

Vineeta Shyam1, Kapil Khatri1, Satish Shilpi1*
1*Pharmaceutical research laboratory, Department of Pharmaceutics,
Ravishankar College of Pharmacy, Bypass Road, Bhanpur Square, Bhopal (MP) India
*Corresponding Author
Dr. Satish Shilpi
Pharmaceutical Research Laboratory
Department of Pharmaceutics,
Ravishankar College of Pharmacy,
Bypass Road, Bhanpur Square, Bhopal (MP), India
Abstract
Objective: Salbutamol is a medication that opens up the medium and large airways in the lungs and it is used to treat asthma, exercise-induced bronchospasm, and chronic obstructive pulmonary disease (COPD). The objective of this dissertation work was to prepare salbutamol loaded microspheres using carbopol, Materials and methods: HPMC and sodium CMC for the purpose of effective management of asthma by delivering drug for long period of time in a controlled manner by making them mucoadhesive. Microspheres prepare by emulsification‐heat stabilizing method and characterize on the basis of size, shape and surface morphology, drug content and drug release. Results: Microspheres were spherical shape and smooth in surface confirmed by scanning electon microscopy (SEM). The average size was found to be 12.14±2.53 µm and drug content was 80.15±1.02%. An in-vitro drug release study was revealed that microsphere release 52.16±1.76% drug in 24 hr. Conclusion: It is concluded that prepared microspheres of salbutamol can be prepared by emulsification heat stabilizing method for high drug encapsulation and controlled drug release.
Keywords: Salbutamol, Microspheres, HPMC, Sodium CMC, Drug release
Introduction
Controlled release technology as rapidly emerged over the past three decades, as a new inter disciplinary science that offers novel approaches to the bioactive agents. Controlled drug delivery design involves the application of physical and polymer chemistry to dosage form design, to produces a well characterized and reproducible drug delivery profile (Celebi et al., 1996; Amperiadou et al., 1995; Cook et al., 2005). If drug targeting and retention time is increase in the target environment then controlled release delivery systems can achieve optimum therapeutic responses, prolong efficacy and decreased toxicity1 (Cevik et al., 2000; Fu et al., 2002; Desai and Park, 2005; Young et al., 2006). It is possible, if drug delivery system provide a therapeutic amount of drug to the target sight in the body. The drug delivery system should deliver drug at a rate detected by the needs of the body over the entire period of treatment (Khar et al., 2009; Chourasia et al., 2004).
Asthma is a common long term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and bronchospasm. Asthma is thought to be caused by a combination of genetic and environmental factors (Murthy and Hiremath, 2001; Martinez et al., 2007). Environmental factors include exposure to air pollution and allergens. There is no cure for asthma but symptoms may be controlled with effective asthma treatment and management. Symptoms can be also be prevented by avoiding allergens and irritants or by use of inhaled corticosteroids. Long-acting beta agonists (LABA) or antileukotriene agents may be used in addition to inhaled corticosteroids if asthma symptoms remain uncontrolled. Treatment of rapidly worsening symptoms is usually with an inhaled short-acting beta-2 agonist such as salbutamol and corticosteroids taken by mouth (Martinez et al., 2007).
Salbutamol is adrenergic bronchodilators. It is generally administered by breath through the mouth to open up the air passages in the lungs. It is used to treat or prevent bronchospasm in patients with asthma, bronchitis, emphysema, and other lung associate complication. This medicine is also used to prevent exercise-induced bronchospasm (wheezing). But its high dosing frequency and side effect make it a suitable candidate to formulate in the form sustained release drug delivery system (Pachuau et al., 2008; Prabakaran et al., 2004).
In this project, salbutamol laded polymeric microspheres is prepared which will administered by nasal route to lung. It mocoadhesiveness will help it to adhere with mucus layer of lungs tissue and release drug (salbutamol) in a sustained and controlled manner which will effectively manage the asthma for longer period of time. The mucoadheshivness of microsphere will further increase it retention and making it available for drug release for longer period at the site of action.
Materials and methods
Materials
Salbutamol was obtained as gift sample from Cadila Pharmaceutical Laboratory Pvt Ltd, Ahmedabad, Gujrat, Indai. HPMC, Sodium carboxymethyl cellulose; tween‐80 and span‐80, cellophane dialysis tube MWCO 12000 were also purchased from Himedia Laboratories Pvt. Ltd, India. All other solvents used in the study were of HPLC grade and triple distilled water was used wherever required.
