several health ailments. The current study was carried out with the aim to investigate the effect of cardamonin on N-formyl-methionyl-leucyl-phenyl-alanine (fMLP), and opsonized zymosan (OZ) stimulated SO generation in neutrophils and their degranulation measured as MPO release.
Materials and Methods
Drugs and solutions
Cardamonin or 2′, 4′-dihydroxy-6′-methoxychalcone with the purity of ≥98%, PMA, fMLP, OZ and cytochalasin B were purchased from Sigma Aldrich (Sigma-Aldrich Chemicals Pvt. Ltd. Bengaluru, India). Cardamonin was freshly prepared by dissolving in DMSO, tween 20 and distilled water at a ratio of 5:5:90 (v/v) (Rajput et al., 2019). All other drugs were dissolved in distilled water. The doses for all freshly prepared drug solutions were expressed in terms of their free bases.
Isolation of neutrophils
Fresh goat blood was obtained from the authorized slaughterhouse. Blood was taken into 3.8% trisodium citrate and erythrocytes were removed by dextran sedimentation and centrifugation by the modified method described by Kuhns et al. (2015).
Superoxide determination
Superoxide formation was measured in isolated neutrophils as superoxide dismutase inhibitable reduction of cytochrome c. Neutrophil suspension (1 × 106 neutrophils/1.5 ml PBS with 0.9 mmol/l CaCl2 and 0.5 mmol/l MgCl2) was pre-incubated for 5 min at 37 °C with cardamonin (0.1–100.0 μmol/l) and subsequently stimulated by addition of fMLP (0.1 μmol/l) for 3 min at 37 °C, or of 0.5 mg/l OZ for 60 min at 37 °C. Controls were included for the effect of each stimulus and of cardamonin on cytochrome c reduction. After centrifugation at 4200 g for 2 min at 4 °C, absorbance was measured at 550 nm in a spectrophotometer. The superoxide production was calculated from the rate of change of absorbance using the extinction coefficient 2.1 × 104 mol–1cm–1 (Pecivova et al., 2007).
Myeloperoxidase release
For determination of MPO release, neutrophils were pre-incubated with cytochalasin B (5 μg/ml) for 5 min at room temperature. Neutrophils (2 × 106/sample) were pre-incubated with cardamonin (0.1–100.0 μmol/l) in a shaker bath at 37 °C for 5 min, followed by 3 min exposure to fMLP (0.1 μmol/l) or 60 min to OZ (0.5 mg/l). The activity of MPO was assayed in the supernatant after centrifugation at 983 g for 10 min at 4 °C by determining the oxidation of o –dianisidine in the presence of hydrogen peroxide in a spectrophotometer at 463 nm (Somersalo et al., 1990).
Statistical analysis
Results were expressed as mean ± S.E.M. The data were analyzed by one way analysis of co-variance (ANCOVA) tracked by Tukey's multiple comparison tests. Probability values less than 0.05 were considered statistically significant in all the cases.
Results
Soluble fMLP and particulate OZ stimuli, operating through different membrane receptors were used to induce respiratory burst, measured as SO generation and MPO release as a marker of degranulation. Cardamonin (0.1–100 μmol/l) had no significant effect on SO generation and MPO release from unstimulated isolated neutrophils.
It is evident from one way ANCOVA followed by post-hoc test that cardamonin dose-dependently decreased SO generation after each stimulus (fMLP or OZ), however a significant (p < 0.001 for all doses) decrease was recorded only with cardamonin concentration of 1.0 to 100.0 μmol/l [for fMLP, F(4, 25) = 56.35, p < 0.0001, R2 = 0.9002] [for OZ, F(4, 25) = 51.83, p < 0.0001, R2 = 0.8924] (Figure 1).
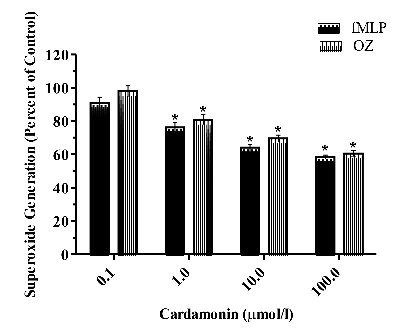
Figure 1. Effect of cardamonin on fMLP and OZ stimulated superoxide generation in neutrophils. Results are expressed as Mean ± S.E.M.; n = 6 in each group. Data was analyzed by one way repeat measure ANCOVA followed by Tuckey’s multiple comparison test. Significance: *p< 0.001 when compared with control group, fMLP: N-formyl-methionyl-leucyl-phenyl-alanine; OZ: opsonized zymosan.