Preaparation of salbutamol loaded microsphere
Microspheres of Salbutamol was prepared by emulsification solvent evaporation method (Chourasia et al., 2004; Amperiadou et al., 1995) with slide modification, in which carbopol, HPMC and sodium CMC were taken in different ration (2:1:1, 4:1:1, 6:1:1) and dissolved in 20 ml of deionised water with continuously stirring on mechanical stirrer. In this solution 0.1% w/v of Tween‐80 was added and stirred continuously for 30 min. Drug (10% w/w of polymer) was added in polymer solution In another container 50mL of castor oil was taken with 1% w/v of span‐80 and stirred it for 20 min. on mechanical stirrer. In this oil phase polymer solution was added drop wise with continuously stirring at 3000 rpm to form w/o emulsion. After 15 min 0.75mL of GST solution was added as cross linking agent and stirred the emulsion for 4 hr to evaporate water from internal phase and got suspension of polymeric microsphere. The suspension containing the micro‐spheres was centrifuged for 5 min at 4000 rpm and the settled microspheres were washed three times with petroleum ether to remove oil from microspheres surfaces. Microspheres were again washed two times with acetone and then pour in Petridis to dry microsphere upon evaporating acetone. Dried microsphere stored in closed vessel until further use.
Characterization of microspheres
Determination of % yield of microspheres
Percent yield of prepared microsphere was determined by taking dried microspheres and weighed accurately. The percentage yield was then calculated using formula given below.
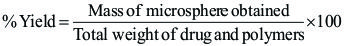
Shape and Surface Morphology
The shape and surface morphology of prepared microsphere were visualized by scanning electron microscopy at AIIMS, New Delhi. Transmission electron microscopy (TEM, Morgagni 268 electron microscope) was used for visualizing particulate shape. Scanning electron microcopy (SEM) (Leo 435 VP, Cambridge, U.K., electron microscope) was also used to characterize the surface morphology of the microsphere. The dried microsphere on a double adhesive tape which was stuck to an aluminum stub and then coated with gold. The sample was then randomly scanned and photographs were taken (Figure 1a & b).
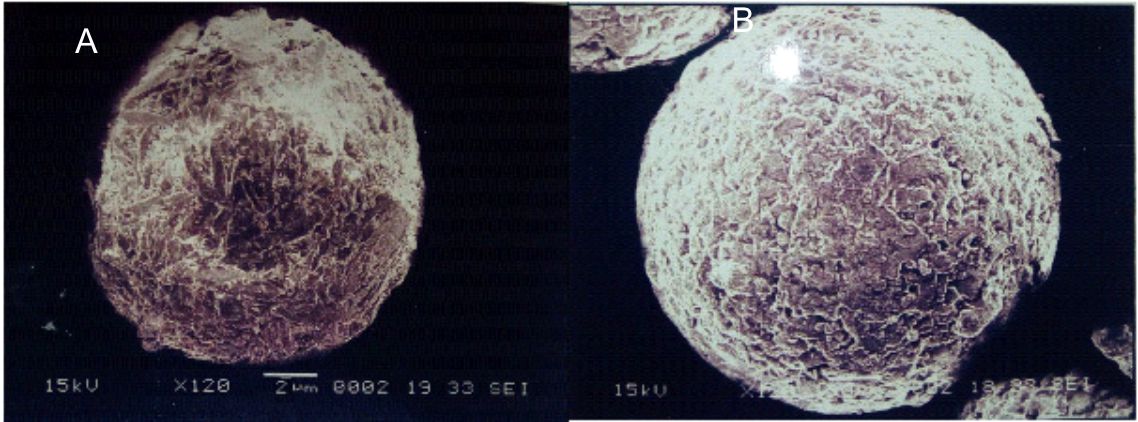
Figure 1. SEM photomicrograph of microsphere (a) without drug loaded (b) drug loaded
Particle Size and Zeta Potential
The size and size distribution analysis of Microsphere was done using Dynamic Light Scattering (DLS) technique using a computerized Malvern zetasizer (DTS ver. 4.10, Malvern instrument, England). Particulate suspension was mixed with the appropriate medium (1:100 dilution using double-distilled water) for particle size measurement. Zeta potential of microsphere dispersion was measured by Malvern zetasizer. Microsphere dispersion was put into the sample cuvette and the particle size was recorded.
Percent Drug Entrapment
Prepared microspheres were further characterized for entrapment efficiency. In this method 50mg of microsphere was taken in a beaker containing 5.0 mL of phosphate buffer pH 7.4 and stored at room temperature for 24 hr to leach out drug from microsphere. Then microspheres were crushed by pestle and mortar to extract out complete drug from microsphere matrix. Solution was centrifuged for 15 min at 3000 rpm and 1.0 mL of supernatant was taken diluted upto 10 mL and analyzes the sample using UV spectrophotometer.
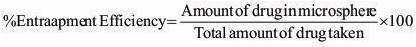
Invitro release studies
In vitro drug release from microsphere was carried out by using dialysis sac method. In whch microspheres equivalent to 100 mg of drug were weighed and transferred into cellophane membrane tubing MWCO 12000 Dalton (Himedia) containing 5.0 mL of phosphate buffer pH 7.4. This sac was transferred in a beaker containing 100 mL of same phosphate buffer pH 7.4. Flask was kept on the magnetic stirrer and the media continuously at 100 rpm and temperature was maintained at 37± 0.5oC. Then 1.0 mL of sample was withdrawn at suitable time interval using a hypodermic syringe attached with 0.45 μm membrane filter. Same volume of medium was replaced by phosphate buffer pH 7.4. After suitable dilution samples were analyzed on UV spectroscope at 276 nm.
Results and discussion
Preparation and Characterization of microspheres
The Microspheres were prepared using emulsification solvent evaporation method. HPMC, Sodium CMC and carbopol were used as a polymer blend to form microsphere. The microspheres were stabilized with the non-ionic surfactants Tween 80 and span-80. The composition of the microsphere significantly affects the physicochemical properties and drug release profiles of Microsphere. The studies of various microspheres consisting of different ratios of HPMC, Sodium CMC and carbopol describe the effect of these variables on the size, and drug entrapment (Table 1). At lower level of HPMC, Sodium CMC, the size was found to be less but particles were of irregular shape and on increasing the same the particle size increases and there is increase in uniformity and entrapment efficiency, but on further increasing the ratio of HPMC, Sodium CMC the size increases abruptly. The formulation F2 was found to be optimum with respect to various parameters as discussed earlier.
Shape and surface morphology
Shape and surface morphology are very critical factor when designing such novel formulation as these are responsible for the structural integrity and stability of the formulation. Surface morphology of the formulation was determined using SEM respectively as shown in figure 1a and b for without drug loaded and salbutamol loaded microsphere respectively. All the microsphere were found to be spherical with somewhat smooth in surface.
Table 1. Optimization of salbutamol laoded microsphere
|
Formulation code |
Polymer Ratio Carbopol:HPMC:NaCMC |
Drug (% w/w) |
% Yield |
Average Size (µm) |
Entrapment Efficiency (%) |
Zeta Potential |
|
F1 |
2:1:1 |
10 |
84.22±2.43 |
08.46±3.22 |
72.63±1.88 |
12.31±0.42 |
|
F2 |
4:1:1 |
10 |
88.26±3.09 |
12.14±2.53 |
80.15±1.02 |
20.45±0.28 |
|
F3 |
6:1:1 |
10 |
85.52±1.68 |
38.86±2.86 |
81.54±2.44 |
21.10±0.56 |
Particle size and zeta potential
To develop such controlled-release formulations particle size and Zeta potential are important parameter for stability of the formulation. Particle sizes and surface charges of the developed microsphere are shown in table 1. It was observed that the size of the optimized microsphere formulation was 12.14±2.53, which was optimum, when prepared with 1% surfactant (Tween 80). As depicted in table 1, the particle size of optimized microsphere when prepared with 4:1:1 ratio Carbopol: HPMC: NaCMC respectively was found to be 12.14±2.53 µm and zeta potentials of the microsphere was 20.45±0.28 mV. On increasing the concentration of Carbopol, i.e. 6:1:1 ratio of Carbopol: HPMC: NaCMC, particle size was increase.
Percent Drug Entrapment
The percent drug entrapment is an essential requirement for the fabrication of any drug delivery system. The results obtained in this study of salbutamol loaded Microsphere are shown in the table 1, which confirms good entrapment efficiency (80.15±1.02%) of optimized microsphere (F2). On increasing the ratio of carbopol, entrapment efficiency was increase but size of microsphere also increases.
In vitro drug release study
In order to estimate potential of the microsphere based gel formulation to retard the release of drug, it is necessary to calculate controlled release profile of the drug from the microsphere. The slow release profile may be the indicator of the temporal efficiency of the formulation. The release profile of salbutamol from microsphere formulations at different time interval are shown in the figure 2 and shows 52.16±1.76% in 24 h.