One way ANCOVA followed by post-hoc test shows the dose-dependent decreasing effect of cardamonin on stimulated MPO release from intact cells. Cardamonin (1.0 to 100.0 μmol/l) significantly (p < 0.05, 0.001 and 0.001 respectively) decreased MPO release after both fMLP and OZ stimulation [for fMLP, F(4, 25) = 20.44, p < 0.0001, R2 = 0.7658] [for OZ, F(4, 25) = 42.69, p < 0.0001, R2 = 0.8723 (Figure 2).
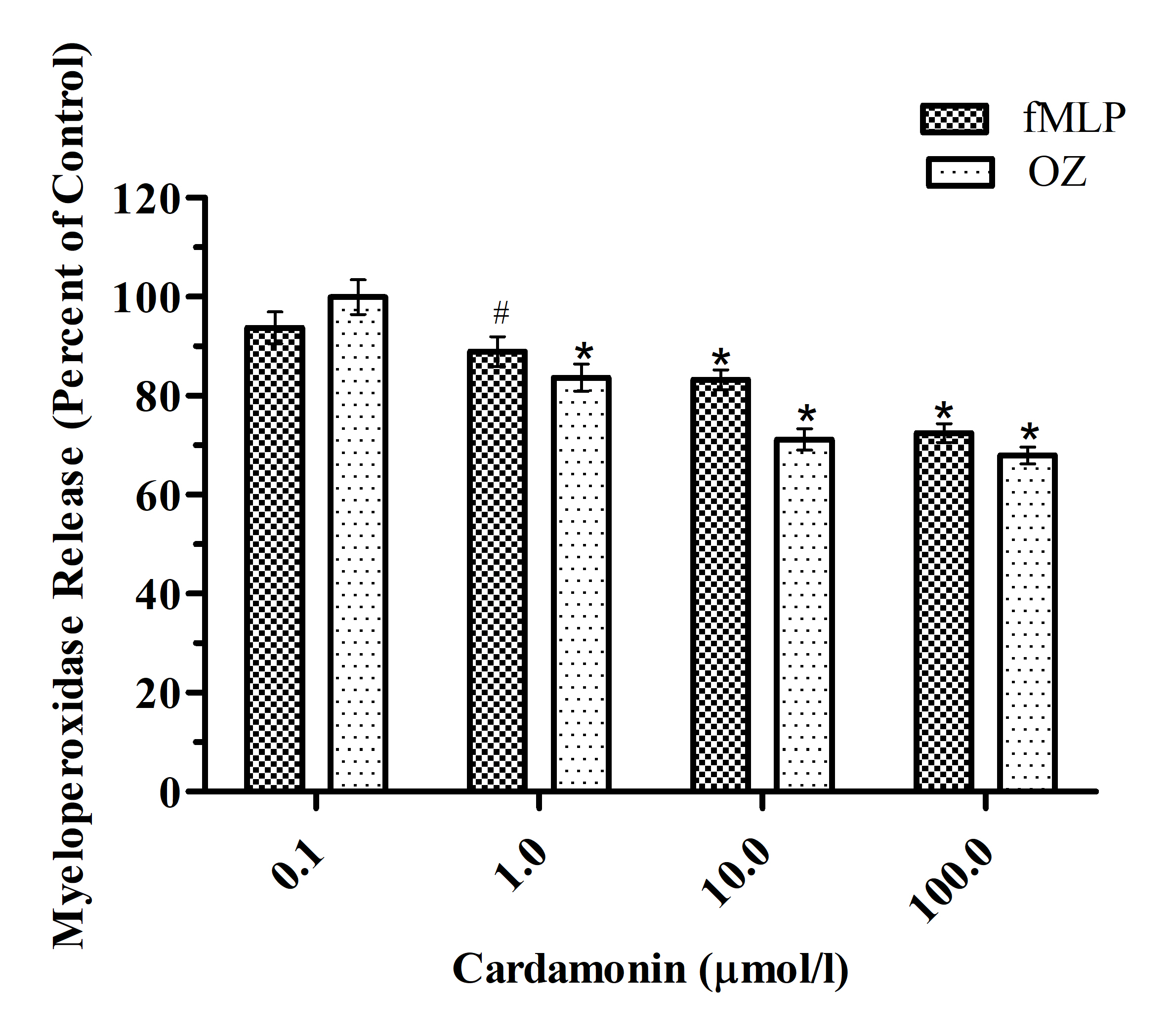
Figure 2. Effect of cardamonin on fMLP and OZ stimulated in myeloperoxidase release in neutrophils. Results are expressed as Mean ± S.E.M.; n = 6 in each group. Data was analyzed by one way repeat measure ANCOVA followed by Tuckey’s multiple comparison test. Significance: #p< 0.05 and *p< 0.001 when compared with control group, fMLP: N-formyl-methionyl-leucyl-phenyl-alanine; OZ: opsonized zymosan.