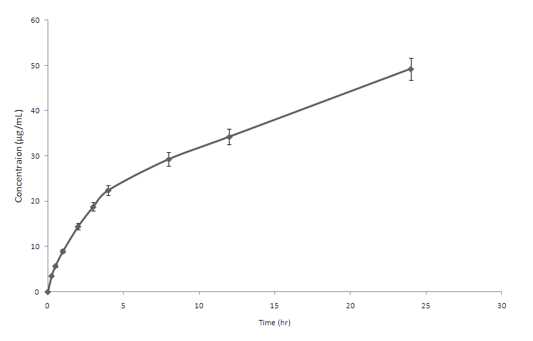
Figure 2. Cumulative percent drug release from salbutamol loaded microsphere
Conclusion
The microspheres of polymer blend (HPMC, sodium CMC and carbopol) were successfully prepared using emulsification solvent evaporation method in which salbutamol was loaded with high loading efficiency. Electron microscopy indicated that the salbutamol loaded microspheres were spherical in shape and smooth in surface. Size and zeta potential result shows that microsphere obtained by this method is in suitable in size and zeta potential represent good stability. The enhanced accumulation of salbutamol via microsphere and release drug well-controlled and sustained manner which will beneficial to manage asthma for longer period of time. The results obtained in this study reveal the merits of developed salbutamol loaded microsphere and justify their potential in strengthening the efficacy and safety of the drug as compared to existing conventional formulations in terms of lungs tolerability by patients. It is concluded that the prepared salbutamol loaded microsphere is a potential tool to deliver drug via nasal route for effective management of asthma.
Acknowledgement
One of the author want to acknowledge to Cadila Pharmaceutical Laboratory Pvt Ltd, Ahmedabad, Gujrat, india for providing Salbutamol as gift sample.
References
Amperiadou A, Georgarakis M. 1995. Controlled release salbutamol sulphate microcapsules prepared by emulsion solvent-evaporation technique and study on the release affected parameters. International Journal of Pharmaceutics, 115:1-8.
Celebi N, Erden N, Turkyilmaz A. 1996. The preparation and evaluation of salbutamol sulphate containing poly(lactic acid-co-glycolic acid) microspheres with factorial design-based studies. International Journal of Pharmaceutics, 136(1–2): 89–100.
Cevik GS. 2000. Sustained release properties of alginate microspheres and tabletted microspheres of diclofenac sodium. Journal of Microencapsulation, 17 (5): 565‐575.
Cook RO, Pannu RK, Kellaway IW. 2005. Novel sustained release microspheres for pulmonary drug delivery. Journal of Control Release, 104 (1): 79‐90.
Desai K, Park H. 2005. Preparation of cross-linked chitosan microspheres by spray drying: effect of cross-linking agent on the properties of spray dried microspheres. Journal of Microencapsulation, 22 (4): 377–395.
Fu J, Fiegel J, Krauland E, Hanes J. 2002. New polymeric carriers for controlled drug delivery following inhalation/ injection. Biomaterials, 23: 4425-4433.
He P, Davis S, Illum L. 1999. Chitosan microspheres prepared by spray drying. International Journal of Pharmaceutics, 187: 53–65.
Khar RK, Mohapatra S, Barik BB. 2009. Design and characterization of controlled release matrix tablets of Salbutamol, Asian Journal of Pharmaceutical and Clinical Research, 2 (2): 5461.
Martinez FD. 2007. Genes, environments, development and asthma: a reappraisal. European Respiratory Journal, 29 (1): 179–84.
Chourasia MK, Jain SK. 2004. Potential of guar gum microspheres for target specific drug release to colon. Journal of Drug Targeting, 12 (7): 435-442.
Murthy SN, Hiremath SR. 2001. Formulation and evaluation of controlled-release transdermal Patches of Theophylline-Salbutamol Sulfate. Drug Development and Industrial Pharmacy, 27: 1057-1062.
Pachuau L, Sarkar S, Mazumder B. 2008. Formulation and evaluation of matrix microspheres for simultaneous delivery of salbutamol sulphate and theophylline. Tropical Journal of Pharmaceutical Research, 7(2): 995-1002.
Prabakaran D, Singh P, Kanaujia P, Jaganathan KS, Rawat A, Vyas SP. 2004. Modified push-pull osmotic system for simultaneous delivery of theophylline and salbutamol: development and in vitro characterization. International Journal of Pharmaceutics, 284: 95-108.
Jain SK, Chourasia MK, Jain AK, Jain RK, Shrivastava AK. 2004. Development and characterization of mucoadhesive microspheres bearing salbutamol for nasal delivery. Drug Delivery, 11 (2): 113-122.
Young HR, Shin CS. 2006. Preparation and characterization of alginate carrageenan complex films. Journal of Applied Polymer Science, 99 (6): 3483‐3490.