Discussion
In present study, to demonstrate the effect of cardamonin on neutrophils in vitro, we used two receptor-operating stimuli (fMLP and OZ) to evoke a respiratory burst. In our experiments, the incubation of isolated neutrophils with cardamonin (100μmol/l) diminished their fMLP and OZ activated production of SO by 58% and 60% respectively in comparison with control. Similarly, cardamonin (100μmol/l) diminished their fMLP and OZ activated release of MPO by 72% and 67% respectively in comparison with control.
MPO is the most abundant enzyme in neutrophils and has been implicated directly or through its metabolites, hypochlorous acid and subsequently derived ROS in many life-endangering diseases (Arnhold, 2004). Our results suggest that through interference with the phospholipase signaling pathway, cardamonin decreases SO generation and MPO release from neutrophils with subsequent decrease of SO and MPO derived potentially harmful metabolites. Concentrations effective in our experiments were about 100 times higher than plasma levels found in patients (Luis et al., 1987). During long term treatment, however, the cellular concentration of lipophilic drugs may increase several times. Therefore, we assume that inhibition of MPO release could participate on beneficial effects of cardamonin. Since the release of MPO (an important auto- and paracrine modulator of neutrophil functions) is reduced, a decrease of both neutrophil infiltration and mutual platelet-neutrophil interactions induced by carvedilol may also be assumed.
Our study provided evidence supporting the potential beneficial effect of cardamonin in diminishing tissue damage at the site of inflammation by inhibiting MPO release and by decreasing the generation of SO and the subsequently derived ROS.
Conflict of Interest
We declare that we do not have any conflict of interest.
References
Abu-Soud H, Hazen S. 2000. Nitric oxide is a physiological substrate for mammalian peroxidases. Journal of Biological Chemistry 275(48): 37524–37532.
Ahmad S, Israf DA, Lajis NH, Shaari K, Mohamed H, Wahab AA, Ariffin KT, Hoo WY, Aziz NA, Kadir AA, Sulaiman MR, Somchit MN. 2006. Cardamonin inhibits pro-inflammatory mediators in activated RAW 264.7 cells and whole blood. European Journal of Pharmacology 538(1-3):188–194.
Arnhold J. 2004. Properties, functions, and secretion of human myeloperoxidase. Biochemistry (Mosc) 69(1): 4–9.
Bheemasankara RC, Namosiva RT, Suryaprakasam, S. 1976. Cardamonin and alpinetin from the seeds of Amomum subulatum. Planta Medica 29(1): 391–392.
Eiserich JP, Hristova M, Cross CE, Jones AD, Freeman BA, Halliwell B, van der Vliet. 1998. Formation of nitric oxide-derived inflammatory oxidants by myeloperoxidase in neutrophils. Nature 391(6665): 393–397.
Kuhns DB, Long PDA, Chu J, Zarember KA. 2015. Isolation and functional analysis of human neutrophils. Current Protocols in Immunology 111(7): 23.
Lominadze G, Powell DW, Luerman GC, Link AJ, Ward RA, McLeish KR. 2005. Proteomic analysis of human neutrophil granules. Molecular and Cellular Proteomics 4(10): 1503–1521.
Luis WJ, McNeil JJ, Workman BS, Drummer OH, Conway EL. 1987. A pharmacokinetic study of carvedilol (BM 14.190) in elderly subjects: preliminary report. Journal of Cardiovascular Pharmacology 10(suppl 11): S89–S93.
Mantovani A, Cassatella MA, Costantini C, Jaillon S. 2011. Neutrophils in the activation and regulation of innate and adaptive immunity. Nature Reviews Immunology 11(8): 519-531.
Pecivova J, Macickova T, Lojek A, Gallova L, Ciz M, Nosal R, Holomanova D. 2007. In vitro effect of carvedilol on professional phagocytes. Pharmacology 79(2): 86-92.
Pecivova J, Macickova T, Svitekova K, Nosal R. 2012. Quercetin inhibits degranulation and superoxide generation in PMA stimulated neutrophils. Interdisciplinary Toxicology 5(2): 81-83.
Ping CP, Mohamad TAST, Akhtar MN, Perimal EK, Akira A, Ali DAI, Sulaiman MR. 2018. Antinociceptive effects of cardamonin in mice: possible involvement of TRPV1, glutamate and opioid receptors. Molecules 23(1): 2237-2239.
Rajput MS, Rathore D, Dahima R. 2019. Anxiolytic potency of cardamonin mediated through brain GABAergic system. Journal of Drug Delivery and Therapeutics 9(1): 248-251.
Somersalo K, Salo OP, Bjorksten F, Mustakalio KK. 1990. A simplified Boyden chamber assay for neutrophil chemotaxis based on quantitation of myeloperoxidase. Anals of Biochemistry 185(2): 238–242.